
Concept explainers
(a)
Interpretation:
The picture of the solution made by mixing the solutions A and B together after the precipitation reaction takes place is to be drawn.
Concept Introduction:
The molarity is the number of moles of the solute dissolved per liter volume of the solution. It is represented in mathematical term such that,
Where, n is the number of moles,
V is the volume of the solution
The limiting regent is that reactant of the reaction which control the amount of the product formed.
(a)
Answer to Problem 63A
The diagram shows the relative volume compared to the both solutions
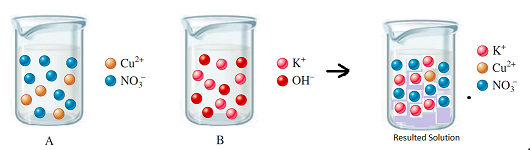
Explanation of Solution
Given information:
The volume and molarity of the copper nitrate solution are
And,
The volume and molarity of the B solution is,
Calculation:
Where, n is the number of moles,
V is the volume of the solution
The number of moles of the solution A is,
Similarly, the number of moles of solution B is,
The balanced chemical rection between the solution A and solution B is,
The number of moles of calcium hydroxide formed from the calcium nitrate is,
And from the potassium hydroxide is,
So, potassium hydroxide is the limiting reagent.
The diagram shows the relative volume compared to the both solutions
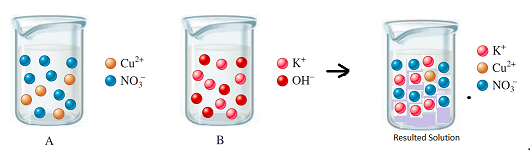
Thus, the diagram shows the relative volume of the given solutions.
(b)
Interpretation:
The concentration of the ions and mass of solid is to be determined.
Concept Introduction:
The molarity is the number of moles of the solute dissolved per liter volume of the solution. It is represented in mathematical term such that,
(b)
Answer to Problem 63A
The concentration of all ions left in the solution are
Explanation of Solution
Given information:
The volume and molarity of the copper nitrate solution are
And,
The volume and molarity of the B solution is,
Calculation:
Where, n is the number of moles,
V is the volume of the solution
The mass of the copper hydroxide is calculated as,
The initial moles of
Similarly, used moles of copper ion is 3 moles. So, the remaining number of moles is,
Total volume is 4.00 L
The concertation of remaining copper ion is,
Similarly, the number of moles of hydroxide ion is,
The used number of moles of hydroxide ion is,
So, the remaining number of moles is zero of hydroxide ions.
The remaining number of moles of nitrate ions is,
The remaining mole of potassium ion is,
The concentration of nitrate and potassium ions is,
And,
Thus, the concentration of all ions left in the solution are
Chapter 15 Solutions
World of Chemistry
- PLEASE ANSWER THE QUESTION!!!arrow_forward3. SYNTHESIS. Propose a sequence of synthetic steps (FGI) that convert the starting material (SM) into the Target molecule. For each FGI in your proposed synthesis, specify the reagents / conditions, and draw the product(s) of that FGI. DO NOT INCLUDE the FGI mxn in the answer you submit. If an FGI requires two reagent sets, specify the order in which the reagent sets are added, e.g., i) Hg(OAc)2 / H₂O; ii) NaBH4/MeOH. Indicate the stereochemistry (if any) of the products of each FGI. FGI 1. Me Starting Material Source of all carbons in the Target molecule (can use multiple copies) Me Me Target molecule + enantiomerarrow_forwardcurved arrows are used to illustate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction mechanism stepsarrow_forward
- At 90ºC the vapor pressure of ortho-xylene is 20 kPa and that of meta-xylene is 18 kPa. What is the composition of the vapor in equilibrium with a mixture in which the mole fraction of o-xylene is 0.60?arrow_forwardDraw the products of this reduction of a ketone with sodium borohydride. Use a dash or wedge bond to indicate the stereochemistry of substituents on asymmetric centers, where applicableIgnore any inorganic byproducts. 1) NaBH4 2) HCI/H2O Select to Drawarrow_forwardWhy do you think people who live at high altitudes are advised to add salt to water when boiling food like pasta? What mole fraction of NaCl is needed to raise the boiling point of H2O by 3˚C? Does the amount of salt added to water (typically about one teaspoon to four quarts of water) substantially change the boiling point? (Kb (H2O) = 0.51˚C/molal.)arrow_forward
- pls help asaparrow_forwardpls help asaparrow_forward9. Consider the following galvanic cell: Fe (s) | Fe(NO3)2 (aq) || Sn(NO3)2 (aq) | Sn (s) a. Write an equation for the half reactions occurring at the anode and cathode. b. Calculate the standard cell potential Show all of your work. c. Draw and label the galvanic cell, including the anode and cathode, direction of electron flow, and direction of ion migration.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





