
Concept explainers
(a)
Interpretation:
Given amide has to be classified as primary, secondary, or tertiary amide.
Concept Introduction:
Organic compounds are the important basis of life. They include gasoline, coal, dyes, and clothing fibers etc. The compounds that are obtained from living organisms are termed as organic compounds and those obtained from the earth are known as inorganic compounds. Organic compounds are found in earth also apart from living organisms. All the organic compounds contain the element carbon. Urea was synthesized in the laboratory which is an organic compound.
Organic compounds contain heteroatom also. Some of them are nitrogen, sulfur, oxygen etc. Nitrogen containing organic compounds are of two important types and they are
One of the
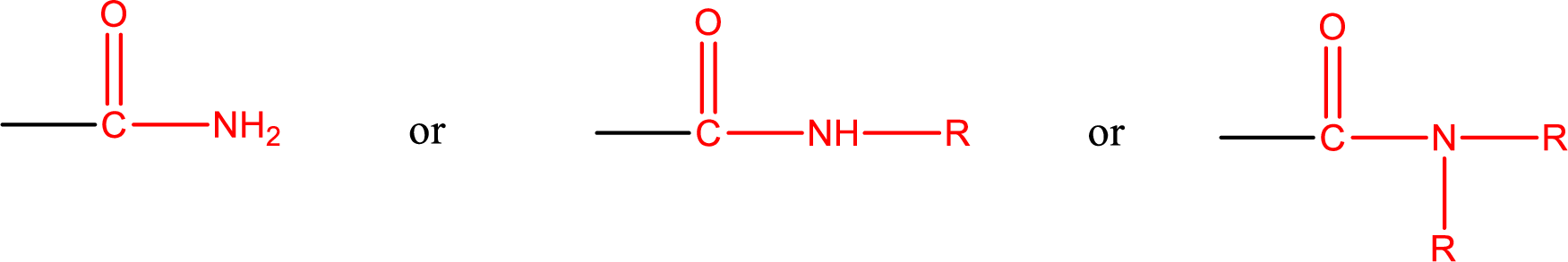
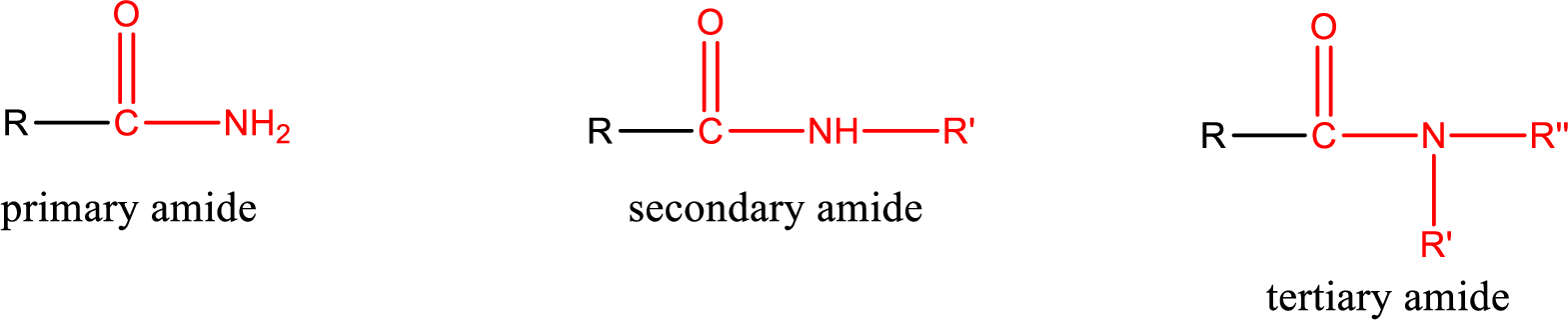
Amides are also classified as primary, secondary, and tertiary amide.
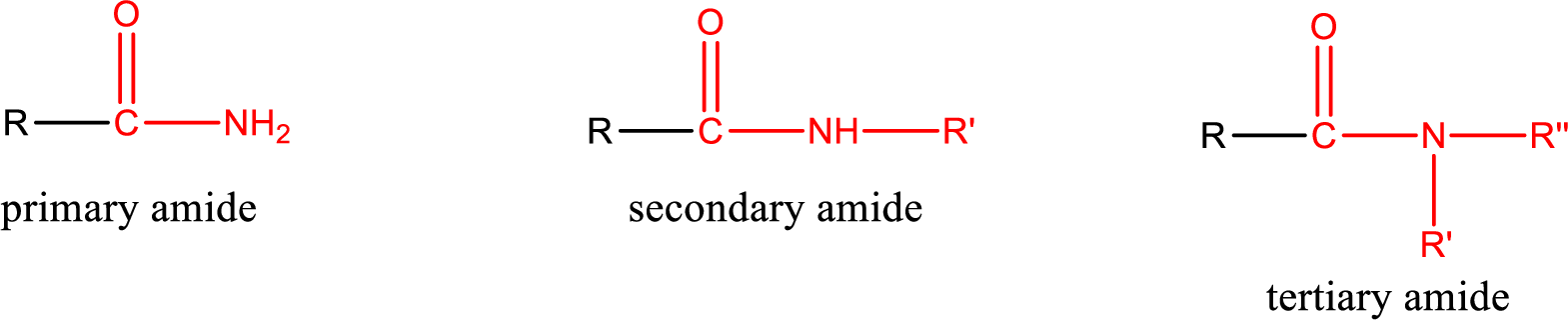
Primary amide is the one that has a nitrogen atom that is bonded to two hydrogen atoms. Primary amides are also known as unsubstituted amides.
Secondary amide is the one that has a nitrogen atom that is bonded to one hydrogen atom and one alkyl (or aryl) group. Secondary amides are also known as monosubstituted amides.
Tertiary amide is the one that has a nitrogen atom that is bonded to two alkyl (or aryl) groups. Tertiary amides are also known as disubstituted amides.
Apart from linear amides, there are also cyclic amides. They are formed by intramolecular condensation. Cyclic amides are also known as lactams.
(b)
Interpretation:
Given amide has to be classified as primary, secondary, or tertiary amide.
Concept Introduction:
Organic compounds are the important basis of life. They include gasoline, coal, dyes, and clothing fibers etc. The compounds that are obtained from living organisms are termed as organic compounds and those obtained from the earth are known as inorganic compounds. Organic compounds are found in earth also apart from living organisms. All the organic compounds contain the element carbon. Urea was synthesized in the laboratory which is an organic compound.
Organic compounds contain heteroatom also. Some of them are nitrogen, sulfur, oxygen etc. Nitrogen containing organic compounds are of two important types and they are amines, amides.
One of the carboxylic acid derivatives is amide. In this the carboxyl
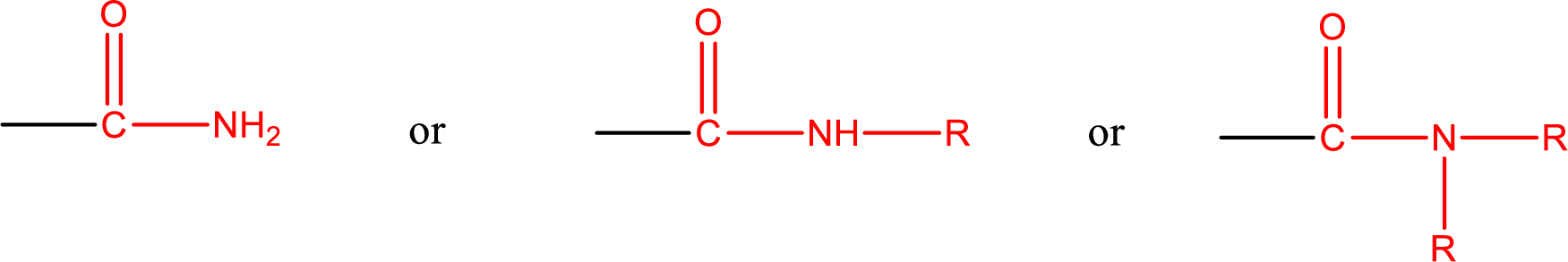
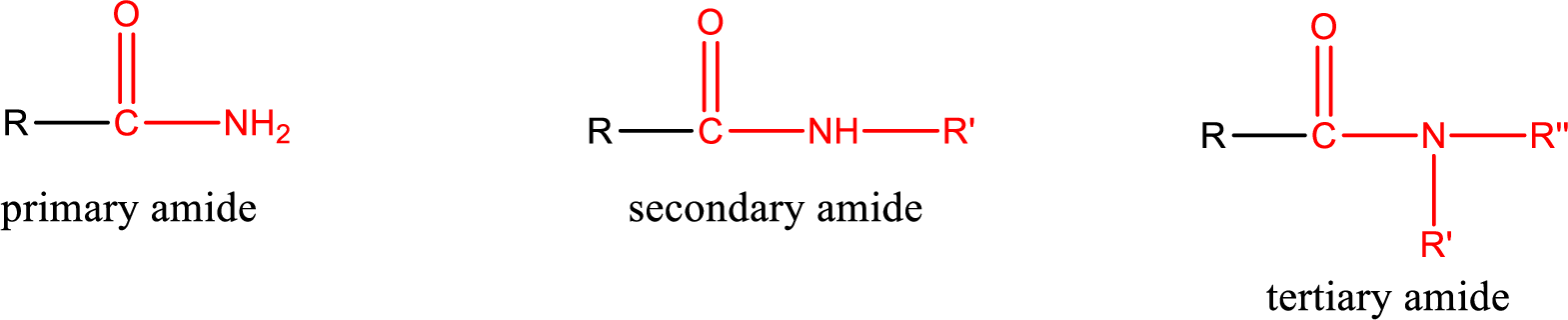
Amides are also classified as primary, secondary, and tertiary amide.
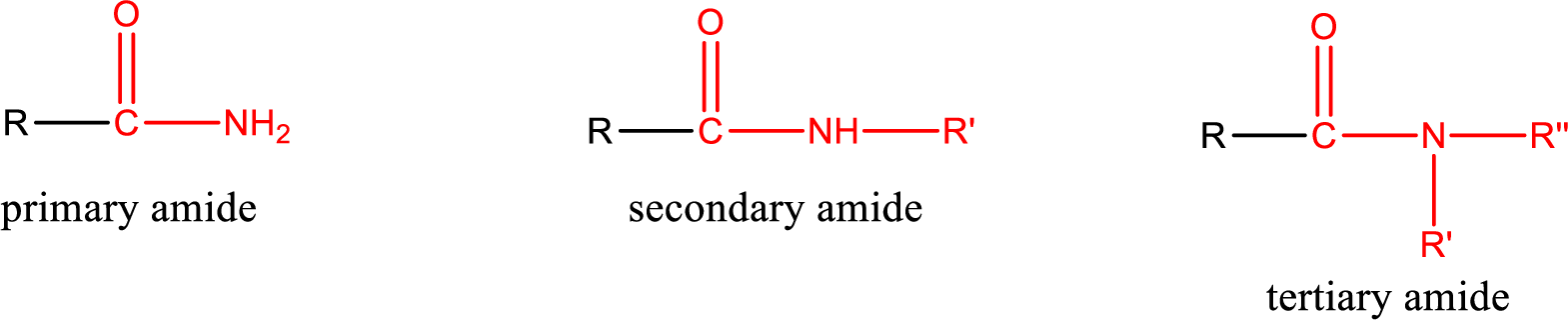
Primary amide is the one that has a nitrogen atom that is bonded to two hydrogen atoms. Primary amides are also known as unsubstituted amides.
Secondary amide is the one that has a nitrogen atom that is bonded to one hydrogen atom and one alkyl (or aryl) group. Secondary amides are also known as monosubstituted amides.
Tertiary amide is the one that has a nitrogen atom that is bonded to two alkyl (or aryl) groups. Tertiary amides are also known as disubstituted amides.
Apart from linear amides, there are also cyclic amides. They are formed by intramolecular condensation. Cyclic amides are also known as lactams.
(c)
Interpretation:
Given amide has to be classified as primary, secondary, or tertiary amide.
Concept Introduction:
Organic compounds are the important basis of life. They include gasoline, coal, dyes, and clothing fibers etc. The compounds that are obtained from living organisms are termed as organic compounds and those obtained from the earth are known as inorganic compounds. Organic compounds are found in earth also apart from living organisms. All the organic compounds contain the element carbon. Urea was synthesized in the laboratory which is an organic compound.
Organic compounds contain heteroatom also. Some of them are nitrogen, sulfur, oxygen etc. Nitrogen containing organic compounds are of two important types and they are amines, amides.
One of the carboxylic acid derivatives is amide. In this the carboxyl
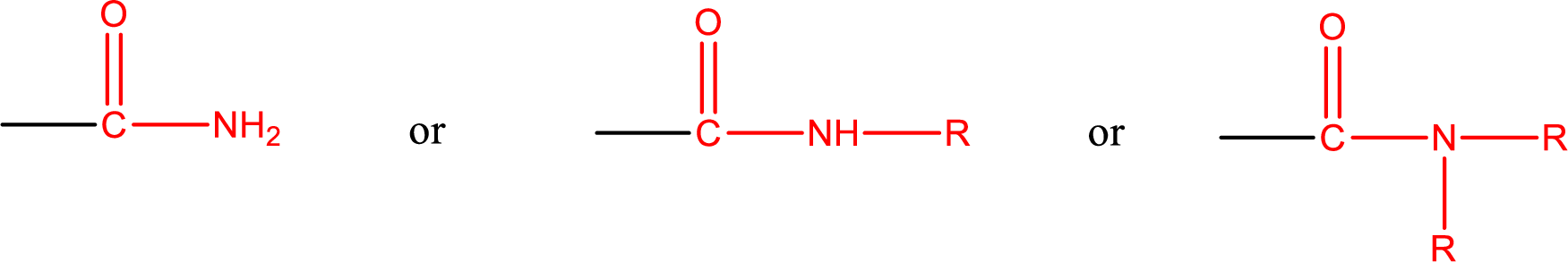
Amides are also classified as primary, secondary, and tertiary amide.
Primary amide is the one that has a nitrogen atom that is bonded to two hydrogen atoms. Primary amides are also known as unsubstituted amides.
Secondary amide is the one that has a nitrogen atom that is bonded to one hydrogen atom and one alkyl (or aryl) group. Secondary amides are also known as monosubstituted amides.
Tertiary amide is the one that has a nitrogen atom that is bonded to two alkyl (or aryl) groups. Tertiary amides are also known as disubstituted amides.
Apart from linear amides, there are also cyclic amides. They are formed by intramolecular condensation. Cyclic amides are also known as lactams.
(d)
Interpretation:
Given amide has to be classified as primary, secondary, or tertiary amide.
Concept Introduction:
Organic compounds are the important basis of life. They include gasoline, coal, dyes, and clothing fibers etc. The compounds that are obtained from living organisms are termed as organic compounds and those obtained from the earth are known as inorganic compounds. Organic compounds are found in earth also apart from living organisms. All the organic compounds contain the element carbon. Urea was synthesized in the laboratory which is an organic compound.
Organic compounds contain heteroatom also. Some of them are nitrogen, sulfur, oxygen etc. Nitrogen containing organic compounds are of two important types and they are amines, amides.
One of the carboxylic acid derivatives is amide. In this the carboxyl
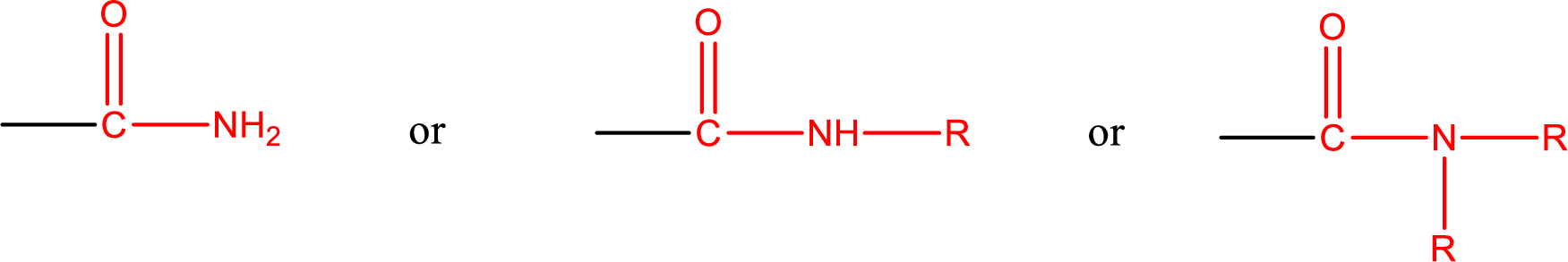
Amides are also classified as primary, secondary, and tertiary amide.
Primary amide is the one that has a nitrogen atom that is bonded to two hydrogen atoms. Primary amides are also known as unsubstituted amides.
Secondary amide is the one that has a nitrogen atom that is bonded to one hydrogen atom and one alkyl (or aryl) group. Secondary amides are also known as monosubstituted amides.
Tertiary amide is the one that has a nitrogen atom that is bonded to two alkyl (or aryl) groups. Tertiary amides are also known as disubstituted amides.
Apart from linear amides, there are also cyclic amides. They are formed by intramolecular condensation. Cyclic amides are also known as lactams.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Bundle: General, Organic, and Biological Chemistry, 7th + OWLv2 Quick Prep for General Chemistry, 4 terms (24 months) Printed Access Card
- Explain why the total E in an Einstein depends on the frequency or wavelength of the light.arrow_forwardIf the dissociation energy of one mole of O2 is 5.17 eV, determine the wavelength that must be used to dissociate it with electromagnetic radiation. Indicate how many Einstein's of this radiation are needed to dissociate 1 liter of O2 at 25°C and 1 atm of pressure.Data: 1 eV = 96485 kJ mol-1; R = 0.082 atm L K-1; c = 2.998x108 m s-1; h = 6.626x10-34 J s; NA = 6.022x 1023 mol-1arrow_forwardIndicate the number of Einsteins that are equivalent to 550 kJ mol⁻¹ of absorbed energy (wavelength 475 nm).arrow_forward
- Indicate the number of einsteins that are equivalent to 550 kJ mol⁻¹ of absorbed energy?arrow_forwardA unit used in photochemistry is the einstein. If 400 kJ mol-1 of energy has been absorbed, how many einsteins is this equivalent to?arrow_forwardFor the condensation reaction between Alanine and histidine write the amididation reaction mechanism using arrows then write the three letter code for the product of the reaction and the one letter code for the product of the reaction.arrow_forward
- Name the following molecules using iupacarrow_forwardWrite the amididation reaction mechanism of a-aminophenol and acetic acid to produce acetaminophenarrow_forwardFor the condensation reaction between Alamine and histamine, please help me write the amididation reaction mechanism. Then write the three letter code for the product of the reaction, then write the one letter code for the product of the reaction. arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning


