
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 22, Problem 47P
Interpretation Introduction
Interpretation:
Each of the letters (A-F) should be labeled in the below diagram as leading strand, lagging strand, template strand, 5-end, or 3-end.
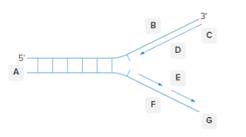
Concept Introduction:
Replication is the process which makes two exact new copies of the original DNA molecule. This process is called as semiconservative replication.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Experts...can any one help me solve these problems?
According to standard reduction potential data in Lecture 4-1, which of the following species is the most difficult to reduce?
Group of answer choices
Zn2+
AgCl(s)
Al3+
Ce4+
Which Group 1 metal reacts with O2(g) to form a metal peroxide (M2O2)?
Group of answer choices
Li
K
Rb
Na
Chapter 22 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 22.1 - Prob. 22.1PPCh. 22.1 - Prob. 22.1PCh. 22.1 - -Fluorouracil is an anticancer drug that...Ch. 22.1 - Prob. 22.3PCh. 22.1 - Prob. 22.4PCh. 22.1 - Label each statement about the compound...Ch. 22.1 - Draw the structure of each nucleotide: (a) UMP;...Ch. 22.1 - Give the name that corresponds to each...Ch. 22.2 - Draw the structure of a dinucleotide formed by...Ch. 22.2 - Draw the structure of each polynucleotide: (a) CU;...
Ch. 22.2 - Label each statement about the polynucleotide...Ch. 22.3 - Write the complementary strand for each of the...Ch. 22.4 - What is the sequence of a newly synthesized DNA...Ch. 22.6 - For each DNA segment: [1] What is the sequence of...Ch. 22.6 - Prob. 22.9PCh. 22.7 - What amino acid is coded for by each codon? GCC...Ch. 22.7 - What codons code for each amino acid? a. glycine...Ch. 22.7 - Drive the amino acid sequence that is coded for by...Ch. 22.7 - Write a possible mRNA sequence that codes for each...Ch. 22.7 - Considering the given sequence of nucleotides in...Ch. 22.8 - Prob. 22.14PCh. 22.8 - Prob. 22.8PPCh. 22.8 - Prob. 22.9PPCh. 22.9 - Prob. 22.10PPCh. 22.9 - Prob. 22.15PCh. 22.10 - Prob. 22.16PCh. 22 - Label each statement as pertaining to DNA, RNA, or...Ch. 22 - Label each statement as pertaining to DNA, RNA, or...Ch. 22 - Prob. 19PCh. 22 - (a) Give the name of each compound shown as a...Ch. 22 - Prob. 21PCh. 22 - Prob. 22PCh. 22 - Prob. 23PCh. 22 - Prob. 24PCh. 22 - Prob. 25PCh. 22 - Draw the structure of each of the following: a...Ch. 22 - Prob. 27PCh. 22 - Prob. 28PCh. 22 - Prob. 29PCh. 22 - Prob. 30PCh. 22 - Prob. 31PCh. 22 - Draw the structures of the two possible...Ch. 22 - Prob. 33PCh. 22 - Draw the structure of each dinucleotide and...Ch. 22 - Draw the deoxyribonucleotide TGA. Label the 5 and...Ch. 22 - Draw the ribonucleotide CGU. Label the 5 and 3...Ch. 22 - Prob. 37PCh. 22 - Describe in detail the DNA double helix with...Ch. 22 - Write the sequence of the complementary strand of...Ch. 22 - Prob. 40PCh. 22 - Prob. 41PCh. 22 - Prob. 42PCh. 22 - Prob. 43PCh. 22 - Prob. 44PCh. 22 - Prob. 45PCh. 22 - Figure 22.4 snows the hydrogen-bonding...Ch. 22 - Prob. 47PCh. 22 - Prob. 48PCh. 22 - Prob. 49PCh. 22 - Prob. 50PCh. 22 - What mRNA is transcribed from each DNA sequence in...Ch. 22 - Prob. 52PCh. 22 - For each DNA segment: [1] What is the sequence of...Ch. 22 - Prob. 54PCh. 22 - For each codon, give its anticodon and the amino...Ch. 22 - For each codon, give its anticodon and the amino...Ch. 22 - Fill in the missing information in the schematic...Ch. 22 - Fill in the missing information in the schematic...Ch. 22 - Derive the amino acid sequence that is coded for...Ch. 22 - Derive the amino acid sequence that is coded for...Ch. 22 - Write a possible mRNA sequence that codes for each...Ch. 22 - Prob. 62PCh. 22 - Prob. 63PCh. 22 - Prob. 64PCh. 22 - Prob. 65PCh. 22 - Prob. 66PCh. 22 - Consider the following mRNA sequence:...Ch. 22 - Consider the following mRNA sequence: 5-ACC UUA...Ch. 22 - Consider the following sequence of DNA: 3-TTA...Ch. 22 - Consider the following sequence of DNA: 3-ATA...Ch. 22 - Prob. 71PCh. 22 - Prob. 72PCh. 22 - Prob. 73PCh. 22 - Prob. 74PCh. 22 - Prob. 75PCh. 22 - Prob. 76PCh. 22 - Prob. 77PCh. 22 - Prob. 78PCh. 22 - Prob. 79PCh. 22 - Prob. 80PCh. 22 - Prob. 81PCh. 22 - Prob. 82PCh. 22 - Fill in the base, codon, anticodon, or amino acid...Ch. 22 - Fill in the base, codon, anticodon, or amino acid...Ch. 22 - Fill in the base, codon, anticodon, or amino acid...Ch. 22 - Prob. 86PCh. 22 - Prob. 87PCh. 22 - Prob. 88PCh. 22 - Prob. 89PCh. 22 - Prob. 90PCh. 22 - Prob. 91CPCh. 22 - Give a possible nucleotide sequence in the...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following statements is true regarding the reaction between Group 1 metals and water? Group of answer choices These reactions result in a basic solution. The metals do not actually react easily with water due to the metals' lack of conductivity. These reaction result in an acidic solution. The metals need their outer coatings of metal oxides to react.arrow_forwardWhich element cannot interact with hydrogen through hydrogen bonds? Group of answer choices O S Br Narrow_forwardWhich of the following statements is false regarding hydrogen gas production? Group of answer choices Steam reforming requires a catalyst. Methanol (CH3OH) can react with water using a ZnO catalyst to form H2(g). Methanol (CH3OH) can react with O2(g) using a Pd catalyst to form H2(g). The reaction between CH4(g) and H2O to form H2(g) requires a temperature of at least 700 oCarrow_forward
- Which of the following forms of hydrogen is the least stable? Group of answer choices H H2 H− H+arrow_forwardConsider the following reduction half reactions and standard reduction potentials: Fe3+ + e− → Fe2+ Eo = +0.77 V Fe2+ + e− → Fe(s) Eo = -0.44 V Which of the following statements is true? Group of answer choices The Fe2+ reduction to Fe(s) is spontaneous. Fe2+ can disproportionate into Fe3+ and Fe(s) The Fe3+ reduction to Fe2+ is not spontaneous. Fe3+ and Fe(s) can undergo a comproportionation reaction to form Fe2+arrow_forwardAccording to standard reduction potential data in Lecture 4-1, which of the following species is the most difficult to reduce? Group of answer choices Zn2+ AgCl(s) Al3+ Ce4+arrow_forward
- Consider the redox reaction: 2 P4 + 8 OH− + 4 H2O → 4 PH3 + 4 HPO32− The element oxidized is ["", "", ""] , the element reduced is ["", "", ""] , one of the oxidizing agents is ["", "", ""] , and the reducing agent is ["", "", ""] .arrow_forwardWhat is the missing reactant in this organic reaction? OH H + R Δ CH3-CH2-CH-CH3 O CH3 CH3-CH2-C-O-CH-CH2-CH3 + H2O Specifically, in the drawing area below draw the condensed structure of R. If there is more than one reasonable answer, you can draw any one of them. If there is no reasonable answer, check the No answe box under the drawing area. Explanation Check Click anywhere to draw the first atom of your structure. C O2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Cerarrow_forwardPredict the product of this organic reaction: CH3 NH2 Δ CH3-CH-CH3 + HO-C-CH2-N-CH3 P+H₂O Specifically, in the drawing area below draw the condensed structure of P. If there is no reasonable possibility for P, check the No answer box under the drawing area. Explanation Check Click anywhere to draw the first atom of your structure. Xarrow_forward
- In the scope of the SCH4U course, please thoroughly go through the second questionarrow_forwardPlease help me solve these two problems. Thank you in advance.arrow_forwardNaming and drawing unsubstituted esters Write the systematic name of each organic molecule: Explanation structure Check name Х 2/5arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co


Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Nucleic acids - DNA and RNA structure; Author: MEDSimplified;https://www.youtube.com/watch?v=0lZRAShqft0;License: Standard YouTube License, CC-BY