
Concept explainers
(a)
Interpretation:
The structure for each of the given thioesters.
Concept Introduction:
Thioesters are sulfur analogs of esters. The S-alkyl or S-aryl thioester has a carbonyl group and a sulfur-linked alkyl or aryl group. The first part of the name indicates that the alkyl or aryl group has bonded to the sulfur atom. The last part of the name indicates the
Answer to Problem 25.1P
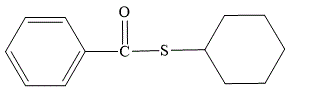
Explanation of Solution
In the given name cyclohexyl thiobenzoate, the cyclohexyl is the alkyl group which is bonded to the sulfur atom. Thiobenzoate consists of a phenyl ring bonded to the sulfur atom through a carbonyl group. The structure for cyclohexyl thiobenzoate is drawn as below:
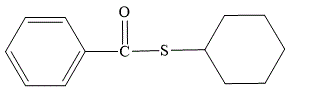
The structure for cyclohexyl thiobenzoate is:
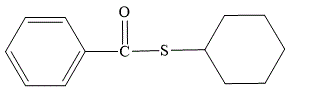
(b)
Interpretation:
The structure for each of the given thioesters.
Concept Introduction:
Thioesters are sulfur analogs of esters. The S-alkyl or S-aryl thioester has a carbonyl group and a sulfur-linked alkyl or aryl group. The first part of the name indicates that the alkyl or aryl group has bonded to the sulfur atom. The last part of the name indicates the carboxylic acid stem. In common usage, the “S-“prefix is understood, and so, it is not mentioned.
Answer to Problem 25.1P
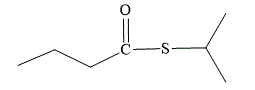
Explanation of Solution
The first part of the name suggests that the isopropyl group must be attached directly to the sulfur atom. The second part of the name suggests that the ester is formed from butanoic acid. In

The structure for S-isopropyl butanethioate is:

(c)
Interpretation:
The structure for each of the given thioesters.
Concept Introduction:
Thioesters are sulfur analogs of esters. The S-alkyl or S-aryl thioester has a carbonyl group and a sulfur-linked alkyl or aryl group. The first part of the name indicates that the alkyl or aryl group has bonded to the sulfur atom. The last part of the name indicates the carboxylic acid stem. In common usage, the “S-“prefix is understood, and so, it is not mentioned.
Answer to Problem 25.1P
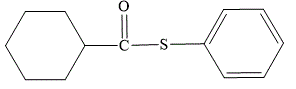
Explanation of Solution
The first part of the name suggests that the phenyl ring must be attached directly to the sulfur atom. The second part of the name suggests that the ester is formed from the carboxylic acid which contains a cyclohexyl ring. In IUPAC nomenclature, “thio-“is inserted before the “ate” suffix in the name of the corresponding oxygen ester.
The structure for S-phenyl cyclohexanecarbothioate is shown as below:

The structure for S-phenyl cyclohexanecarbothioate is:

Want to see more full solutions like this?
Chapter 25 Solutions
Organic Chemistry
- Predict the intermediate 1 and final product 2 of this organic reaction: NaOMe ག1, ད།་, - + H You can draw 1 and 2 in any arrangement you like. 2 work up Note: if either 1 or 2 consists of a pair of enantiomers, just draw one structure using line bonds instead of 3D (dash and wedge) bonds at the chiral center. Explanation Check Click and drag to start drawing a structure. Х © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Parrow_forwardWhat is the total energy cost associated with the compound below adopting the shown conformation? CH3 HH DH CH3arrow_forwardΗΝ, Draw Final Product C cyclohexanone pH 4-5 Edit Enamine H3O+ CH3CH2Br THF, reflux H Edit Iminium Ionarrow_forward
- How many hydrogen atoms are connected to the indicated carbon atom?arrow_forwardIdentify the compound with the longest carbon - nitrogen bond. O CH3CH2CH=NH O CH3CH2NH2 CH3CH2C=N CH3CH=NCH 3 The length of all the carbon-nitrogen bonds are the samearrow_forwardIdentify any polar covalent bonds in epichlorohydrin with S+ and 8- symbols in the appropriate locations. Choose the correct answer below. Η H's+ 6Η Η Η Η Η Ηδ Η Ο Ο HH +Η Η +Η Η Η -8+ CIarrow_forward
- H H:O::::H H H HH H::O:D:D:H HH HH H:O:D:D:H .. HH H:O:D:D:H H H Select the correct Lewis dot structure for the following compound: CH3CH2OHarrow_forwardRank the following compounds in order of decreasing boiling point. ннннн -С-С-Н . н-с- ННННН H ΗΤΗ НННН TTTĪ н-с-с-с-с-о-н НННН НН C' Н н-с-с-с-с-н НН || Ш НННН H-C-C-C-C-N-H ННННН IVarrow_forwardRank the following compounds in order of decreasing dipole moment. |>||>||| ||>|||>| |>|||>|| |||>||>| O ||>>||| H F H F H c=c || H c=c F F IIIarrow_forward
- choose the description that best describes the geometry for the following charged species ch3-arrow_forwardWhy isn't the ketone in this compound converted to an acetal or hemiacetal by the alcohol and acid?arrow_forwardWhat is the approximate bond angle around the nitrogen atom? HNH H Harrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
