
Concept explainers
(a)
Interpretation:
To identify the reactant which reacts with dilute hydrochloric acid
Concept introduction:
Nucleophiles are those species which are electron rich in nature. They are negatively charged. Carboxylate ion is a very good nucleophile. So, it can reacts with dilute
(b)
Interpretation:
To identify the reactant that undergoes hydrolysis.
Concept introduction:
The structure of ester is,
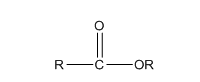
The ester undergoes hydrolysis reaction to form
(c)
Interpretation:
To identify the reactant that neutralizses dilute NaOH.
Concept introduction:
The reaction of an acid and base is called neutralization reaction. The products of neutralization reaction are salt and water. So, the reactant that can neutralize
Want to see the full answer?
Check out a sample textbook solution
Chapter 27 Solutions
EP GENERAL CHEMISTRY-MOD.MASTERINGCHEM.
- (ME EX2) Problems 12-14 Could you please explain problems 12 through 14 to me in detail, step by step? Thank you so much! If necessary, please color-code them for me.arrow_forwardCan you explain problems 9-11 for me step by step. Potentially color code for instance the resonance structuresarrow_forward(SE EX 2) Problems 12-14, can you please explain them to me in detail and color-code anything if necessary?arrow_forward
- (ME EX2) Prblms 5-7 Can you please explain problems 12-14 to me in detail, step by step? Thank you so much! If needed color code them for me.arrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forwardCan you explain problems 12-16 for me please. (step by step) and in detailarrow_forward
- please solve this question. It is very sensentive with spelling and characters so if it is "no errors" please be specific. thanks!arrow_forwardDon't used hand raiting and don't used Ai solutionarrow_forwardH2(g) + I2(g) ⇔ 2HI(g) Using the above equilibrium, find the equilibrium concentration of H2 if the intial concentration of both H2 and I2 are 2.0. K at this temperature is 55.64.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



