
Concept explainers
Hydrocarbons like benzene are
phenols. This is an example of a general process in the body, in which an unwanted compound
(benzene) is converted to a more water-soluble derivative called a metabolite, so that it can be
excreted more readily from the body.
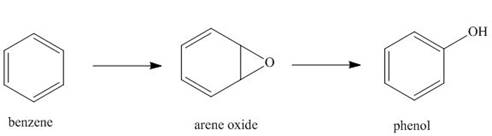
a. Classify each of these reactions as oxidation, reduction, or neither.
b. Explain why phenol is more water soluble than benzene. This means that phenol dissolves in urine, which is largely water, to a greater extent than benzene.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
Additional Science Textbook Solutions
Organic Chemistry - Standalone book
General, Organic, & Biological Chemistry
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Chemistry & Chemical Reactivity
Organic Chemistry As a Second Language: Second Semester Topics
Basic Chemistry (5th Edition)
- How many water molecules would be produced by the complete combustion of one straight chained butane molecule? A. 4 B. 5 C. 6 D. 8arrow_forwardThe oxidation products of ethylene glycol (glycoaldehyde, glycolic acid, and oxalic acid) cause a variety of problems in the body. What happens to the oxalic acid? Which organs are damaged?arrow_forward23. What reactant and catalyst is(are) needed to change ethene into ethane? a. H2, Pt Ob. H20, H2SO4 C. HCI d. heat, pressurearrow_forward
- What is the name of the alcohol produced by the fermentation of sugars and starches and container in alcoholic beverages A.Methyl alcohol B.Ethanol C. Isopropyl alcohol D. Methanol E. Propanolarrow_forwardMULTIPLE CHOICE 1. They impart fragrance of fruit and flowers. * A. alcohol B. ether C. carboxylic acid D. ester 2. They are the final oxidation product of primary alcohol. * A. ester B. aldehyde C. carboxylic acid D. ketonearrow_forward11) Classify the reaction below as an oxidation, a reduction, or neither. cis-2-pentene · → pentane A) oxidation B) reduction C) neitherarrow_forward
- What does the catalytic converter promote combustion?arrow_forwardAcid hydrolysis of ------------ produces carboxylic acids. a. Cyanides b. Alcohol c. Ester d. Hydrocarbonarrow_forward9. What is the major organic product obtained from the following reaction? a. 1 b. 2 c. 3 d. 4 OH H₂SO4 3 SH 2arrow_forward
- Write a balanced chemical equation for the complete combustion of benzenearrow_forwardWhat hydrocarbons enter the atmosphere from internal combustion engines? A. Carboxylics B. Alkynes C. Alkenes D. Alkyl hydrocarbonsarrow_forwardBased on the image attached, identify the product form in the reduction reaction of methyl salicylate. Give an explanation of the reaction and how the product form.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning  Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



