
Inside a distillation column (see Problem 4.8), a downward-flowing liquid and an upward-flowing vapor maintain contact with each other. For reasons we will discuss in greater detail in Chapter 6. the vapor stream becomes increasingly rich in the more volatile components of the mixture as it moves up the column, and the liquid stream is enriched in the less volatile components as it moves down. The vapor leaving the lop of the column goes to a condenser. A portion of the condensate is taken off as a product (the overhead product), and the remainder (the reflux) is returned to the top of the column to begin its downward journey as the liquid stream. The condensation process can be represented as shown below:
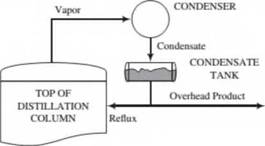
A distillation column is being used to separate a liquid mixture of ethanol (more volatile) and water (less volatile). A vapor mixture containing 89.0 moleSt ethanol and the balance water enters the overhead condenser at a rate of 100 lb-mole/h. The liquid condensate has a density of 49.0 lbnl/ft’. and
the reflux ratio is 3 lbm reflux/lbm overhead product. When the system is operating at steady state, the tank collecting the condensate is half full of liquid and the mean residence time in the tank (volume of liquid/volumetrie flow rate of liquid) is 10.0 minutes. Determine the overhead product volumetric flow rate (ft3/min) and the condenser tank volume (gal).
Trending nowThis is a popular solution!
Learn your wayIncludes step-by-step video

Chapter 4 Solutions
ELEM.PRIN.OF CHEMICAL PROC.-W/ACCESS
Additional Engineering Textbook Solutions
Java How to Program, Early Objects (11th Edition) (Deitel: How to Program)
Degarmo's Materials And Processes In Manufacturing
Computer Science: An Overview (13th Edition) (What's New in Computer Science)
Electric Circuits. (11th Edition)
Management Information Systems: Managing The Digital Firm (16th Edition)
Java: An Introduction to Problem Solving and Programming (8th Edition)
- A thick nickel wall is exposed to pure 5 bar H2(g) at 85 oC on one side of its surface (13 pts).(a) Assuming thermodynamic gas-solid equilibrium, calculate the H2 concentration at the surface ofthe nickel wall. (b) Assuming that the concentration of H2 at the surface is constant, determine the concentration ofH2 at the penetration depth in percentage of its concentration at the wall surfacearrow_forwardCan you provide me the answer of these pleasearrow_forwardA constant-volume process involving 2.0 moles of diatomic ideal gas, for which cV = (5/2)R, is at an initial state of 111 kPa and 277 K and is then heated reversibly to 356 K. Calculate the P2 (kPa), dU (kJ), heat transfer q (kJ mol–1), and w (kJ mol–1).arrow_forward
- Problem 1 Marks: 60 Section: 1a): 30 marks, Section 1b):30 marks A laboratory scale fluidized bed is considered for studying a catalytic ozone decomposition. a) It is requested to derive model equations under the following assumptions: ■ Operation of the catalytic reactor under steady state conditions, There is no influence of thermal ozone decomposition reactions. The fluidized bed includes bubbles and dense phase. □ The dense phase can be simulated using a CSTR The fluidized bed bubbles contain catalyst particles and can be simulated as a DSTR (batch). □ The jets contain particles and can be simulated with a PFR. The influence of the freeboard has to be considered using a PFR model. The available catalytic reaction rate model is r (moles/gcat.s)= -k CA b) Same than on a) under unsteady state conditions, using an absorbable and reactive tracer. Note: A step-by step derivation of the model equations is required here. A quick answer will not do. Problem 2 Marks: 40 Section 2a: 30 marks,…arrow_forwardPenicillin process Penicillium chrysogenum is used to produce penicillin in a 90,000-litre fermenter. The volumetric rate of oxygen uptake by the cells ranges from 0.45 to 0.85 mmol l^−1 min^−1 depending on time during the culture. Power input by stirring is 2.9 W l^−1. Estimate the cooling requirements.arrow_forwardProduction of bakers’ yeast Bakers’ yeast is produced in a 50,000-litre fermenter under aerobic conditions. The carbon substrate is sucrose; ammonia is provided as the nitrogen source. The average biomass composition is CH_1.83O_0.55N_0.17 with 5% ash. Under conditions supporting efficient growth, biomass is the only major product and the biomass yield from sucrose is 0.5 g g^−1. If the specific growth rate is 0.45 h^−1, estimate the rate of heat removal required to maintain constant temperature in the fermenter when the yeast concentration is 10 g l^−1.arrow_forward
- Sensible energy change Calculate the enthalpy change associated with the following processes:(a) m-Cresol is heated from 25°C to 100°C(b) Ethylene glycol is cooled from 20°C to 10°C(c) Succinic acid is heated from 15°C to 120°C(d) Air is cooled from 150°C to 65°Carrow_forward▼ Enzyme conversion An immobilised enzyme process is used in an ice-cream factory to hydrolyse lactose (C12H22O11) to glucose (C6H12O6) and galactose (C6H1206): C12H22O11 + H2O →→ C6H12O + C6H12O6 Gel beads containing ß-galactosidase are packed into a column reactor; 2500 kg of lactose enters the reactor per day as a 10% solution in water at 25°C. The reactor operates at steady state and 32°C; all of the lactose is converted. Because the heat of reaction for enzyme conversions is not as great as for cell culture, sensible heat changes and heats of mixing cannot be ignored. Lactose Water Ah (kJ gmol¹) C, (cal g¹ ºC-¹) Ahm (kcal gmol¹) 3.7 -5652.5 0.30 1.0 Glucose -2805.0 0.30 5.6 Galactose -2805.7 0.30 5.6 (a) What is the standard heat of reaction for this enzyme conversion? (b) Estimate the heating or cooling requirements for this process. State explicitly whether heating or cooling is needed.arrow_forwardBacterial production of alginate Azotobacter vinelandii is investigated for production of alginate from sucrose. In a continuous fermenter at 28°C with ammonia as nitrogen source, the yield of alginate was found to be 4 g g^−1 oxygen consumed. It is planned to produce alginate at a rate of 5 kg h^−1. Since the viscosity of alginate in aqueous solution is considerable, energy input due to mixing the broth cannot be neglected. The fermenter is equipped with a flat-blade disc turbine; at a satisfactory mixing speed and air flow rate, the power requirements are estimated at 1.5 kW. Calculate the cooling requirements.arrow_forward
- Preheating nutrient medium Steam is used to heat nutrient medium in a continuous-flow process. Saturated steam at 150°C enters a coil on the outside of the heating vessel and is completely condensed. Liquid medium enters the vessel at 15°C and leaves at 44°C. Heat losses from the jacket to the surroundings are estimated as 0.22 kW. If the flow rate of medium is 3250 kg h^−1 and its heat capacity is 0.9 cal g^−1 °C^−1, how much steam is required?arrow_forwardQ3] Determine the optimal operating conditions (XA, t, and CR) in a mixed flow reactor to maximize the concentration of R (CR) in the effluent, where an aqueous feed A with an initial concentration of CA0-40 mol/m³ enters the reactor, undergoes decomposition, and exits as a mixture containing A, R, and S. K₁ AR, FR = k₁C, k₁ = 0.4 m³/(mol min) SA AS, rs = k₂CA, k₂ = 2(min), CA0 = 40 mol/m³arrow_forwardConsider the parallel decomposition of A of different orders with FR = 1, rs = 2CA and IT = C. Determine the maximum concentration of desired product obtainable in mixed flow reactor and plug flow reactor. (1) R is desired product and CA0 = 2. (2) S is desired product and CA0 = 4. R S Tarrow_forward
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The





