
Concept explainers
a.
Interpretation:
Chiral centers in the given monosaccharide has to be labelled, the monosaccharide has to be classified as D or L, the enantiomer of the given monosaccharide has to be drawn and also the Fischer projection formula has to be given.
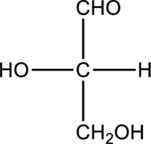
Concept Introduction:
A carbon atom that is bonded to four different groups is known as a chiral carbon atom. This can rotate the plane polarized light. D- and L- isomers of monosaccharide can be identified by looking into the chiral center that is farther from the carbonyl group. In a Fischer projection, if the
Enantiomers are two stereoisomers of a compound that rotate the plane polarized light exactly opposite. The configuration present in the enantiomers will be exactly opposite to each other.
Fischer projection formula simply uses cross for representing tetrahedral carbon atom. The carbon atom that is present in the intersection point in cross. The horizontal bonds means they are coming forward and they are present on wedge bond. The vertical bonds means they are pointing away and they are present on dashed lines.
b.
Interpretation:
Chiral centers in the given monosaccharide has to be labelled, the monosaccharide has to be classified as D or L, the enantiomer of the given monosaccharide has to be drawn and also the Fischer projection formula has to be given.
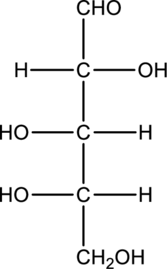
Concept Introduction:
Refer part “a.”.
c.
Interpretation:
Chiral centers in the given monosaccharide has to be labelled, the monosaccharide has to be classified as D or L, the enantiomer of the given monosaccharide has to be drawn and also the Fischer projection formula has to be given.
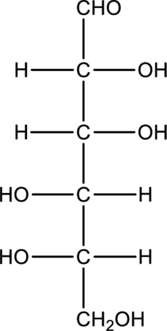
Concept Introduction:
Refer part “a.”.
Want to see the full answer?
Check out a sample textbook solution
Chapter 14 Solutions
Connect 1-Semester Online Access for Principles of General, Organic & Biochemistry
- Steps and explanations. Also provide, if possible, ways to adress this kind of problems in general.arrow_forwardk₁ Given the reaction A B, indicate k-1 d[A] (A). the rate law with respect to A: (B). the rate law with respect to B: d[B] dt dtarrow_forwardk₁ Given the reaction R₂ R + R, indicate k-1 (A). the rate law with respect to R2: (B). the rate law with respect to R: d[R₂] dt d[R] dtarrow_forward
- Given the reaction R+ Q → P, indicate (A). the rate law with respect to P: (B). the rate law with respect to R: (C). the rate law with respect to Q: d[P] dt d[R] dt d[Q] dtarrow_forwardThe reaction for obtaining NO2 from NO and O2 has the rate equation: v = k[NO]2[O2]. Indicate which of the following options is correct.(A). This rate equation is inconsistent with the reaction consisting of a single trimolecular step.(B). Since the overall order is 3, the reaction must necessarily have some trimolecular step in its mechanism.(C). A two-step mechanism: 1) NO + NO ⇄ N2O2 (fast); 2) N2O2 + O2 → NO2 + NO2 (slow).(D). The mechanism must necessarily consist of three unimolecular elementary steps with very similar rate constants.arrow_forwarda. What is the eluent used in the column chromatography here (a “silica plug filtration” is essentially a very short column)? b. The spectroscopy of compound 5b is described in the second half of this excerpt, including 1H-NMR and 13C-NMR (which you will learn about in CHEM 2412L), MS (which you will learn about later in CHEM 2411L) and IR. One of the IR signals is at 3530 cm-1. What functional group does this indicate might be present in compound 5b?arrow_forward
- Steps and explanations. Also provide, if possible, ways to adress this kind of problems in general.arrow_forwarda. The first three lines of this procedure describe the reaction used to make compound 5b. In the fourth line, hexane and sodium bicarbonate are added. What organic lab technique is being used here? b. What is the purpose of the Na2SO4? c. What equipment would you use to “concentrate [a solution] under reduced pressure”?arrow_forwardWhen N,N-dimethylaniline is treated with bromine both the ortho and para products are observed. However when treated with a mixture of nitric acid and sulfuric acid only the meta product is observed. Explain these results and support your answer with the appropriate drawings *Hint amines are bases* N HNO3 H2SO4 N NO2 N Br2 N Br + N 8-8-8 FeBr3 Brarrow_forward
- Draw a mechanism that explains the formation of compound OMe SO3H 1. Fuming H2SO4arrow_forwardConsider the following two acid-base reactions: OH OHI Based on what you know about the compounds and their acidity, which direction would you expect both of these reactions to proceed? Show your reasoning. A pKa table has been provided in case you need it. Functional group Example pka CHA -50 Alkane -35 Amine : NH3 Alkyne RH 25 Water HO-H 169 16 10 Protonated amines NH 10 5 Carboxylic acids OH Hydrochloric acid HCI A chemist intends to run the following reaction on the three substrates shown below: H₂O R-CI product room temp. Cl Cl (1) (2) (3) They find one will react quickly, one slowly, and one will not react at all. Which is which, and why? HINT: What is the reaction they're trying to do? Does that mechanism tell you anything about why something would be favored?arrow_forwardNH3 decomposes through an equilibrium reaction between NH3, H2, and N2. Only one of the options is correct:(A). The mechanism of the NH3 decomposition reaction must necessarily involve the collision of two NH3 molecules to induce a rearrangement of the atoms in this molecule.(B). The molecular weight of the NH3 decomposition reaction is 2 since two NH3 molecules must collide.(C). The rate of the NH3 decomposition reaction must be greater than that of NH3 synthesis, since the former requires two molecules to collide and the latter, four.(D). The NH3 decomposition reaction cannot occur in a single step.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





