
CHEMISTRY-TEXT
8th Edition
ISBN: 9780134856230
Author: Robinson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 4.40CP
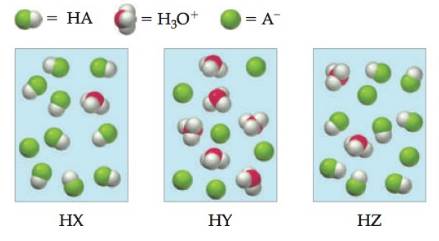
The following pictures represent aqueous solutions of three acids HA (A = X, Y, or Z), with surrounding water molecules omitted for clarity. Which of the three is the strongest acid, and which is the weakest?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Diels Alder Cycloaddition: Focus on regiochemistry (problems E-F) –> match + of thedienophile and - of the diene while also considering stereochemistry (endo).
HELP! URGENT! PLEASE RESOND ASAP!
Question 4
Determine the rate order and rate constant for sucrose hydrolysis.
Time (hours)
[C6H12O6]
0
0.501
0.500
0.451
1.00
0.404
1.50
0.363
3.00
0.267
First-order, k = 0.210 hour 1
First-order, k = 0.0912 hour 1
O Second-order, k =
0.590 M1 hour 1
O Zero-order, k = 0.0770 M/hour
O Zero-order, k = 0.4896 M/hour
O Second-order, k = 1.93 M-1-hour 1
10 pts
Chapter 4 Solutions
CHEMISTRY-TEXT
Ch. 4 - Prob. 4.1PCh. 4 - Prob. 4.2ACh. 4 - How many moles of solute are present in 125 mLof...Ch. 4 - Prob. 4.4ACh. 4 - Prob. 4.5PCh. 4 - APPLY 4.7 Sulfuric acid is normally purchased at a...Ch. 4 - Prob. 4.7PCh. 4 - Conceptual APPLY 4.9 Three different substances,...Ch. 4 - Write ne ionic equations for the following...Ch. 4 - Prob. 4.10A
Ch. 4 - Will a precipitation reaction occur when aqueous...Ch. 4 - APPLY 4.13 How might you use a precipitation...Ch. 4 - Conceptual PRACTICE 4.14 An aqueous solution...Ch. 4 - Conceptual APPLY 4.15 A solution containing the...Ch. 4 - Name the acids HI and HBrO2 .Ch. 4 - APPLY 4.17 Give likely chemical formulas...Ch. 4 - PRACTICE 4.18 Write a balanced ionic equation and...Ch. 4 - Milk of magnesia (active ingredient: magnesium...Ch. 4 - Prob. 4.19PCh. 4 - Prob. 4.20ACh. 4 - Prob. 4.21PCh. 4 - Assume that the buret contains H+ ions, the flask...Ch. 4 - PRACTICE 4.24 Assign an oxidation number to each...Ch. 4 - APPLY 4.25 Chlorine can have several different...Ch. 4 - Identify the oxidizing agent and reducing agent in...Ch. 4 - APPLY 4.27 Police often use a Breathalyzer test to...Ch. 4 - PRACTICE 4.28 Predict whether the following...Ch. 4 - Use the following reactions to arrange the...Ch. 4 - Prob. 4.29PCh. 4 - Prob. 4.30ACh. 4 - A viramin-fortified brand of a sports...Ch. 4 - The nutritional label on Power ade specifies that...Ch. 4 - Prob. 4.33PCh. 4 - One way to analyze a sports drink for the...Ch. 4 - To measure the concentration of chloride ions ina...Ch. 4 - The flavor of the first batch of Gatorade was...Ch. 4 - Prob. 4.37CPCh. 4 - Prob. 4.38CPCh. 4 - Assume that an aqueous solution Of a cation,...Ch. 4 - The following pictures represent aqueous solutions...Ch. 4 - Prob. 4.41CPCh. 4 - The concentration of an aqueous solution of NaOCl...Ch. 4 - Assume that the electrical conductivity of a...Ch. 4 - Based on the positions in the periodic table,...Ch. 4 - The following two redox reactions occur between...Ch. 4 - Prob. 4.46SPCh. 4 - How many grams of solute would you use to prepare...Ch. 4 - How many milliliters of a 0.45 M BaCl2 solution...Ch. 4 - How many milliliters of a 0.350 M KOH solution...Ch. 4 - The sterile saline solution used to rinse contact...Ch. 4 - Prob. 4.51SPCh. 4 - Copper reacts with dilute nitric acid according to...Ch. 4 - Prob. 4.53SPCh. 4 - How many grams of solute would you use to prepare...Ch. 4 - Prob. 4.55SPCh. 4 - How would you prepare 250 mL of a 0.100 M solution...Ch. 4 - Pennies minted after 1982 are mostly zinc (97.5%)...Ch. 4 - A bottle of 12.0 M hydrochloric acid has only 35.7...Ch. 4 - What is the volume of the solution that would...Ch. 4 - How would you prepare 250 mL of a 0.100 M solution...Ch. 4 - Prob. 4.61SPCh. 4 - The following aqueous solutions were tested with a...Ch. 4 - Prob. 4.63SPCh. 4 - Individual solutions of Ba(OH)2 and H2SO4 both...Ch. 4 - A solution of HCl in water conducts electricity,...Ch. 4 - Classify each of the following substances as a...Ch. 4 - Is it possible for a molecular substance to be a...Ch. 4 - What is the total molar concentration of ions in...Ch. 4 - What is the total molar concentration of ions in...Ch. 4 - Ringer’s solution, used in the treatment of burns...Ch. 4 - What is the molarity of each ion in a solution...Ch. 4 - Prob. 4.72SPCh. 4 - Prob. 4.73SPCh. 4 - Write net ionic equations for the reactions listed...Ch. 4 - Write net ionic equations for the reactions listed...Ch. 4 - Which of the following substances are likely to be...Ch. 4 - Which of the following substances are likely to be...Ch. 4 - Predict whether a precipitation reaction will...Ch. 4 - Predict whether a precipitation reaction will...Ch. 4 - hich of the following solutions will not form a...Ch. 4 - Which of the following solutions will not form a...Ch. 4 - How would you prepare the following substances by...Ch. 4 - How would you prepare the following substances by...Ch. 4 - What are the mass and the identity of the...Ch. 4 - What are the mass and the identity of the...Ch. 4 - Assume that you have an aqueous mixture of NaNO3...Ch. 4 - Assume that you have an aqueous mixture of BaCl2...Ch. 4 - Assume that you have an aqueous solution of an...Ch. 4 - Assume that you have an aqueous solution of an...Ch. 4 - How could you use a precipitation reaction to...Ch. 4 - How could you u a precipitation reaction to...Ch. 4 - The following three solutions are mixed: 100.0 mL...Ch. 4 - A 250.0 g sample of a white solid is known to be a...Ch. 4 - Assume that you are given a solution of an unknown...Ch. 4 - Why do we use a double arrow () to show the...Ch. 4 - Write balanced ionic equations for the following...Ch. 4 - Write balanced ionic equations for the following...Ch. 4 - Write balanced net ionic equations for the...Ch. 4 - Write balanced net ionic equations for the...Ch. 4 - A flask containing 450 mL of 0.500 M HBr was...Ch. 4 - The odor of skunks is caused by chemical compounds...Ch. 4 - Prob. 4.102SPCh. 4 - Prob. 4.103SPCh. 4 - How many milliliters of 1.00 M KOH must be added...Ch. 4 - How many milliliters of 2.00 M HCl must be added...Ch. 4 - If the following solutions are mixed, is the...Ch. 4 - If the following solutions are mixed, is the...Ch. 4 - Prob. 4.108SPCh. 4 - Assign oxidation numbers to each element in the...Ch. 4 - Assign oxidation numbers to each element in the...Ch. 4 - Assign oxidation numbers to each element in the...Ch. 4 - Nitrogen can have several different oxidation...Ch. 4 - Prob. 4.113SPCh. 4 - Prob. 4.114SPCh. 4 - Where in the periodic table are the most easily...Ch. 4 - In each of the following instances, tell whether...Ch. 4 - Tell for each of the following substances whether...Ch. 4 - Which element is oxidized and which is reduced in...Ch. 4 - Which element is oxidized and which is reduced in...Ch. 4 - Use the activity series of metals (Table 4.5) to...Ch. 4 - Neither strontium (Sr) nor antimony (Sb) is shown...Ch. 4 - (a) Use the following reactions to arrange the...Ch. 4 - Prob. 4.123SPCh. 4 - Iodine, I2 , reacts with aqueous thiosulfate ion...Ch. 4 - How many milliliters of 0.250MNa2S2O3 solution is...Ch. 4 - Dichromate ion, Cr2O72 reacts with aqueous...Ch. 4 - A volume of 18.72 mL of 0.1500MK2Cr2O7 solution...Ch. 4 - What is the molar concentration of As(III) in a...Ch. 4 - Standardized solutions of KBrO3 are frequently...Ch. 4 - The metal content of iron in ores can be...Ch. 4 - Prob. 4.131SPCh. 4 - Alcohol levels in blood can he determined by a...Ch. 4 - Calcium levels in blood can be determined by...Ch. 4 - Assume that you have 1.00 g of a mixture of...Ch. 4 - A compound with the formula XOCl2 , reacts with...Ch. 4 - Prob. 4.136MPCh. 4 - Some metals occur naturally in their elemental...Ch. 4 - A sample weighing 14.98 g and containing a small...Ch. 4 - The solubility of an ionic compound can be...Ch. 4 - Prob. 4.140MPCh. 4 - Prob. 4.141MPCh. 4 - Prob. 4.142MPCh. 4 - A sample of metal (M) reacted with both steam and...Ch. 4 - Prob. 4.144MPCh. 4 - Prob. 4.145MPCh. 4 - Iron content in ores can be determined by a redox...Ch. 4 - Prob. 4.147MPCh. 4 - Salicylic acid, used in the manufacture of...Ch. 4 - Compound X contains only the elements C, H, O, and...Ch. 4 - Prob. 4.150MPCh. 4 - Element M is prepared industrially by a two-step...Ch. 4 - Prob. 4.152MPCh. 4 - Four solutions are prepared and mixed in the...Ch. 4 - To 100.0 mL of a solution that contains 0.120MCr(...Ch. 4 - Sodium nitrite, NaNO2 , is frequently added to...Ch. 4 - Brass is an approximately 4:1 alloy of copper and...Ch. 4 - Prob. 4.157MPCh. 4 - Prob. 4.158MP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Determine the rate order and rate constant for sucrose hydrolysis. Time (hours) [C6H12O6] 0 0.501 0.500 0.451 1.00 0.404 1.50 0.363 3.00 0.267arrow_forwardDraw the products of the reaction shown below. Use wedge and dash bonds to indicate stereochemistry. Ignore inorganic byproducts. OSO4 (cat) (CH3)3COOH Select to Draw ઘarrow_forwardCalculate the reaction rate for selenious acid, H2SeO3, if 0.1150 M I-1 decreases to 0.0770 M in 12.0 minutes. H2SeO3(aq) + 6I-1(aq) + 4H+1(aq) ⟶ Se(s) + 2I3-1(aq) + 3H2O(l)arrow_forward
- Problem 5-31 Which of the following objects are chiral? (a) A basketball (d) A golf club (b) A fork (c) A wine glass (e) A spiral staircase (f) A snowflake Problem 5-32 Which of the following compounds are chiral? Draw them, and label the chirality centers. (a) 2,4-Dimethylheptane (b) 5-Ethyl-3,3-dimethylheptane (c) cis-1,4-Dichlorocyclohexane Problem 5-33 Draw chiral molecules that meet the following descriptions: (a) A chloroalkane, C5H11Cl (c) An alkene, C6H12 (b) An alcohol, C6H140 (d) An alkane, C8H18 Problem 5-36 Erythronolide B is the biological precursor of erythromycin, a broad-spectrum antibiotic. How H3C CH3 many chirality centers does erythronolide B have? OH Identify them. H3C -CH3 OH Erythronolide B H3C. H3C. OH OH CH3arrow_forwardPLEASE HELP! URGENT! PLEASE RESPOND!arrow_forward2. Propose a mechanism for this reaction. ہلی سے ملی N H (excess)arrow_forward
- Steps and explanationn please.arrow_forwardProblem 5-48 Assign R or S configurations to the chirality centers in ascorbic acid (vitamin C). OH H OH HO CH2OH Ascorbic acid O H Problem 5-49 Assign R or S stereochemistry to the chirality centers in the following Newman projections: H Cl H CH3 H3C. OH H3C (a) H H H3C (b) CH3 H Problem 5-52 Draw the meso form of each of the following molecules, and indicate the plane of symmetry in each: OH OH (a) CH3CHCH2CH2CHCH3 CH3 H3C. -OH (c) H3C CH3 (b) Problem 5-66 Assign R or S configurations to the chiral centers in cephalexin, trade-named Keflex, the most widely prescribed antibiotic in the United States. H2N H IHH S Cephalexin N. CH3 CO₂Harrow_forwardSteps and explanationn please.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY