
Organic Chemistry
7th Edition
ISBN: 9780321803221
Author: Paula Y. Bruice
Publisher: Prentice Hall
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2.3, Problem 9P
Draw the conjugate acid of each of the following:
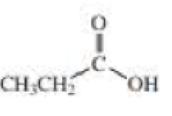
- a. CH3CH2OH
- b. CH3CH2O–
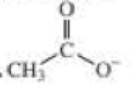
- c. CH3CH2NH2
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a Haworth projection of a common cyclic form of this monosaccharide:
H
HO
H
HO
H
HO
H
H
-OH
CH2OH
Click and drag to start drawing a
structure.
Х
:
D
:
Draw the structure of valylasparagine, a dipeptide made from valine and asparagine, as it would appear at physiological pH.
Click and drag to start drawing a
structure.
P
D
Draw the Haworth projection of α-L-mannose. You will find helpful information in the ALEKS Data resource.
Click and drag to start drawing a
structure.
:
ཊི
Х
D
Chapter 2 Solutions
Organic Chemistry
Ch. 2.1 - Which of the following are not acids? CH3COOH CO2...Ch. 2.1 - Draw the products of the addbase renc1 ion when a....Ch. 2.1 - a. What is the conjugate acid of each or the...Ch. 2.2 - a. Which is a stronger acid: one with a pKa of 5.2...Ch. 2.2 - An acid has a Ka of 4.53 106 in water. What is...Ch. 2.2 - Prob. 6PCh. 2.2 - Antacids are compounds that neutralize stomach...Ch. 2.2 - Are the following body fluids acidic or basic? a....Ch. 2.3 - Draw the conjugate acid of each of the following:...Ch. 2.3 - a. Write an equation showing CH3OH reacting as an...
Ch. 2.3 - Estimate the pKa values of the following...Ch. 2.3 - a. Which is a stronger base: CH3COO or HCOO? (The...Ch. 2.3 - Using the pKa values in Section 2.3, rank the...Ch. 2.4 - Prob. 14PCh. 2.5 - a. For each of the acid-base reactions in Section...Ch. 2.5 - Ethyne has a pKa value of 25, water has a pKa...Ch. 2.5 - Which of the following bases can remove a proton...Ch. 2.5 - Calculate the equilibrium constant for the...Ch. 2.6 - Rank the ions (CH3, NH2, HO, and F) from most...Ch. 2.6 - Rank the carbanions shown in the margin from most...Ch. 2.6 - Which is the stronger acid?Ch. 2.6 - Prob. 22PCh. 2.6 - Rank the halide ions (F, Cl, Br, and l) from...Ch. 2.6 - a. Which is more electronegative, oxygen or...Ch. 2.6 - Which is a stronger acid? a. HCl or HBr b....Ch. 2.6 - a. Which of the halide ions (F, Cl, Br, and l) is...Ch. 2.6 - Prob. 27PCh. 2.7 - What is a stronger acid? a. CH3OCH2CH2OH or...Ch. 2.7 - Prob. 29PCh. 2.7 - What is a stronger base?Ch. 2.8 - Fosamax (shown on the previous page) has six...Ch. 2.8 - Which is a stronger acid? Why?Ch. 2.8 - Prob. 34PCh. 2.9 - Using the table of pKa values given in Appendix I,...Ch. 2.10 - Prob. 36PCh. 2.10 - As long as the pH is not less than _______, at...Ch. 2.10 - A naturally occurring amino acid such as alanine...Ch. 2.10 - For each of the following compounds, indicate the...Ch. 2.11 - Write the equation that shows how a buffer made by...Ch. 2.12 - Draw the products of the following react ions. Use...Ch. 2.12 - What product are formed when each of the following...Ch. 2 - a. Rank the following alcohols from strongest to...Ch. 2 - Which is a stronger base? a. HS or HO b. CH3O or...Ch. 2 - According to the explanations by Lewis, if a...Ch. 2 - a. Rank the following carboxylic acids from...Ch. 2 - Prob. 51PCh. 2 - For the following compound. a. draw its conjugate...Ch. 2 - Rank the following compounds from strongest to...Ch. 2 - Prob. 54PCh. 2 - Prob. 55PCh. 2 - a. Rank the following alcohols from strongest to...Ch. 2 - For each compound, indicate the atom that is most...Ch. 2 - a. Given the Ka values, estimate the pKa value of...Ch. 2 - A single bond between two carbons with different...Ch. 2 - Tenormin, a member of the group of drugs known as...Ch. 2 - From which of the following compounds can HO...Ch. 2 - a. For each of the following pairs of reactions,...Ch. 2 - Prob. 63PCh. 2 - Which is a stronger acid? a. b. c. d.Ch. 2 - Prob. 65PCh. 2 - Given that pH+ pOH = 14 and that the concentration...Ch. 2 - How could you separate a mixture of the following...Ch. 2 - Prob. 68PCh. 2 - a. If an add with a pKa of 5.3 is in an aqueous...Ch. 2 - Calculate the pH values of the following...Ch. 2 - Prob. 1PCh. 2 - Prob. 2PCh. 2 - Prob. 3PCh. 2 - Which of the reactions in Problem 3 favor...Ch. 2 - Prob. 5PCh. 2 - Prob. 6PCh. 2 - Prob. 7PCh. 2 - Which is the stronger acid? a. ClCH2CH2OH or...Ch. 2 - Prob. 9PCh. 2 - Prob. 10PCh. 2 - Which is a more stable base? Remembering that the...Ch. 2 - Which is the Stronger acid?Ch. 2 - Prob. 13PCh. 2 - a. Draw the structure of (CH3COOH (pKa = 4.7) at...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the structure of serine at pH 6.8. Click and drag to start drawing a structure. : d كarrow_forwardTake a look at this molecule, and then answer the questions in the table below it. CH2OH H H H OH OH OH CH2OH H H H H OH H H OH H OH Is this a reducing sugar? yes α β ロ→ロ no ☑ yes Does this molecule contain a glycosidic bond? If you said this molecule does contain a glycosidic bond, write the symbol describing it. O no 0+0 If you said this molecule does contain a glycosidic bond, write the common names (including anomer and enantiomer labels) of the molecules that would be released if that bond were hydrolyzed. If there's more than one molecule, separate each name with a comma. ☐arrow_forwardAnswer the questions in the table below about this molecule: H₂N-CH₂ -C—NH–CH–C—NH–CH—COO- CH3 CH CH3 What kind of molecule is this? 0= CH2 C If you said the molecule is a peptide, write a description of it using 3-letter codes separated ☐ by dashes. polysaccharide peptide amino acid phospolipid none of the above Хarrow_forward
- Draw a Haworth projection of a common cyclic form of this monosaccharide: CH₂OH C=O HO H H -OH H OH CH₂OH Click and drag to start drawing a structure. : ☐ Х S '☐arrow_forwardNucleophilic Aromatic Substitution 22.30 Predict all possible products formed from the following nucleophilic substitution reactions. (a) (b) 9 1. NaOH 2. HCI, H₂O CI NH₁(!) +NaNH, -33°C 1. NaOH 2. HCl, H₂Oarrow_forwardSyntheses 22.35 Show how to convert toluene to these compounds. (a) -CH,Br (b) Br- -CH3 22.36 Show how to prepare each compound from 1-phenyl-1-propanone. 1-Phenyl-1-propanone ہتی. Br. (b) Br (racemic) 22.37 Show how to convert ethyl benzene to (a) 2,5-dichlorobenzoic acid and (b) 2,4-dichlorobenzoic acid. 22.38 Show reagents and conditions to bring about the following conversions. (a) 9 NH2 8 CO₂H NH2 CO₂Et (d) NO2 NH2 S NH₂ NO2 CHS CHarrow_forward
- ive the major organic product(s) of each of the following reactions or sequences of reactions. Show all rant stereochemistry. [10 only] A. B. NaN3 1. LiAlH4, ether Br 2. H₂O CH3 HNO3 H₂/Pt H₂SO ethanol C. 0 0 CH3CC1 NaOH NHCCH AICI H₂O . NH₂ CH3CH2 N CH2CH3 + HCI CH₂CH 3 1. LIAIH, THE 2. H₂Oarrow_forwardCalculate the stoichiometric amount of CaCl2 needed to convert all of the CuSO4 into CuCl2.arrow_forwardH CH تنی Cl 1. NaCN, DMF 2. LIAIH4, ether H₂O pyridine N NH₂ 5 CH H 1 HNO, H₂SO 2. Nal NH2 Br Br HNO₂ CuCl H₂SO HCI CH3 H3C NN HSO KCN CuCN 1. HNO₂, H₂SO O₂N NH2 2. OH ཀ་ལས། །ས་ཅན་ :i་དེ་མ་མ་སེ་ NH₂ CH3 1. HNO₂, H₂SO4 2. H3PO₂ 1 HNO2, H2SO4 2. Nalarrow_forward
- ive the major organic product(s) of each of the following reactions or sequences of reactions. Show all rant stereochemistry. [10 only] A. B. NaN3 1. LiAlH4, ether Br 2. H₂O CH3 HNO3 H₂/Pt H₂SO ethanol C. 0 0 CH3CC1 NaOH NHCCH AICI H₂O . NH₂ CH3CH2 N CH2CH3 + HCI CH₂CH 3 1. LIAIH, THE 2. H₂Oarrow_forwardIf a pharmacy chain sold 65 million 500-mg tablets of aspirin, how many US tons of aspirin does this represent? Report your answer to 2 significant figures.arrow_forwardHere are the options: reducing a monosaccharide a non reducing disaccharide amylopectin cellulose 1,4' beta- glycosidearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY