
Interpretation: The set up of oxidation-reduction in a galvanic cell needs to be explained.
Concept Introduction: By transfer of electrons,
Answer to Problem 28A
Explanation of Solution
From an oxidation-reduction reaction, a galvanic cell uses energy to produce an electric current. Electrons are transferred quickly and collide with each other if oxidizing and reducing agent are in contact with each other.
The electrons must travel via external circuit from the reducing agent to oxidizing agent. Oxidizing agent is separated from the reducing agent. Salt bridge is used to avoid the accumulation of charges in the beakers. The below figure represents a galvanic cell:
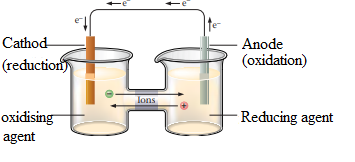
Reducing agent releases electrons, so is oxidized and oxidizing agent gains electrons and reduced in the other electrode.
An oxidizing agent gains electrons and a reducing agent lose electrons in a redox reaction.
Chapter 18 Solutions
World of Chemistry
- + Draw the product of the E2 reaction shown below. Include the correct stereochemistry. Ignore any inorganic byproducts. Ph CH2CH3 H H3C H Br DBN [૪] Drawing Atoms, Bonds and Rings H | OH Charges ―00 H. C | Undo Reset Br I Remove Done Drag To Pan +arrow_forwardReaction A Now the production A Œ In the product of reaction i 12 Dear the product of actionarrow_forwardMacmillan Learnin When an unknown amine reacts with an unknown acid chloride, an amide with a molecular mass of 163 g/mol (M* = 163 m/z) is formed. In the infrared spectrum, important absorptions appear at 1661, 750 and 690 cm-1. The 13C NMR and DEPT spectra are provided. Draw the structure of the product as the resonance contributor lacking any formal charges. 13C NMR DEPT 90 200 160 120 80 40 0 200 160 120 80 DEPT 135 200 160 120 80 40 0 Draw the unknown amide. 40 40 0arrow_forward
- Draw the major product karmed when I reach with the epoxide. Use walge dah bonds, including hydrogen al alcach genic center, to show the chemistry of the product Beeldraw any hydrogen akams on coxygen where applicablearrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. H I Select to Add Arrows + H H 'H Q H2O H2O CI:O .H H H H I Select to Add Arrows I : C H2O H H H Select to Add Arrows 'Harrow_forward+ Draw an alkyl halide that produces ONLY the following alkene in an E2 elimination. Ignore any inorganic byproducts. Drawing Strong Base Q Atoms, Bonds and Rings Charges HO Br H2N Undo Reset Remove Done Drag To Panarrow_forward
- For the dehydrohalogenation (E2) reaction shown, draw the major organic product. Хок Br tert-butanol heat Select Drew Templates More Erase CH QQQarrow_forwardMacmillan Learning Draw the major, neutral organic product for each substitution reaction. For this question, assume that each substitution reaction goes to completion. Disregard elimination. Reaction A. CI H₂O Select Draw Templates More Erase C Harrow_forwardMacmillan Learning Reaction B: CI HO_ 곳으 / Select Draw Templates More с € H D Erasearrow_forward
- When 2-bromo-93-dimethylbutane is heated with sodium methoxide, one majors.. në la formed. 4th attempt Part 1 (0.5 point) t Ji See Periodic Table See Hint Draw the major alkene product and all other byproducts. Be sure to include lone-pair electrons and charges. Part 2 (0.5 point) What type of mechanism is occuring? Choose one: AS1 3rd attempt X H 41 See Hint Part 1 (0.5 point) Feedback See Periodic Table See Hintarrow_forwardComplete the mechanism for the E1 reaction below by following the directions written above each of the five boxes. Be sure to include lone pair electrons and nonzero formal charges. 2nd attempt 1 Provide the missing curved arrow notation. E+ RUDDA 1st attempt Feedback See Periodic Table See Hint Iir See Periodic Table See Hintarrow_forwardHeating an alcohol in the presence of sulfuric or phosphoric acid will cause a dehydration to occur: the removal of the elements of water from a molecule, forming an alkene. The reaction usually follows an E1 mechanism. The SN1 pathway is suppressed by using a strong acid whose conjugate base is a poor nucleophile. Further, heating the reaction mixture causes a greater increase in the rate of E1 compared to the rate of Sy1. 3rd attempt h Draw curved arrow(s) to show how the alcohol reacts with phosphoric acid. TH © 1 0 0 +1% # 2nd attempt Feedback H Ju See Periodic Table See Hint H Jud See Periodic Table See Hintarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





