
Concept explainers
(a)
Interpretation: To identify the heme degradation product (1) bilirubin, (2) biliverdin, (3) stercobilin, and (4) urobilin in whose production “ring-opening” occurs.
Concept introduction: Hemoglobin is a heme protein present in the red blood cells. The protein part is called globin and the non-protein part is heme. Heme is the prosthetic group that contains 4 pyrrole groups bonded together and has an iron atom in the center. The structure of the heme group is:
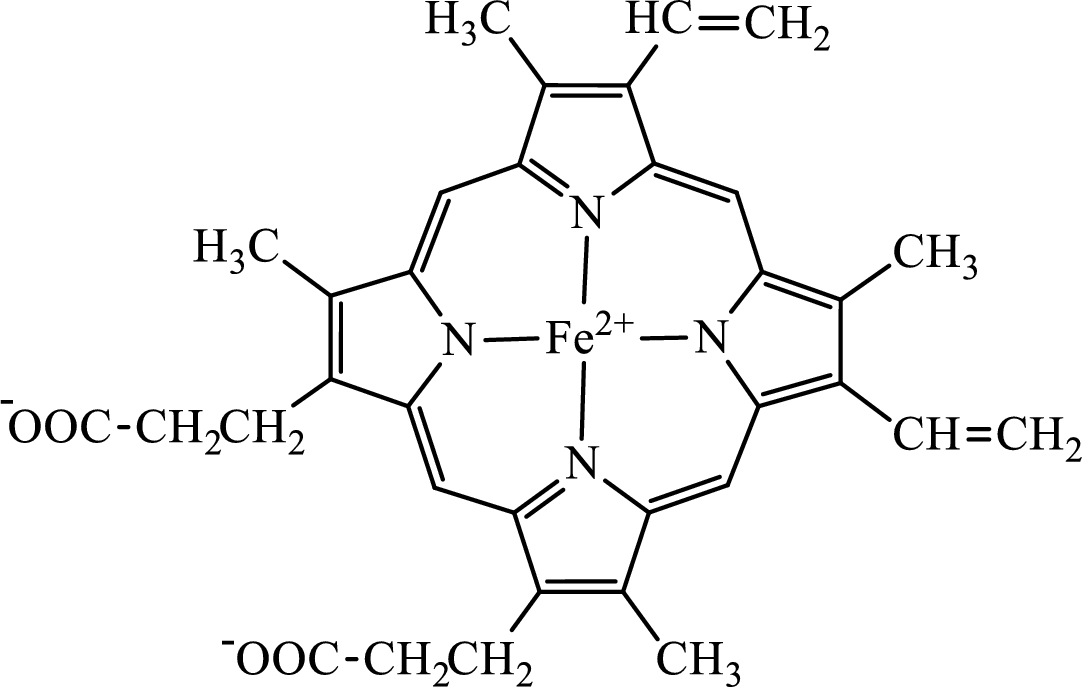
The first step of degradation of heme involves opening of pyrrole ring with the release of the iron atom and production of biliverdin. The iron atom released becomes part of ferritin protein. Biliverdin produced is converted bilirubin in the spleen. Bilirubin is then transported to the liver where attachment of sugar residues to the propionate side chains of the bilirubin occurs to make it more soluble. Then more solubilized bilirubin is excreted in bile and finally to the small intestine. In the small intestine, it is converted into stercobilin for excretion in feces or urobilin for excretion in urine.
(b)
Interpretation: To identify the heme degradation product (1) bilirubin, (2) biliverdin, (3) stercobilin, and (4) urobilin in whose production a carbon-carbon double bond is changed to a carbon-carbon single bond.
Concept introduction: Hemoglobin is a heme protein present in the red blood cells. The protein part is called globin and the non-protein part is heme. Heme is the prosthetic group that contains 4 pyrrole groups bonded together and has an iron atom in the center.
The structure of the heme group is:
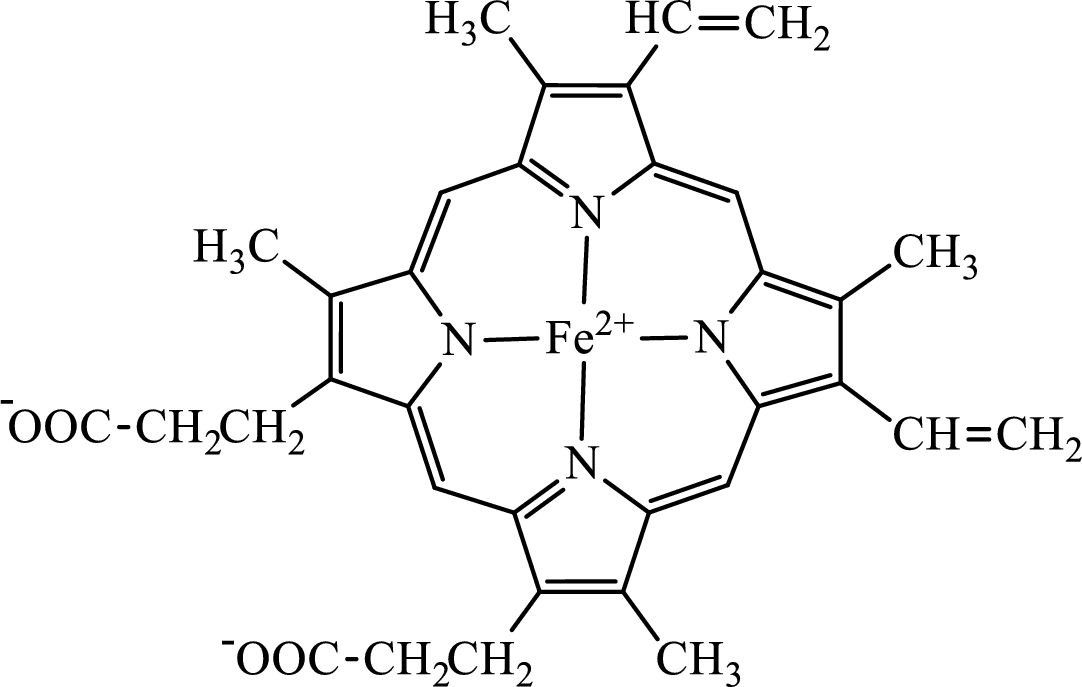
The first step of degradation of heme involves opening of pyrrole ring with the release of the iron atom and production of biliverdin. The iron atom released becomes part of ferritin protein. Biliverdin produced is converted bilirubin in the spleen. Bilirubin is then transported to the liver where attachment of sugar residues to the propionate side chains of the bilirubin occurs to make it more soluble. Then more solubilized bilirubin is excreted in bile and finally to the small intestine. In the small intestine, it is converted into stercobilin for excretion in feces or urobilin for excretion in urine.
(c)
Interpretation: To identify the heme degradation product (1) bilirubin, (2) biliverdin, (3) stercobilin, and (4) urobilin that is rendered more water soluble by use of a glucose derivative.
Concept introduction: Hemoglobin is a heme protein present in the red blood cells. The protein part is called globin and the non-protein part is heme. Heme is the prosthetic group that contains 4 pyrrole groups bonded together and has an iron atom in the center.
The first step of degradation of heme involves opening of pyrrole ring with the release of the iron atom and production of biliverdin. The iron atom released becomes part of ferritin protein. Biliverdin produced is converted bilirubin in the spleen. Bilirubin is then transported to the liver where attachment of sugar residues to the propionate side chains of the bilirubin occurs to make it more soluble. Then more solubilized bilirubin is excreted in bile and finally to the small intestine. In the small intestine, it is converted into stercobilin for excretion in feces or urobilin for excretion in urine.
The structure of the heme group is:
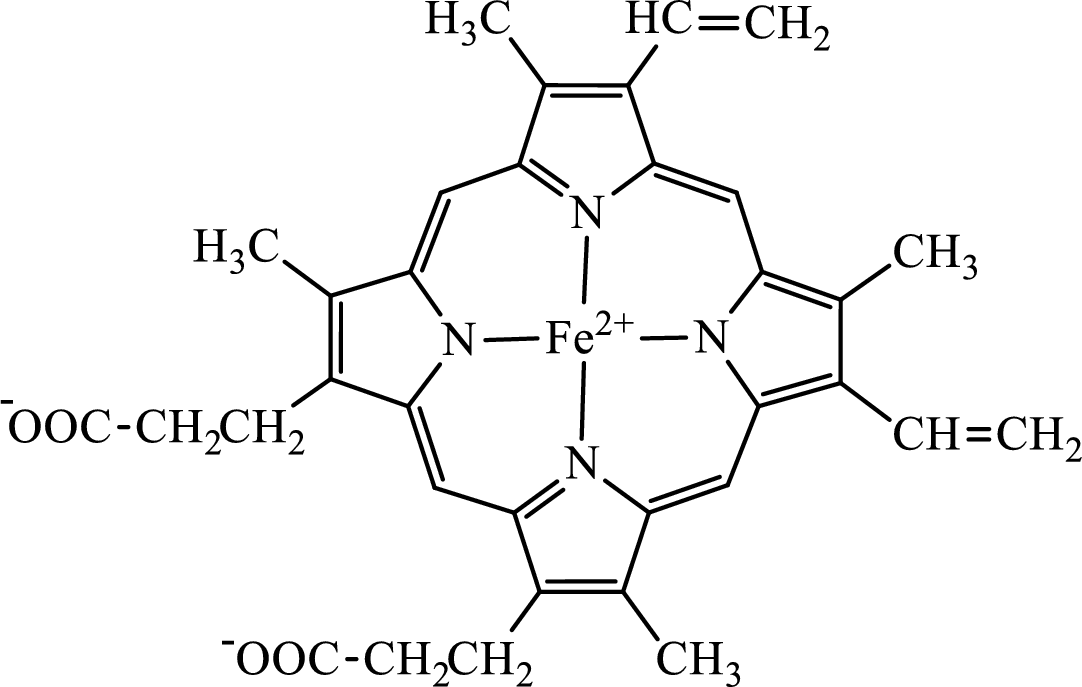
(d)
Interpretation: To identify the heme degradation product (1) bilirubin, (2) biliverdin, (3) stercobilin, and (4) urobilin which is bile pigment and has a yellowish color.
Concept introduction: Hemoglobin is a heme protein present in the red blood cells. The protein part is called globin and the non-protein part is heme. Heme is the prosthetic group that contains 4 pyrrole groups bonded together and has an iron atom in the center.
The structure of the heme group is:
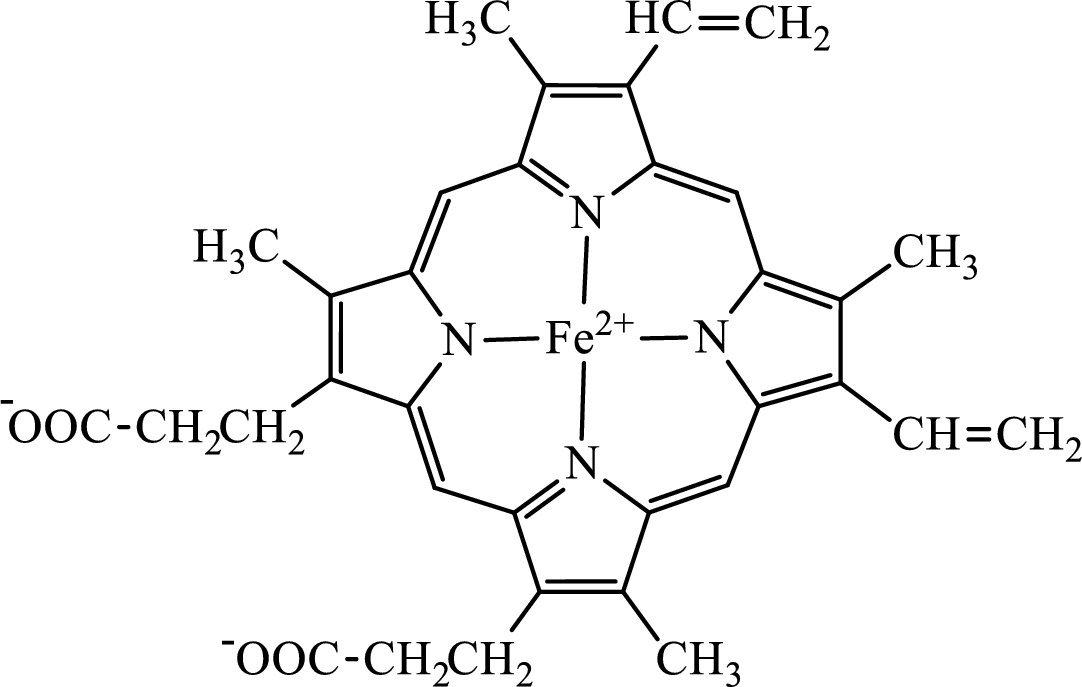
The first step of degradation of heme involves opening of pyrrole ring with the release of the iron atom and production of biliverdin. The iron atom released becomes part of ferritin protein. Biliverdin produced is converted bilirubin in the spleen. Bilirubin is then transported to the liver where attachment of sugar residues to the propionate side chains of the bilirubin occurs to make it more soluble. Then more solubilized bilirubin is excreted in bile and finally to the small intestine. In the small intestine, it is converted into stercobilin for excretion in feces or urobilin for excretion in urine.
Bile pigments are the colored degradation product of tetrapyrrole carbon arrangement of heme portion of hemoglobin. These are excreted in bile and give characteristic color to urine and feces.
Want to see the full answer?
Check out a sample textbook solution
Chapter 15 Solutions
Organic And Biological Chemistry
- 19) Which metal is most commonly used in galvanization to protect steel structures from oxidation? Lead a. b. Tin C. Nickel d. Zinc 20) The following molecule is an example of a: R₁ R2- -N-R3 a. Secondary amine b. Secondary amide c. Tertiary amine d. Tertiary amidearrow_forwardpls helparrow_forwardIndicate the product of the reaction OH OH CH3-CC- Ph + H2SO4 a 20°C | CH3 Pharrow_forward
- 35) Complete the following equation by drawing the line the structure of the products that are formed. Please note that in some cases more than one product is possible. You must draw all possible products to recive full marks! a. ethanol + 2-propanol + H2SO4 → b. OH conc. H2SO4 CH2 H3C CH + K2Cr2O7 C. d. H3C A pressure CH3 + H2 CH Pt catalystarrow_forward21) The rate of reaction depends upon: a. the concentration and nature of reactants b. the temperature of the reaction C. whether or not a catalyst was used d. all of the above 22) A Maxwell-Boltzmann curve shows the distribution of molecular energies in a reaction system. When the temperature in this system is increased, the peak is a. higher and further to the right. b. higher and further to the left. c. lower and further to the right. d. lower and further to the left. 23) Which of the following correctly describes the reaction represented by the reaction below? CaCO3 (s) + energy → CaO (s) + CO2 (g) a. It is exothermic and the potential energy is greater in the reactants than the products. b. c. It is exothermic and the potential energy is greater in the products than the reactants. It is endothermic and the potential energy is greater in the products than the reactants. d. It is endothermic and the potential energy is equal for the products and reactants.arrow_forwardpls helparrow_forward
- 27) Draw the energy level diagram and write the full and shorthand electron configuration for a neutral sulfur atom.arrow_forwardIndicate whether these compounds are isomers, enantiomers, or tautomers. OCH OCH محمد ممدarrow_forward30) Substance A to E below are listed with several of their properties. The identities of the substances are identified in random order below: Iron, ethane, ethanol, sodium nitrate, graphite First classify each substance as either a polar covalent compound, non-polar covalent compound, ionic compound, metallic solid, or network solid. Write your predictions in the sixth coloumn of the chart, under "type of substance." Then, identify the identity of the substance in the last coloumn. Substance Melting Point Boiling Point Solubility in H₂O Electrical Conductivity Type of Substance Identity of Substance (°C) (°C) as: Solid, Liquids, Solution A -182 -88 Insoluble No/No/- B 1538 2862 Insoluble Yes/Yes/- C 308 380 Soluble Yes/Yes/Yes Ꭰ 3456 Insoluble No/-/- E -114 78 Soluble No/No/Noarrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning



