
(a)
Interpretation: The aldol product that formed by the given compound is to be drawn.
Concept introduction: Aldol reaction is the condensation reaction of the
Answer to Problem 24.1P
The aldol product that formed by the given compound is
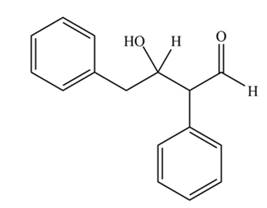
Explanation of Solution
The aldol product that formed by the given compound is shown below.
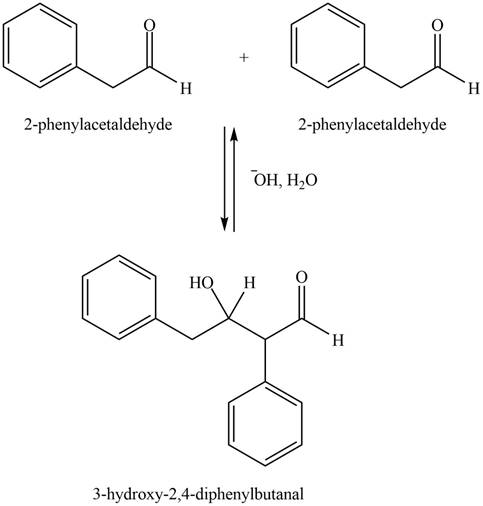
Figure 1
In this aldol reaction, one equivalent of
The aldol product that formed by the given compound is
(b)
Interpretation: The aldol product that formed by the given compound is to be drawn.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.1P
The aldol product that formed by the given compound is
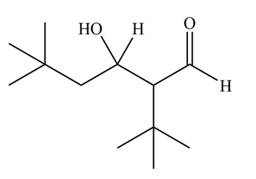
Explanation of Solution
The aldol product that formed by the given compound is shown below.
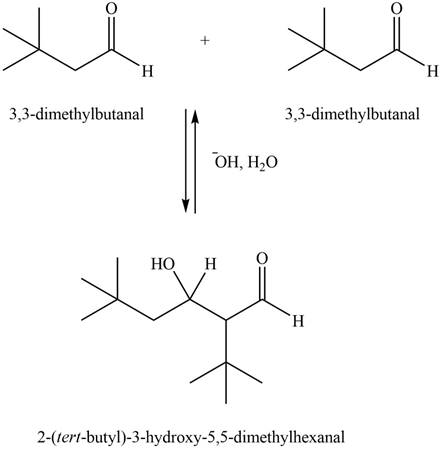
Figure 2
In this aldol reaction, one equivalent of
The aldol product that formed by the given compound is
(c)
Interpretation: The aldol product that formed by the given compound is to be drawn.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.1P
The aldol product that formed by the given compound is
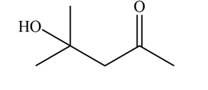
Explanation of Solution
The aldol product that formed by the given compound is shown below.
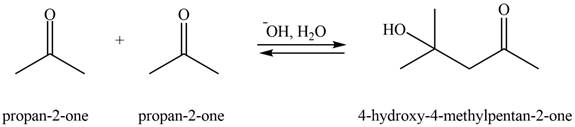
Figure 3
In this aldol reaction, one equivalent of
The aldol product that formed by the given compound is
(d)
Interpretation: The aldol product that formed by the given compound is to be drawn.
Concept introduction: Aldol reaction is the condensation reaction of the organic chemistry. In this reaction an enolate ion or an enol reacts with the carbonyl compound that leads to the formation of
Answer to Problem 24.1P
The aldol product that formed by the given compound is
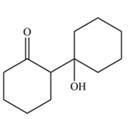
Explanation of Solution
The aldol product that formed by the given compound is shown below.
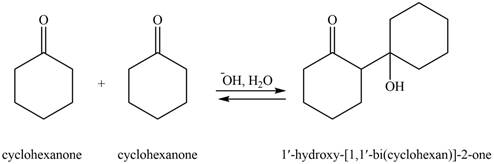
Figure 4
In this aldol reaction, one equivalent of cyclohexanone is treated with strong base that results in the formation of resonance-stabilized enolate ion. The second equivalent of cyclohexanone is then reacts with enolate ion. After that, hydrolysis of the intermediate compound takes place that results the formation of the desired product,
The aldol product that formed by the given compound is
Want to see more full solutions like this?
Chapter 24 Solutions
Organic Chemistry
- CH3CH2CH=CH2 + H₂O − H+arrow_forwardГ C-RSA CHROMATOPAC CH=1 DATA 1: @CHRM1.C00 ATTEN=10 SPEED= 10.0 0.0 b.092 0.797 1.088 1.813 C-RSA CHROMATOPAC CH=1 Report No. =13 ** CALCULATION REPORT ** DATA=1: @CHRM1.000 11/03/05 08:09:52 CH PKNO TIME 1 2 0.797 3 1.088 4 1.813 AREA 1508566 4625442 2180060 HEIGHT 207739 701206 V 287554 V MK IDNO CONC NAME 18.1447 55.6339 26.2213 TOTAL 8314067 1196500 100 C-R8A CHROMATOPAC CH=1 DATA 1: @CHRM1.C00 ATTEN=10 SPEED= 10.0 0. 0 087 337. 0.841 1.150 C-R8A CHROMATOPAC CH=1 Report No. =14 DATA=1: @CHRM1.000 11/03/05 08:12:40 ** CALCULATION REPORT ** CH PKNO TIME AREA 1 3 0.841 1099933 41.15 4039778 HEIGHT MK IDNO 170372 649997¯¯¯ CONC NAME 21.4007 78.5993 TOTAL 5139711 820369 100 3 C-R8A CHROMATOPAC CH=1 DATA 1: @CHRM1.C00 ATTEN=10 SPEED= 10.0 0.100 0:652 5.856 3 1.165 C-RSA CHROMATOPAC CH-1 Report No. =15 DATA=1: @CHRM1.000 11/03/05 08:15:26 ** CALCULATION REPORT ** CH PKNO TIME AREA HEIGHT MK IDNO CONC NAME 1 3 3 0.856 4 1.165 TOTAL 1253386 4838738 175481 708024 V 20.5739 79.4261 6092124…arrow_forwardIndicate the product that is obtained if the benzotriazole reacts with the use of a medium basic product.arrow_forward
- Indicate the product that is obtained if the benzotriazol reacts with dimethyl sulfate.arrow_forwardIndicate how to obtain 2-metilbencimidazol from 1,2-diaminobenzene.arrow_forwardbreak down both reactions shown and explain it correctly using the bromonium ion mechanism, instead of the (disproven) carbocation-based mechanism.arrow_forward
- Indicate how from 1,2-diaminobenzene to obtain 1-metilbenzotriazol.arrow_forward-C = C - C - + Br₂ + I" -> -C-C-c -C = C -C- + Br² + I₂ -C=C Br I + Brū + Iz -7- C - C-C- I Br Mechanism; - C = c - c - + Br - Br > - C-c-c- Br -C-C-C- + 1 - - -Ċ-Ċ'-c' - Br Br Iarrow_forwardWrite the mechanism of the esterification reaction (please show the mechanism included line pairs and arrows)arrow_forward
- How do I break down the reaction shown on the chalkboard and explain it correctly using the bromonium ion mechanism, instead of the (disproven) carbocation-based mechanismarrow_forward¿Qué the product is obtained from tetraethoxypropano and hidrazina?. Indicate the reason why the corresponding dial is used.arrow_forwardIf CH3COCH2CH(OCH3)2 is reacted with hydrazine, two isomeric products are formed. Indicate their structures and the major product.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
