
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 16.3, Problem 16.7P
Acetone and progesterone are two 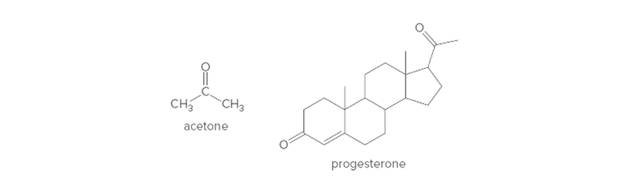
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Given a chemical test to distinguish between:(i) Ethane and ethane(ii) Ethanol and ethanoic acid(iii) Soaps and Detergents
(a) Write the molecular formula of acetaminophen (see Fig. 7.34b).(b) A tablet of Extra Strength Tylenol contains 500 mg acetaminophen. Calculate the chemical amount (in moles) of that compound in the tablet.
Describe the solubility, density and physical properties of Hexane, Cyclohexane and Toluene.
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 16.1 - Prob. 16.1PCh. 16.1 - Draw the structure of the three constitutional...Ch. 16.2 - Give the IUPAC name for each aldehyde. a. (...Ch. 16.2 - Give the structure corresponding to each TUPAC...Ch. 16.2 - Prob. 16.4PCh. 16.2 - Prob. 16.2PPCh. 16.2 - Give the structure corresponding to each name. a....Ch. 16.3 - Which compound in each pair has the higher boiling...Ch. 16.3 - Acetone and progesterone are two ketones that...Ch. 16.4 - Prob. 16.8P
Ch. 16.5 - What product is formed when each carbonyl compound...Ch. 16.5 - Prob. 16.4PPCh. 16.5 - Prob. 16.9PCh. 16.6 - Prob. 16.5PPCh. 16.6 - Prob. 16.10PCh. 16.6 - Prob. 16.11PCh. 16.7 - Prob. 16.12PCh. 16.8 - Prob. 16.6PPCh. 16.8 - Prob. 16.7PPCh. 16.8 - Prob. 16.13PCh. 16.8 - Prob. 16.8PPCh. 16.8 - Label the three acetalsin solanine, the toxic...Ch. 16.8 - Prob. 16.14PCh. 16.8 - Prob. 16.10PPCh. 16 - Prob. 15PCh. 16 - Prob. 16PCh. 16 - Prob. 17PCh. 16 - Prob. 18PCh. 16 - Prob. 19PCh. 16 - Prob. 20PCh. 16 - Prob. 21PCh. 16 - Prob. 22PCh. 16 - Prob. 23PCh. 16 - Prob. 24PCh. 16 - Give an acceptable name for each ketone. a. b. c....Ch. 16 - Prob. 26PCh. 16 - Prob. 27PCh. 16 - Draw the structure corresponding to each name. a....Ch. 16 - Prob. 29PCh. 16 - Prob. 30PCh. 16 - Prob. 31PCh. 16 - Prob. 32PCh. 16 - Draw out the structure of benzaldehyde, including...Ch. 16 - Prob. 34PCh. 16 - Which compound in each pair has the higher boiling...Ch. 16 - Prob. 36PCh. 16 - Prob. 37PCh. 16 - Prob. 38PCh. 16 - Prob. 39PCh. 16 - Prob. 40PCh. 16 - Prob. 41PCh. 16 - Prob. 42PCh. 16 - Prob. 43PCh. 16 - Prob. 44PCh. 16 - Prob. 45PCh. 16 - Consider the following ball-and-stick model....Ch. 16 - Prob. 47PCh. 16 - Prob. 48PCh. 16 - Prob. 49PCh. 16 - Prob. 50PCh. 16 - Prob. 51PCh. 16 - Prob. 52PCh. 16 - Prob. 53PCh. 16 - Prob. 54PCh. 16 - Prob. 55PCh. 16 - Prob. 56PCh. 16 - Prob. 57PCh. 16 - Prob. 58PCh. 16 - Prob. 59PCh. 16 - Label the functional group(s) in each compound as...Ch. 16 - Prob. 61PCh. 16 - Prob. 62PCh. 16 - What acetal is formed when each aldehyde or ketone...Ch. 16 - What acetal is formed when each aldehyde or ketone...Ch. 16 - Prob. 65PCh. 16 - Prob. 66PCh. 16 - Prob. 67PCh. 16 - Prob. 68PCh. 16 - Prob. 69PCh. 16 - Prob. 70PCh. 16 - Prob. 71PCh. 16 - Prob. 72PCh. 16 - Prob. 73PCh. 16 - Answer each question about phenylacetaldehyde,...Ch. 16 - Prob. 75PCh. 16 - Prob. 76PCh. 16 - Prob. 77PCh. 16 - Prob. 78PCh. 16 - Three constitutional isomers of molecular formula...Ch. 16 - Identify A—C in the following reaction sequenceCh. 16 - Androsterone is a male sex hormone that controls...Ch. 16 - Prob. 82PCh. 16 - Prob. 83PCh. 16 - Prob. 84PCh. 16 - Prob. 85PCh. 16 - Paraldehyde, a hypnotic and sedative once commonly...Ch. 16 - Prob. 87PCh. 16 - Prob. 88PCh. 16 - Prob. 89CPCh. 16 - Prob. 90CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- . (a) Write the molecular formula of acetylsalicylic acid (see Fig. 7.34a).(b) An aspirin tablet contains 325 mg acetylsalicylic acid. Calculate the number of moles of that compound in the tablet.arrow_forwardWrite the reaction, occurring in the liver, that causes theoxidation of methanol. What is the product of this reaction, and what is the possible result of the accumulation ofthe product in the body?arrow_forwardWrite an equation for the reaction of chloroacetic acid (Ka=1.5103) with trimethylamine (Kb=5.9105) . Calculate the equilibrium constant for the reaction. If 0.10 M solutions of these two species are mixed, what will be their concentrations at equilibrium?arrow_forward
- Intermolecular interactions play a key role in the binding of morphine to its receptor. State the types of interaction that may occur and discuss the key structural features of morphine associated with these interactions.arrow_forwardwhat physical property explains this difference in toxicity between methylmercury chloride and Calomel?arrow_forwardWrite a generalized statement regarding the solubility of hydrocarbons in a polar solvent (water) and a less polar solvent (dicloromethane) .What conclusion can you make regarding the polarity of hydrocarbons?arrow_forward
- Write the products of the reaction of diphenhydramine (a base) with the acid HCI shown below. H COCH₂CH₂NCH3 + HC1 CH3 Consider the chemical reaction from the previous question. Are the reactants or products more soluble in water? Briefly explain.arrow_forwardUsing the relationship between the structure and the boiling point. Obtain a table for the listed chemicals below showing how the boiling point will be different with (1) the different chemical structures, (2) with different functional groups. Boiling Point and Functionalized Compounds: Chemical: Cyclohexanol Cyclohexanone Cyclohexene Benzaldehyde Benzyl alcoholarrow_forwardWrite the skeletal structures of propanal, acetone and cyclohexanone. What is the major intermolecular force (IMF) found in them? Based on their major intermolecular force and molecular weight, what can you predict on their solubility in water? Chemical Name Skeletal Structures Major IMF Solubility in water Propanal Acetone Cyclohexanonearrow_forward
- which has the highest solubility between methanol and hexane?arrow_forwardWhat is meant by the following terms? Explain with an example for each.(i) Target molecules as used in medicinal chemistry(ii) Food preservative(iii) Non-ionic detergentsarrow_forwardWhat is an oxide of nitrogen that serves as an anesthetic and analgesic.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning  EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

EBK A SMALL SCALE APPROACH TO ORGANIC L
Chemistry
ISBN:9781305446021
Author:Lampman
Publisher:CENGAGE LEARNING - CONSIGNMENT

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning
07 Physical Properties of Organic Compounds; Author: Mindset;https://www.youtube.com/watch?v=UjlSgwq4w6U;License: Standard YouTube License, CC-BY