
Concept explainers
(a)
Interpretation:
pH of
Concept Introduction:
pH definition:
The concentration of hydrogen ion is measured using pH scale. The acidity of aqueous solution is expressed by pH scale.
The pH of a solution is defined as the negative base-10 logarithm of the hydrogen or hydronium ion concentration.
The
(a)
Explanation of Solution
Given,
Volume of acid in liters:
Original mole
The given solution contains only
(b)
Interpretation:
pH of the solution has to be calculated, after addition of
Concept Introduction:
Refer part (a).
(b)
Explanation of Solution
After the addition of the titrant,
To determine the equilibrium concentration of
(c)
Interpretation:
pH of the solution has to be calculated, after addition of
Concept Introduction:
Refer part (a).
(c)
Explanation of Solution
Used the equation in (b), again
Volume of base (L)
(d)
Interpretation:
pH of the solution has to be calculated, after addition of
Concept Introduction:
Refer part (a).
(d)
Explanation of Solution
The volume and total moles of base can be calculated as
Total moles of
(e)
Interpretation:
pH of the solution has to be calculated, after addition of
Concept Introduction:
Refer part (a).
(e)
Explanation of Solution
At equivalent point, the moles of base begin to exceed the moles of acid, so we adapt the expression we used in (b) for a solution with excess base to determine the equilibrium concentration of
The solution has a
(f)
Interpretation:
pH of the solution has to be calculated, after addition of
Concept Introduction:
Refer part (a).
(f)
Explanation of Solution
The pH of equivalent point of titrant can be calculated as
Hence, the results in a pH verses volume of titrant to make a titration curve, including equivalence point:
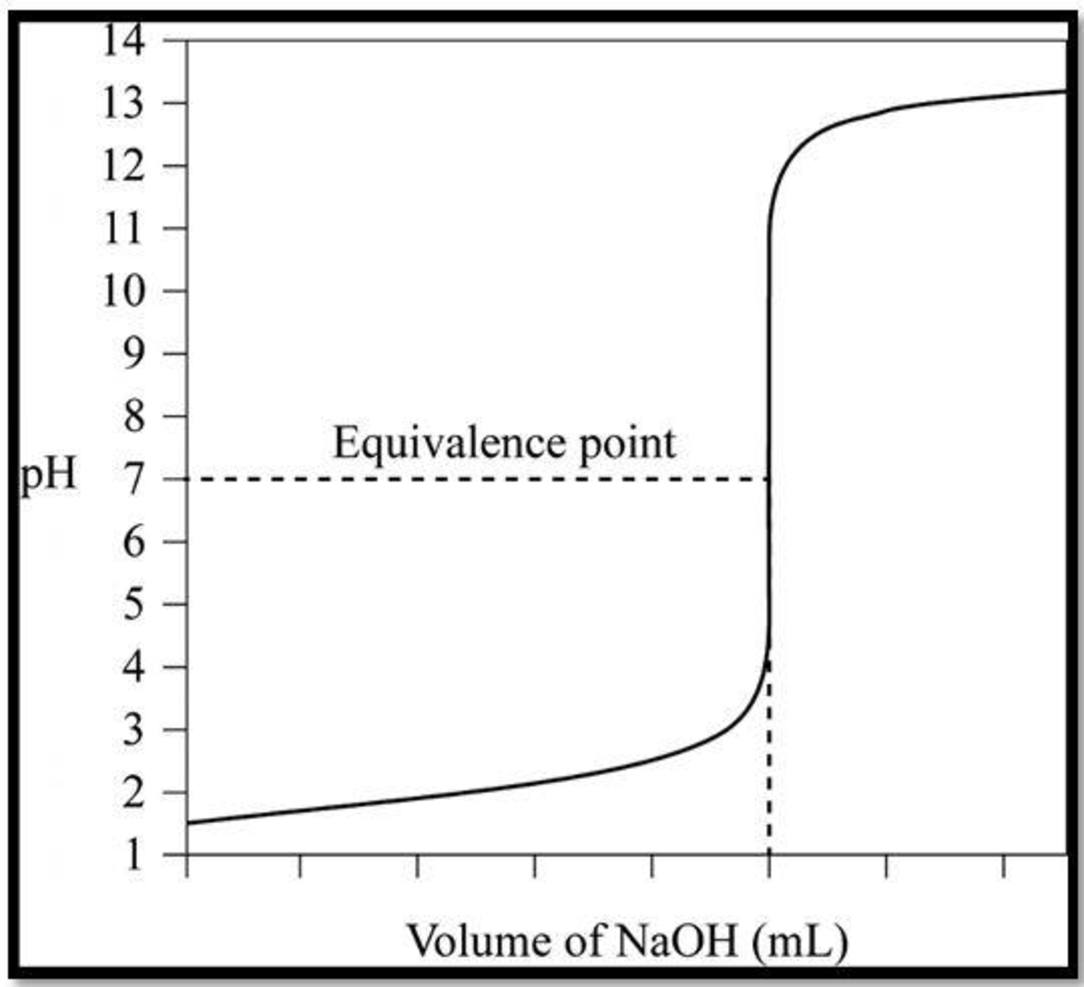
Want to see more full solutions like this?
Chapter 15 Solutions
Chemistry: The Molecular Science
- 1. Using a Model set Build a model for the following compound [CHBRIF] 2. Build another model of the mirror image of your first molecule. 3. Place the two models next to each other and take a picture which shows the differences between the two models. 4. Determine the absolute stereochemistry R or S for the two models. 5. Write or type a paragraph to Discuss the stereochemical relationship between the two models of CHBгCIF. You must provide an explanation for your conclusions also provide a description for the colors used to representarrow_forwardThe specific rotation of a sample depends upon measured angle of rotation, the density of the sample, and the pathway length of the light. True Falsearrow_forwardConsider the molecule A,B, C and D shown below, (1 x 4) Br NH2 A OH Br 边 H B C D 1. Assign the R/S configuration to each chiral center and identify by circling all the chiral centers. 2. Draw an image for the enantiomer of each of the compounds A, B, C and D.arrow_forward
- Could you crystallize one enantiomer of mandelic acid from a racemic mixture (using the typical achiral solvents found in our lab) without preparing a diastereomeric salt? Why or why not? No, because both enantiomers have the same solubility in achiral solvents. than the other. ооо Yes, because one enantiomer has a higher melting point No, because both enantiomers are liquids. Yes, because one enantiomer is more crystalline than the other.arrow_forwardIf the literature value of specific rotation for a chiral compound is -53.6°, what is the enantiomeric excess of a compound with a measured specific rotation of -40.5°?arrow_forwardThe process to determine the configuration, starts by placing the lowest priority substituent toward the back. If the substituents pointing forward decrease in priority in a clockwise order, the configuration is S. If the substituents decrease in priority in a counterclockwise order, the configuration is R. True Falsearrow_forward
- In the drawing area below, create a hemiacetal with 1 hydroxyl group, 1 methoxy group, and a total of 3 carbon atoms. Click and drag to start drawing a structure. Explanation Check Х PO 18 Ar B © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibilityarrow_forwardPredict the product of the reaction below (3 pts). hydrazine Ph H₂NNH2 KOH Write the mechanism for the above reaction using curved arrows to show electron movements. show all intermediates in the process (7 pts).arrow_forward↓ Feedback (8/10) Draw the major product of this reaction. Ignore inorganic byproducts. Incorrect, 2 attempts remaining N H3O+ 0 × Select to Draw + V Retryarrow_forward
- 2. Calculate the branching ratio of the reaction of the methyl peroxy radical with either HO, NO 298K) (note: rate constant can be found in the tropospheric chemistry ppt CH,O,+NO-HCHO+HO, + NO, CH₂O+HO, CH₂00H +0₂ when the concentration of hydroperoxyl radical is DH01-1.5 x 10 molecules and the nitrogen oxide maxing ratio of 10 ppb when the concentration of hydroperoxyl radicalis [H0] +1.5x10 molecules cm" and the nitrogen oxide mixing ratio of 30 p Under which condition do you expect more formaldehyde to be produced and whyarrow_forwardIndicate the product of the reaction of benzene with 1-chloro-2,2-dimethylpropane in the presence of AlCl3.arrow_forwardIn what position will N-(4-methylphenyl)acetamide be nitrated and what will the compound be called.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning





