
Concept explainers
The amino acid alanine is a solid at room temperature and has a melting point of
Interpretation:
The reason for alanine which is solid to have a melting point of
Concept introduction:
Structure of alanine is as follows:
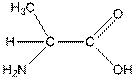
Alanine is a hydrophobic amino acid as it has only one
Structure of pyruvic acid is as follows:
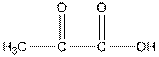
Answer to Problem 21P
Due to the strong electrostatic force of attraction alanine is solid and has a high melting point of
Explanation of Solution
Alanine is a hydrophobic amino acid also an ionic salt with strong electrostatic forces. Therefore, alanine has a high melting point of
Want to see more full solutions like this?
Chapter 21 Solutions
General, Organic, and Biological Chemistry - 4th edition
- How would you use infrared spectroscopy to distinguish between the following pairs of constitutional isomers? (a) CH3C=CCH3 || and CH3CH2C=CH (b) CH3CCH=CHCH3 and CH3CCH2CH=CH2 Problem 12-41 The mass spectrum (a) and the infrared spectrum (b) of an unknown hydrocarbon are shown. Propose as many structures as you can. (a) 100 Relative abundance (%) 80 60 60 40 200 20 (b) 100 Transmittance (%) 10 20 20 80- 60- 40- 20 40 60 80 100 120 140 m/z 500 4000 3500 3000 2500 2000 1500 Wavenumber (cm-1) 1000arrow_forwardPropagation of uncertainty. You have a stock solution certified by the manufacturer to contain 150.0±0.03 µg SO42-/mL. You would like to dilute it by a factor of 100 to obtain 1.500 µg/mL. Calculate the uncertainty in the two methods of dilution below. Use the following uncertainty values for glassware: Glassware Uncertainty (assume glassware has been calibrated and treat the values below as random error) 1.00 mL volumetric pipet 0.01 mL 10.00 mL volumetric pipet 0.02 mL 100.00 mL volumetric flask 0.08 mL Transfer 10.00 mL with a volumetric pipet and dilute it to 100 mL with a volumetric flask. Then take 10.00 mL of the resulting solution and dilute it a second time with a 100 mL flask. 2. Transfer 1.00 mL with a volumetric pipet and dilute it to 100 mL with a volumetric flask.arrow_forwardDraw all resonance structures for the following ion: CH₂ Draw all resonance structures on the canvas by choosing buttons from the Tools (for bonds), Atoms, and Advanced Template toolbars, including charges where needed. The single bond is active by default. 2D ד CONT HD EXP CON ? 1 [1] Α 12 Marvin JS by Chemaxon A DOO H C N Br I UZ OSPFarrow_forward
- What is the average mass of the 10 pennies? Report your value with correct significant figures. What is the error (uncertainty) associated with each mass measurement due to the equipment? What is the uncertainty associated with the average value? Note that the uncertainty of the balance will propagate throughout the calculation. What is the standard deviation of the 10 mass measurements? Explain the difference between the propagated uncertainty and the standard deviation. Which number would you use to describe the uncertainty in the measurement? Calculate the total mass of the pennies with associated uncertainty. Calculate the average density of a penny based on these data. Propagate the uncertainty values for both mass and volume in your calculations.arrow_forwardCan you help me and explain the answers please.arrow_forwardB 1 of 2 Additional problems in preparation to Midterm #1: 1.) How can the following compounds be prepared using Diels-Alder reaction: CH3 O CN (a) (b) CN CH3 2.) What is the missing reagent in the shown reaction? H3C + ? H3C H3C CN H3C ''CN (၁) H 3.) Write the products 1,2-addition and 1,4-addition of DBr to 1,3-cyclohexadiene. Remember, D is deuterium, a heavy isotope of hydrogen. It reacts exactly like hydrogen. 4.) In the shown reaction, which will be the kinetic product and which will be the thermodynamic product? H3C CI H3C HCI H3C + 5.) Which of the following molecules is aromatic? (a) (b) (c) H 6.) Which of the following molecules is aromatic? (a) (b) (c) 7.) Write the mechanism for the shown reaction. + Ха AICI 3 CI 8.) Suggest reagents that would convert benzene into the shown compounds. CI NO2 -8-6-6-8-a (a) (b) (c) (d) (e) (a) SO3H Brarrow_forward
- The number of 2sp^2 hybridized atoms in is: A. 8; B. 6; C.4; D.2; E.0;arrow_forwardThe highest boiling compound from among the following isA. 2-methylheptane; B. 3-methylheptane; C. 2,2-dimethylhexane;D. octane; E. 2,2,3-trimethylpentanearrow_forwardWhich of the following features are found in the most stable structure ofCH5NO that does not have a CO bond?w. a π bond, x. two NH bonds, y. one OH bond, z. 3 lone pairsA. w, x; B. x, y; C. y, z; D. x, y, z; E. all of them.arrow_forward
- Which one of the following functional groups is not present in thecompound shownA. amine; B. aldehyde, C. ether; D. amide. E. ketonearrow_forwardWhich of the following formulas correspond to at least one compound inwhich resonance is important?w. C2H5N x. C3H5Br; y. C3H4; z. C4H6.A. w, x, y; B. x, y, z; C. w, x, z; D. w, y, z; E. all of themarrow_forwardPredict the product(s) that are formed after each step for reactions 1-4. In each case, consider formation of any chiral center(s) and draw all expected stereoisomers. 1) OH 1) HBr (SN2) 2) NaOH, heat 3) BH3, THF 4) H2O2, NaOH 2) OH 1) SOCI 2, py 2) NaOEt 3) Br2, H₂O 3) OH 1) H2SO4 conc. 2) HBr, ROOR 3) KOtBu 4) OH 1) TsCl, py 2) NaOEt 3) 03 4) DMSarrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





