
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4.9, Problem 30P
What is the configuration of the following compounds? (Use the given structures to answer the question.)
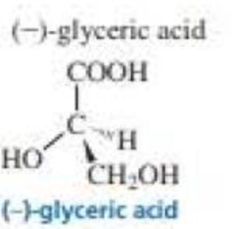
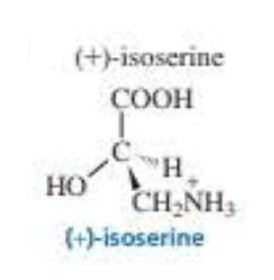
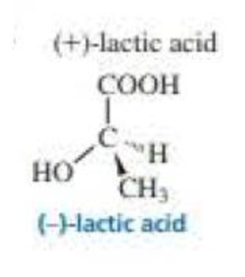
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the final product when D-galactose reacts with hydroxylamine?
Indicate the formula of the product obtained by reacting methyl 5-chloro-5-oxopentanoate with 1 mole of 4-penten-1-ylmagnesium bromide.
In the two chair conformations of glucose, the most stable is the one with all the OH groups in the equatorial position. Is this correct?
Chapter 4 Solutions
Organic Chemistry (8th Edition)
Ch. 4.1 - Prob. 2PCh. 4.1 - Prob. 3PCh. 4.1 - Prob. 4PCh. 4.1 - Prob. 5PCh. 4.1 - Which of the roll owing compounds have a dipole...Ch. 4.2 - Draw and label the E and Z isomers for each of the...Ch. 4.2 - Assign relative priorities to each set of...Ch. 4.2 - Tamoxifen slows the growth of some breast tumors...Ch. 4.2 - Prob. 10PCh. 4.2 - Name each of the following:
Ch. 4.2 - Draw the Z isomer of an alkene that has a CH3 and...Ch. 4.3 - Prob. 13PCh. 4.4 - Prob. 14PCh. 4.5 - Prob. 16PCh. 4.6 - Prob. 17PCh. 4.7 - Prob. 18PCh. 4.8 - Prob. 20PCh. 4.8 - Prob. 22PCh. 4.8 - Prob. 23PCh. 4.8 - Prob. 24PCh. 4.8 - Draw a perspective formula for each or the...Ch. 4.8 - Prob. 27PCh. 4.9 - Prob. 28PCh. 4.9 - What is the configuration of the following...Ch. 4.9 - Prob. 32PCh. 4.10 - Prob. 33PCh. 4.10 - (S)-(+)-Monosodium glutamate (MSG) is a flavor...Ch. 4.11 - Prob. 35PCh. 4.11 - Prob. 36PCh. 4.12 - Prob. 38PCh. 4.12 - Prob. 39PCh. 4.12 - The stereoisomer of cholesterol found in nature is...Ch. 4.12 - Prob. 41PCh. 4.13 - 1-Bromo-2-methylcyclopentane has four pairs of...Ch. 4.13 - Prob. 43PCh. 4.13 - Draw all possible stereoisomers for each of the...Ch. 4.13 - Prob. 45PCh. 4.13 - Of all the possible cyclooctanes that have one...Ch. 4.13 - Prob. 47PCh. 4.13 - Prob. 48PCh. 4.14 - Which of the following compounds has a...Ch. 4.14 - Draw all the stereoisomers for each of the...Ch. 4.15 - Prob. 52PCh. 4.15 - Name the isomers you drew in Problem 52.Ch. 4.15 - Chloramphenicol is a broad-spectrum antibiotic...Ch. 4.15 - Draw a perspective formula for each of the...Ch. 4.15 - Name the following:Ch. 4.15 - Prob. 57PCh. 4.15 - Prob. 59PCh. 4.15 - Convert the perspective formula to a skeletal...Ch. 4.15 - Prob. 62PCh. 4.16 - Prob. 63PCh. 4.17 - Limonene exists as two different stereoisomers....Ch. 4 - a. Draw three constitutional isomers with...Ch. 4 - Prob. 65PCh. 4 - Prob. 66PCh. 4 - Which of the following has an asymmetric center?...Ch. 4 - Prob. 68PCh. 4 - Prob. 69PCh. 4 - Prob. 70PCh. 4 - Prob. 71PCh. 4 - Assign relative priorities to each set of...Ch. 4 - Prob. 73PCh. 4 - Which of the following are optically active?Ch. 4 - Prob. 75PCh. 4 - Name the following:Ch. 4 - Which of the following has an achiral...Ch. 4 - Using skeletal structures, draw the stereoisomers...Ch. 4 - Prob. 79PCh. 4 - Citrate synthase, one of the enzymes in the series...Ch. 4 - Prob. 81PCh. 4 - Prob. 82PCh. 4 - Prob. 83PCh. 4 - Prob. 84PCh. 4 - Prob. 85PCh. 4 - Prob. 86PCh. 4 - Prob. 87PCh. 4 - Prob. 88PCh. 4 - Prob. 89PCh. 4 - a. Draw all the isomers with molecular formula...Ch. 4 - Prob. 91PCh. 4 - Prob. 92PCh. 4 - Draw structures for the following: a....Ch. 4 - For each of the following structures, draw the...Ch. 4 - Prob. 95PCh. 4 - Prob. 96PCh. 4 - Prob. 97PCh. 4 - a. Using the wedge-and-dash notation, draw the...Ch. 4 - Prob. 99PCh. 4 - Prob. 100PCh. 4 - Prob. 101PCh. 4 - a. Draw the two chair conformers for each of the...Ch. 4 - Prob. 103PCh. 4 - Is the following compound optically active?Ch. 4 - Prob. 105P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- please help me with my homeworkarrow_forwardhelparrow_forwardThe temperature on a sample of pure X held at 1.25 atm and -54. °C is increased until the sample boils. The temperature is then held constant and the pressure is decreased by 0.42 atm. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 2 0 0 200 400 temperature (K) Xarrow_forward
- QUESTION: Answer Question 5: 'Calculating standard error of regression' STEP 1 by filling in all the empty green boxes *The values are all provided in the photo attached*arrow_forwardpressure (atm) 3 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 0 0 200 temperature (K) 400 аarrow_forwarder your payment details | bar xb Home | bartleby x + aleksogi/x/isl.exe/1o u-lgNskr7j8P3jH-1Qs_pBanHhviTCeeBZbufuBYT0Hz7m7D3ZcW81NC1d8Kzb4srFik1OUFhKMUXzhGpw7k1 O States of Matter Sketching a described thermodynamic change on a phase diagram 0/5 The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. pressure (atm) 1 3- 0- 0 200 Explanation Check temperature (K) 400 X Q Search L G 2025 McGraw Hill LLC. All Rights Reserved Terms of Use Privacy Cearrow_forward
- 5.arrow_forward6.arrow_forward0/5 alekscgi/x/sl.exe/1o_u-IgNglkr7j8P3jH-IQs_pBaHhvlTCeeBZbufuBYTi0Hz7m7D3ZcSLEFovsXaorzoFtUs | AbtAURtkqzol 1HRAS286, O States of Matter Sketching a described thermodynamic change on a phase diagram The pressure on a sample of pure X held at 47. °C and 0.88 atm is increased until the sample condenses. The pressure is then held constant and the temperature is decreased by 82. °C. On the phase diagram below draw a path that shows this set of changes. 3 pressure (atm) + 0- 0 5+ 200 temperature (K) 400 Explanation Check X 0+ F3 F4 F5 F6 F7 S 2025 McGraw Hill LLC All Rights Reserved. Terms of Use Privacy Center Accessibility Q Search LUCR + F8 F9 F10 F11 F12 * % & ( 5 6 7 8 9 Y'S Dele Insert PrtSc + Backsarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY