
Concept explainers
(a)
Interpretation:
The structure of alcohol that could be used to prepare the given compound by oxidation reaction has to be drawn.
Concept Introduction:
In
In organic chemistry, reduction reaction is referred to the number
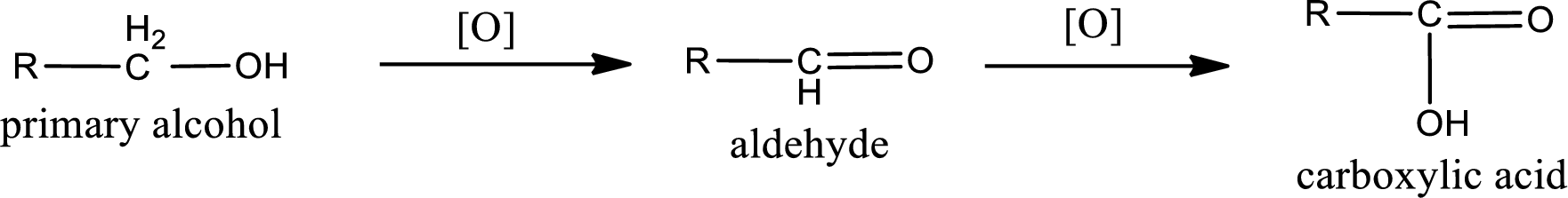
Alcohols do undergo
Primary alcohol:
Secondary alcohol:
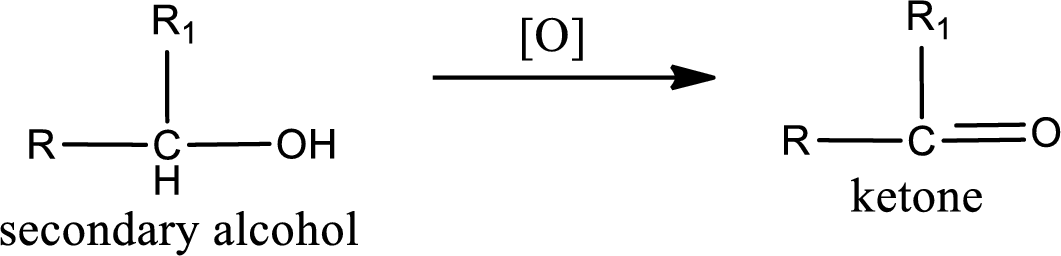
(a)
Answer to Problem 14.68EP
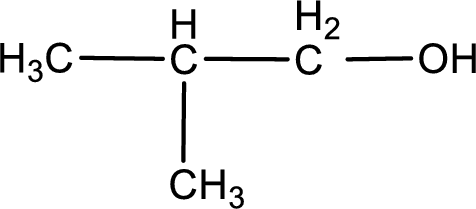
The structure of alcohol is,
Explanation of Solution
In organic chemistry, oxidation reaction is said to be decrease in
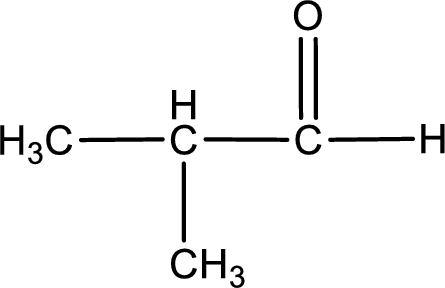
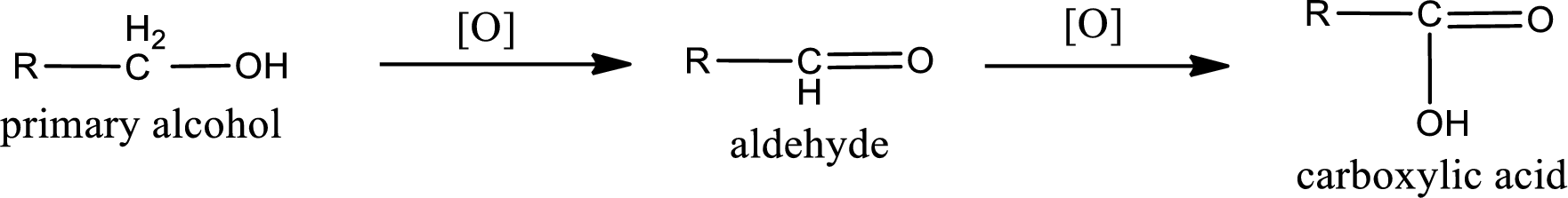
The given compound is a aldehyde. The aldehyde must have formed from primary alcohol by oxidation. The structure of the primary alcohol can be drawn considering the general scheme. Therefore, the structure can be given as shown below,
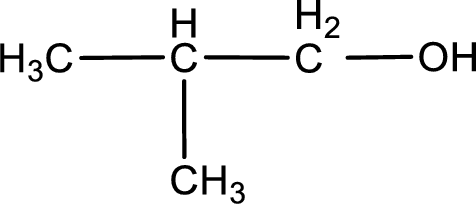
Structure of alcohol is drawn from which the given product is obtained by oxidation.
(b)
Interpretation:
The structure of alcohol that could be used to prepare the given compound by oxidation reaction has to be drawn.
Concept Introduction:
In organic chemistry, oxidation reaction is referred to the number
In organic chemistry, reduction reaction is referred to the number
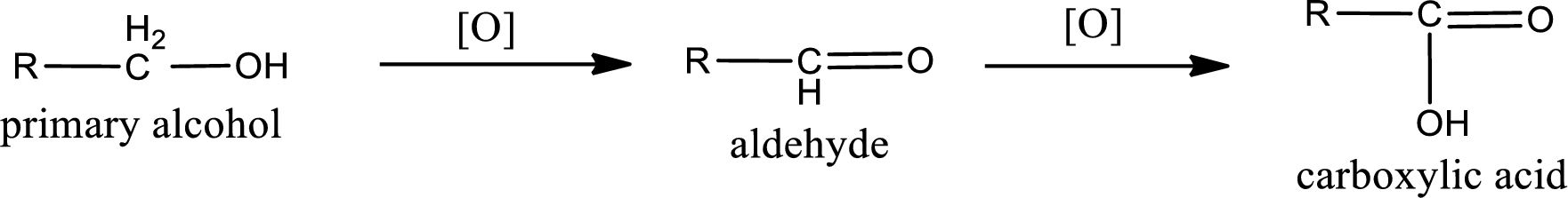
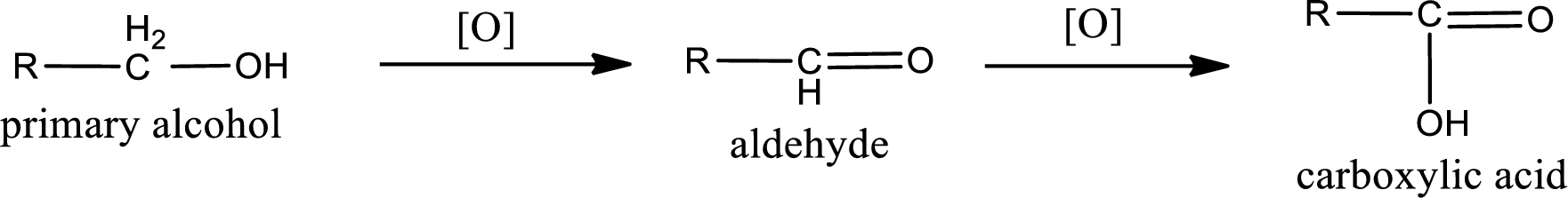
Alcohols do undergo oxidation reaction and reduction reaction. This depends upon the number of hydrogen atoms that is bonded to the alpha carbon atom. Primary and secondary alcohol undergoes oxidation reaction while tertiary alcohol does not undergo oxidation reaction. Primary alcohols undergo oxidation to give aldehyde and this id further oxidized to give carboxylic acid as product. Secondary alcohol undergoes oxidation to give ketone as the product.
Primary alcohol:
Secondary alcohol:
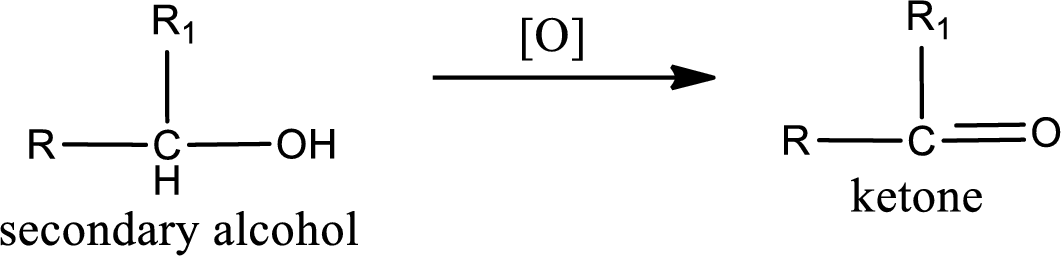
(b)
Answer to Problem 14.68EP
The structure of alcohol is,
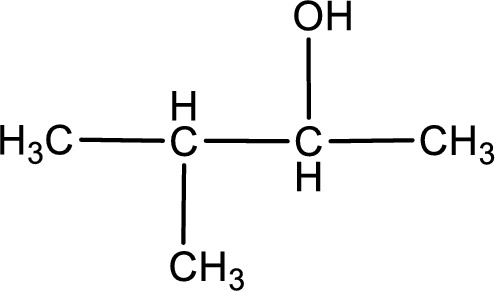
Explanation of Solution
In organic chemistry, oxidation reaction is said to be decrease in
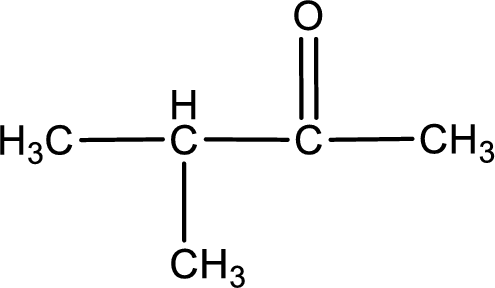
The given compound is a ketone. This has to be formed from a secondary alcohol only on oxidation. The structure of the secondary alcohol can be drawn considering the general scheme. Therefore, the structure can be given as shown below,
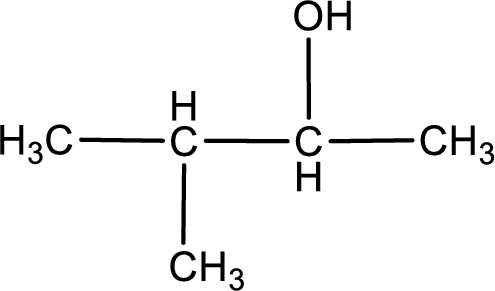
Structure of alcohol is drawn from which the given product is obtained by oxidation.
(c)
Interpretation:
The structure of alcohol that could be used to prepare the given compound by oxidation reaction has to be drawn.
Concept Introduction:
In organic chemistry, oxidation reaction is referred to the number
In organic chemistry, reduction reaction is referred to the number
Alcohols do undergo oxidation reaction and reduction reaction. This depends upon the number of hydrogen atoms that is bonded to the alpha carbon atom. Primary and secondary alcohol undergoes oxidation reaction while tertiary alcohol does not undergo oxidation reaction. Primary alcohols undergo oxidation to give aldehyde and this id further oxidized to give carboxylic acid as product. Secondary alcohol undergoes oxidation to give ketone as the product.
Primary alcohol:
Secondary alcohol:
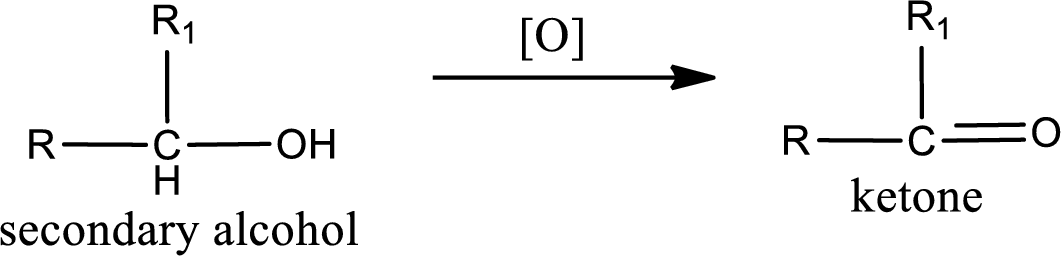
(c)
Answer to Problem 14.68EP
The structure of alcohol is,
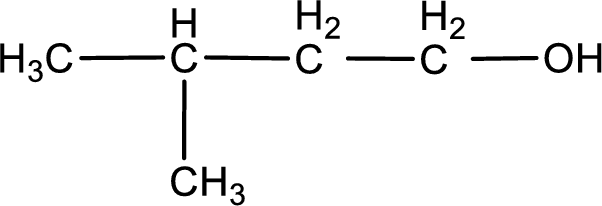
Explanation of Solution
In organic chemistry, oxidation reaction is said to be decrease in
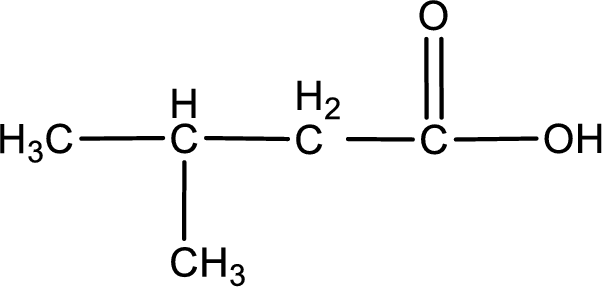
The given compound is a carboxylic acid. This has to be formed from aldehyde by oxidation. The aldehyde must have formed from primary alcohol by oxidation. The structure of the primary alcohol can be drawn considering the general scheme. Therefore, the structure can be given as shown below,
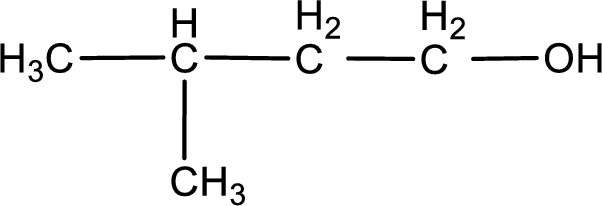
Structure of alcohol is drawn from which the given product is obtained by oxidation.
(d)
Interpretation:
The structure of alcohol that could be used to prepare the given compound by oxidation reaction has to be drawn.
Concept Introduction:
In organic chemistry, oxidation reaction is referred to the number
In organic chemistry, reduction reaction is referred to the number
Alcohols do undergo oxidation reaction and reduction reaction. This depends upon the number of hydrogen atoms that is bonded to the alpha carbon atom. Primary and secondary alcohol undergoes oxidation reaction while tertiary alcohol does not undergo oxidation reaction. Primary alcohols undergo oxidation to give aldehyde and this id further oxidized to give carboxylic acid as product. Secondary alcohol undergoes oxidation to give ketone as the product.
Primary alcohol:
Secondary alcohol:
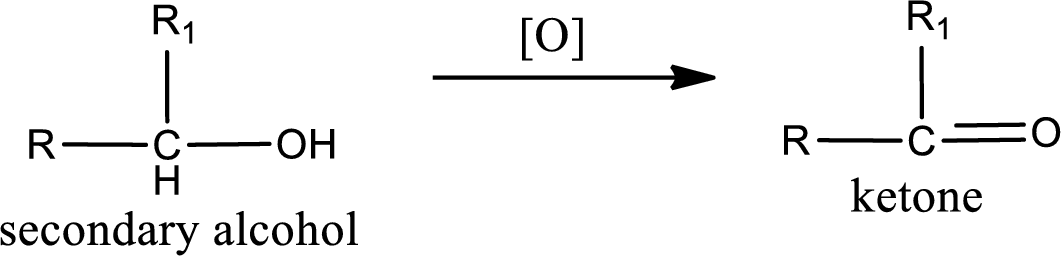
(d)
Answer to Problem 14.68EP
The structure of alcohol is,
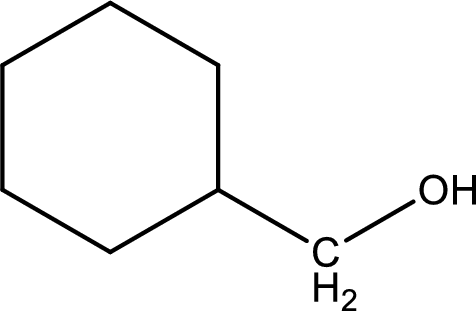
Explanation of Solution
In organic chemistry, oxidation reaction is said to be decrease in
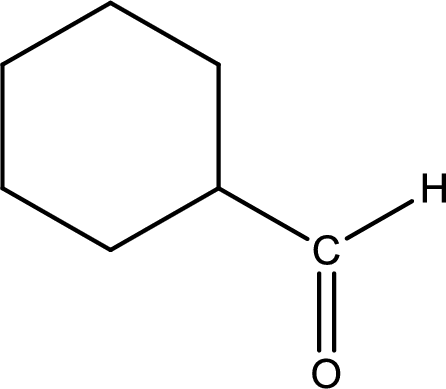
The given compound is a aldehyde. The aldehyde must have formed from primary alcohol by oxidation. The structure of the primary alcohol can be drawn considering the general scheme. Therefore, the structure can be given as shown below,
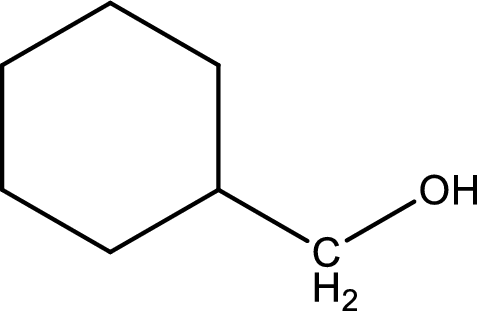
Structure of alcohol is drawn from which the given product is obtained by oxidation.
Want to see more full solutions like this?
Chapter 14 Solutions
General, Organic, And Biological Chemistry, Hybrid (with Owlv2 Quick Prep For General Chemistry Printed Access Card)
- Can you please help me solve these homework questionsarrow_forwardAn einstein is the amount of energy needed to dissociate 1 mole of a substance. If we have 0.58 moles, do we need 0.58 einsteins to dissociate that substance?arrow_forwardIf the energy absorbed per mole of gas is 480 kJ mol-1, indicate the number of Einsteins per mole.Data: Energy of each photon: 0.7835x10-18 J.arrow_forward
- If the energy absorbed per mole of gas is 480 kJ mol-1, indicate the number of Einsteins per mole.arrow_forwardThe quantum yield of the photochemical decay of HI is 2. Calculating the moles of HI per kJ of radiant energy can be decayed knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forwardThe quantum yield of the photochemical decay of HI is 2. Calculate the number of Einsteins absorbed per mole knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forward
- The quantum yield of the photochemical decay of HI is 2. How many moles of HI per kJ of radiant energy can be decayed knowing that the energy absorbed per mole of photons is 490 kJ.arrow_forwardIf the energy absorbed per mole of photons is 450 kJ, the number of Einsteins absorbed per 1 mole.arrow_forwardWhen propionic aldehyde in vapor form at 200 mmHg and 30°C is irradiated with radiation of wavelength 302 nm, the quantum yield with respect to the formation of CO is 0.54. If the intensity of the incident radiation is 1.5x10-3 W, find the rate of formation of CO.arrow_forward
 Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





