
Concept explainers
(a)
Interpretation:
The product formed in the reaction of
Concept Introduction:
Potassium dichromate is a good oxidizing agent and it has the tendency to oxidize alcohols to
Answer to Problem 41P
Explanation of Solution
Aldehydes when made to react with potassium dichromate are known to form the corresponding carboxylic acids. The reaction of hexanal in the presence of potassium dichromate will result in the formation of hexanoic acid as shown below in the chemical equation:
Hence the reaction of hexanal with potassium dichromate results in the formation of the corresponding
(b)
Interpretation:
The product formed in the reaction of

Concept Introduction:
Potassium dichromate is a good oxidizing agent and it has the tendency to oxidize alcohols to aldehydes and aldehydes to carboxylic acids as well. An oxidizing agent is a chemical species which has the tendency to oxidize other to possible higher oxidation states by making them loose electron and thus itself gets reduced.
Answer to Problem 41P
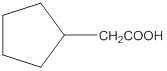
Explanation of Solution
Aldehydes when made to react with potassium dichromate are known to form the corresponding carboxylic acids. The reaction of 2-cyclopentylacetaldehyde in the presence of potassium dichromate will result in the formation of 2-cyclopentanoic acid as shown below in the chemical equation:
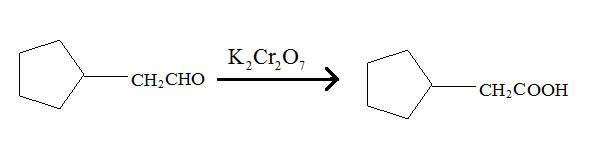
Hence the reaction of 2-cyclopentylacetaldehyde with potassium dichromate results in the formation of the corresponding carboxylic acid which is 2-cyclopentylacetic acid.
(c)
Interpretation:
The product formed in the reaction of
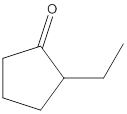
Concept Introduction:
Potassium dichromate is a good oxidizing agent and it has the tendency to oxidize alcohols to aldehydes and aldehydes to carboxylic acids as well. An oxidizing agent is a chemical species which has the tendency to oxidize other to possible higher oxidation states by making them loose electron and thus itself gets reduced.
Answer to Problem 41P
No product formed.
Explanation of Solution
The given compound is 2-ethylcyclopentanone and it cannot react with potassium dichromate as it contains ketone as a
(d)
Interpretation:
The product formed in the reaction of
Concept Introduction:
Potassium dichromate is a good oxidizing agent and it has the tendency to oxidize alcohols to aldehydes and aldehydes to carboxylic acids as well. An oxidizing agent is a chemical species which has the tendency to oxidize other to possible higher oxidation states by making them loose electron and thus itself gets reduced.
Answer to Problem 41P
Explanation of Solution
The given structure clearly shows that this compound is hexanol. It is an aliphatic alcohol and it will react with potassium reagent to form the corresponding aldehyde which will further get oxidized and the reaction will finally result in the formation of a carboxylic acid.
Want to see more full solutions like this?
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- Calculating standard reaction free energy from standard reduction... Using standard reduction potentials from the ALEKS Data tab, calculate the standard reaction free energy AG° for the following redox reaction. Be sure your answer has the correct number of significant digits. 3Cu+ (aq) + Cro²¯ (aq) +4H₂O (1) → 3Cu²+ (aq) +Cr(OH)3 (s)+5OH˜¯ (aq) 0 kJ ☐ x10 00. 18 Ararrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 241.7 mL of a 0.4900M solution of methylamine (CH3NH2) with a 0.7800M solution of HNO3. The pK of methylamine is 3.36. Calculate the pH of the base solution after the chemist has added 17.7 mL of the HNO3 solution to it. Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HNO3 solution added. Round your answer to 2 decimal places. pH = ☑ ? 18 Ararrow_forwardThe following is two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 223.4 148.7 353.7 278.2 334.6 268.7 305.6 234.4 340.0 262.7 304.3 283.2 244.7 143.6 QUESTION: For both groups of data calculate the answers attached in the image.arrow_forward
- The following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 340.0mmol/L 262.7mmol/L QUESTION: For both groups (Regular & Salt Reduced tomato sauce) of data provide answers to the following calculations below: 1. Standard Deviation (Sx) 2. T Values (t0.05,4) 3. 95% Confidence Interval (mmol/L) 4. [Na+] (mg/100 mL) 5. 95% Confidence Interval (mg/100 mL)arrow_forwardIf we have leucine (2-amino-4-methylpentanoic acid), alanine (2-aminopropanoic acid) and phenylalanine (2-amino-3-phenylpropanoic acid), indicate the tripeptides that can be formed (use the abbreviated symbols Leu., Ala and Phe).arrow_forwardBriefly state why trifluoroacetic acid is more acidic than acetic acid.arrow_forward
- Explain why acid chlorides are more reactive than amides in reactions with nucleophiles.arrow_forwardCalculating the pH of a weak base titrated with a strong acid An analytical chemist is titrating 101.7 mL of a 0.3500M solution of piperidine (C5H10NH) with a 0.05700M solution of HClO4. The pK of piperidine is 2.89. Calculate the pH of the base solution after the chemist has added 682.9 mL of the HClO solution to it. 4 Note for advanced students: you may assume the final volume equals the initial volume of the solution plus the volume of HClO solution added. 4 Round your answer to 2 decimal places. pH = .11 00. 18 Ararrow_forwardThe following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 340.0 262.7 QUESTION: For both groups of data provide answers to the calculations attached in the imagearrow_forward
- 7. Concentration and uncertainty in the estimate of concentration (class data) Class mean for sample (Regular) |[Cl-] (mmol/L) class mean Sn za/2 95% Confidence Interval (mmol/L) [Na+] (mg/100 mL) 95% Confidence Interval (mg/100 mL)arrow_forwardThe following is a two groups (Regular tomato sauce & Salt Reduced Tomato Sauce) of data recorded by a team analysising salt content in tomato sauce using the MOHR titration method: Regular Tomato Sauce Salt Reduced Tomato Sauce 223.4 148.7 353.7 278.2 334.6 268.7 305.6 234.4 340.0 262.7 304.3 283.2 244.7 143.6 QUESTION: For both groups of data calculate the answers attached in the image.arrow_forwardGive reason(s) for six from the followings [using equations if possible] a. Addition of sodium carbonate to sulfanilic acid in the Methyl Orange preparation. b. What happened if the diazotization reaction gets warmed up by mistake. c. Addition of sodium nitrite in acidified solution in MO preparation through the diazotization d. Using sodium dithionite dihydrate in the second step for Luminol preparation. e. In nitroaniline preparation, addition of the acid mixture (nitric acid and sulfuric acid) to the product of step I. f. What is the main reason of the acylation step in nitroaniline preparation g. Heating under reflux. h. Fusion of an organic compound with sodium. HAND WRITTEN PLEASEarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
