
Interpretation:
The IUPAC name of the following compound should be determined:
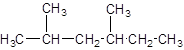
Concept introduction:
The hydrocarbon compounds that contains single bond(s) between carbon atom(s) are said to be saturated hydrocarbon. Hydrocarbon compounds containing single bonds only are said to be
Answer to Problem 8PP
2, 4-dimethylhexane.
Explanation of Solution
In order to give the IUPAC name to the alkane following steps are followed:
1. The parent (longest) continuous carbon chain is identified.
2. The ending of the parent chain for alkane (-e) is -ane.
3. Name should be written in alphabetical order and numbering should be done in such a way that the substituent group gets lowest number.
4. Hyphen is used to connect the number to the name.
For number of carbons atoms in alkane chain, the prefix is given as:

The given structure is:
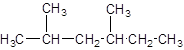
The parent chain contains 2 substituents so numbering is done in such a way that substituents gets lower number as shown:
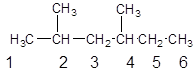
Since, the parent chain contains 6 carbon atoms and only single bonds are present so, the parent chain is hexane. Since, the substituents,
Interpretation:
The IUPAC name of the following compound should be determined:
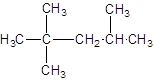
Concept introduction:
The hydrocarbon compounds that contains single bond(s) between carbon atom(s) are said to be saturated hydrocarbon. Hydrocarbon compounds containing single bonds only are said to be alkane.
Answer to Problem 8PP
2, 2, 4-trimethylpentane.
Explanation of Solution
The given structure is:
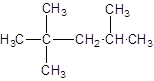
The parent chain contains 3 substituents so numbering is done in such a way that substituents gets lower number as shown:
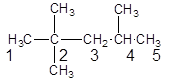
Since, the parent chain contains 5 carbon atoms and only single bonds are present so, the parent chain is pentane. Since, the substituents,
Interpretation:
The IUPAC name of the following compound should be determined:
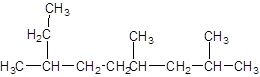
Concept introduction:
The hydrocarbon compounds that contains single bond(s) between carbon atom(s) are said to be saturated hydrocarbon. Hydrocarbon compounds containing single bonds only are said to be alkane.
Answer to Problem 8PP
2, 4, 7-trimethylnonane.
Explanation of Solution
The given structure is:
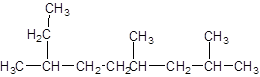
The parent chain contains 3 substituents so numbering is done in such a way that substituents gets lower number as shown:
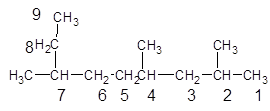
Since, the parent chain contains 9 carbon atoms and only single bonds are present so, the parent chain is nonane. Since, the substituents,
Chapter 21 Solutions
Glencoe Chemistry: Matter and Change, Student Edition
Additional Science Textbook Solutions
Genetic Analysis: An Integrated Approach (3rd Edition)
Physics for Scientists and Engineers: A Strategic Approach, Vol. 1 (Chs 1-21) (4th Edition)
Campbell Biology (11th Edition)
Applications and Investigations in Earth Science (9th Edition)
Brock Biology of Microorganisms (15th Edition)
Cosmic Perspective Fundamentals
- 6. Draw the molecular orbital diagram shown to determine which of the following is paramagnetic. B22+ B22+, B2, C22, B22 and N22+ Molecular Orbital Diagram B2 C22- B22- N22+ Which molecule is paramagnetic?arrow_forward3. Put the following species in order of increasing bond length by using molecular orbital diagrams and calculating their bond orders: F2, F2, F2+ Molecular Orbital Diagram F2 F2 F2+ Bond Order Shortest bond: Longest bondarrow_forward3. Put the following species in order of increasing bond length by using molecular orbital diagrams and calculating their bond orders: F2, F2, F2+ Molecular Orbital Diagram F2 F2 F2+ Bond Orderarrow_forward
- 4. The superoxide ion, Oz, plays an important role in the ageing processes that take place in organisms. Judge whether Oz is likely to have larger or smaller dissociation energy than 02. Molecular Orbital Diagram 02 02 Does O2 have larger or smaller dissociation energy?: Bond Orderarrow_forward1. How many molecular orbitals can be built from the valence shell orbitals in O2?arrow_forwardSho reaction mechanism. Don't give Ai generated solutionarrow_forward
- Is this aromatic, antiaromatic, or nonaromatic?arrow_forwardOn what basis are Na and Nb ranked against each other?arrow_forwardStep 1: add a curved arrow. Select Draw Templates More / " C H Br 0 Br : :o: Erase H H H H Q2Q Step 2: Draw the intermediates and a curved arrow. Select Draw Templates More MacBook Air / " C H Br 0 9 Q Erase 2Qarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





