
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22.SE, Problem 40AP
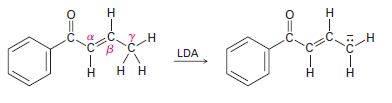
Base treatment of the following α, β-unsaturated carbonyl compound yields an anion by removal of H+ from the ᵞ carbon. Why are hydrogens on the g carbon atom acidic?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
how many moles of H2O2 are required to react with 11g of N2H4 according to the following reaction? (atomic weights: N=14.01, H=1.008, O= 16.00) 7H2O2 + N2H4 -> 2HNO3 + 8H20
calculate the number of moles of H2 produced from 0.78 moles of Ga and 1.92 moles HCL? 2Ga+6HCL->2GaCl3+3H2
an adult human breathes 0.50L of air at 1 atm with each breath. If a 50L air tank at 200 atm is available, how man y breaths will the tank provide
Chapter 22 Solutions
Organic Chemistry
Ch. 22.1 - Prob. 1PCh. 22.1 - How many acidic hydrogens does each of the...Ch. 22.1 - Prob. 3PCh. 22.3 - Write the complete mechanism for the deuteration...Ch. 22.3 - Prob. 5PCh. 22.4 - If methanol rather than water is added at the end...Ch. 22.5 - Prob. 7PCh. 22.5 - Draw a resonance structure of the acetonitrile...Ch. 22.6 - If methanol rather than water is added at the end...Ch. 22.7 - Prob. 10P
Ch. 22.7 - Draw a resonance structure of the acetonitrile...Ch. 22.7 - Why do you suppose ketone halogenations in acidic...Ch. 22.7 - Prob. 13PCh. 22.7 - Prob. 14PCh. 22.7 - Prob. 15PCh. 22.7 - Prob. 16PCh. 22.SE - Prob. 17VCCh. 22.SE - Prob. 18VCCh. 22.SE - Prob. 19VCCh. 22.SE - Prob. 20MPCh. 22.SE - Predict the product(s) and provide the mechanism...Ch. 22.SE - Predict the product(s) and provide the mechanism...Ch. 22.SE - Prob. 23MPCh. 22.SE - In the Hell–Volhard–Zelinskii reaction, only a...Ch. 22.SE - Prob. 25MPCh. 22.SE - Nonconjugated , -unsaturated ketones, such as...Ch. 22.SE - Prob. 27MPCh. 22.SE - Using curved arrows, propose a mechanism for the...Ch. 22.SE - Prob. 29MPCh. 22.SE - One of the later steps in glucose biosynthesis is...Ch. 22.SE - The Favorskii reaction involves treatment of an...Ch. 22.SE - Treatment of a cyclic ketone with diazomethane is...Ch. 22.SE - Prob. 33MPCh. 22.SE - Amino acids can be prepared by reaction of alkyl...Ch. 22.SE - Amino acids can also be prepared by a two-step...Ch. 22.SE - Heating carvone with aqueous sulfuric acid...Ch. 22.SE - Identify all the acidic hydrogens (pKa 25) in the...Ch. 22.SE - Rank the following compounds in order of...Ch. 22.SE - Prob. 39APCh. 22.SE - Base treatment of the following , -unsaturated...Ch. 22.SE - Prob. 41APCh. 22.SE - Prob. 42APCh. 22.SE - Prob. 43APCh. 22.SE - Which, if any, of the following compounds can be...Ch. 22.SE - Prob. 45APCh. 22.SE - Prob. 46APCh. 22.SE - Prob. 47APCh. 22.SE - How might you convert geraniol into either ethyl...Ch. 22.SE - Prob. 49APCh. 22.SE - One way to determine the number of acidic...Ch. 22.SE - Prob. 51APCh. 22.SE - Prob. 52APCh. 22.SE - Prob. 53APCh. 22.SE - Prob. 54APCh. 22.SE - Prob. 55APCh. 22.SE - Prob. 56APCh. 22.SE - All attempts to isolate primary and secondary...Ch. 22.SE - How would you synthesize the following compounds...Ch. 22.SE - Prob. 59APCh. 22.SE - Prob. 60APCh. 22.SE - Prob. 61APCh. 22.SE - Prob. 62APCh. 22.SE - As far back as the 16th century, South American...Ch. 22.SE - The key step in a reported laboratory synthesis of...Ch. 22.SE - Prob. 65AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Using reaction free energy to predict equilibrium composition Consider the following equilibrium: 2NO2 (g) = N2O4(g) AGº = -5.4 kJ Now suppose a reaction vessel is filled with 4.53 atm of dinitrogen tetroxide (N2O4) at 279. °C. Answer the following questions about this system: Under these conditions, will the pressure of N2O4 tend to rise or fall? Is it possible to reverse this tendency by adding NO2? In other words, if you said the pressure of N2O4 will tend to rise, can that be changed to a tendency to fall by adding NO2? Similarly, if you said the pressure of N2O4 will tend to fall, can that be changed to a tendency to '2' rise by adding NO2? If you said the tendency can be reversed in the second question, calculate the minimum pressure of NO 2 needed to reverse it. Round your answer to 2 significant digits. 00 rise ☐ x10 fall yes no ☐ atm G Ar 1arrow_forwardWhy do we analyse salt?arrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. H H CH3OH, H+ H Select to Add Arrows H° 0:0 'H + Q HH ■ Select to Add Arrows CH3OH, H* H. H CH3OH, H+ HH ■ Select to Add Arrows i Please select a drawing or reagent from the question areaarrow_forward
- What are examples of analytical methods that can be used to analyse salt in tomato sauce?arrow_forwardA common alkene starting material is shown below. Predict the major product for each reaction. Use a dash or wedge bond to indicate the relative stereochemistry of substituents on asymmetric centers, where applicable. Ignore any inorganic byproducts H Šali OH H OH Select to Edit Select to Draw 1. BH3-THF 1. Hg(OAc)2, H2O =U= 2. H2O2, NaOH 2. NaBH4, NaOH + Please select a drawing or reagent from the question areaarrow_forwardWhat is the MOHR titration & AOAC method? What is it and how does it work? How can it be used to quantify salt in a sample?arrow_forward
- Predict the major products of this reaction. Cl₂ hv ? Draw only the major product or products in the drawing area below. If there's more than one major product, you can draw them in any arrangement you like. Be sure you use wedge and dash bonds if necessary, for example to distinguish between major products with different stereochemistry. If there will be no products because there will be no significant reaction, just check the box under the drawing area and leave it blank. Note for advanced students: you can ignore any products of repeated addition. Explanation Check Click and drag to start drawing a structure. 80 10 m 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility DII A F1 F2 F3 F4 F5 F6 F7 F8 EO F11arrow_forwardGiven a system with an anodic overpotential, the variation of η as a function of current density- at low fields is linear.- at higher fields, it follows Tafel's law.Calculate the range of current densities for which the overpotential has the same value when calculated for both cases (the maximum relative difference will be 5%, compared to the behavior for higher fields).arrow_forwardUsing reaction free energy to predict equilibrium composition Consider the following equilibrium: N2 (g) + 3H2 (g) = 2NH3 (g) AGº = -34. KJ Now suppose a reaction vessel is filled with 8.06 atm of nitrogen (N2) and 2.58 atm of ammonia (NH3) at 106. °C. Answer the following questions about this system: rise Under these conditions, will the pressure of N2 tend to rise or fall? ☐ x10 fall Is it possible to reverse this tendency by adding H₂? In other words, if you said the pressure of N2 will tend to rise, can that be changed to a tendency to fall by adding H2? Similarly, if you said the pressure of N will tend to fall, can that be changed to a tendency to rise by adding H₂? If you said the tendency can be reversed in the second question, calculate the minimum pressure of H₂ needed to reverse it. Round your answer to 2 significant digits. yes no ☐ atm Х ด ? olo 18 Ararrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
IR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=_TmevMf-Zgs;License: Standard YouTube License, CC-BY