
The data in the table are for the reaction of NO and O2 at 660 K.
NO(g) + ½ O2(g) → NO2(g)
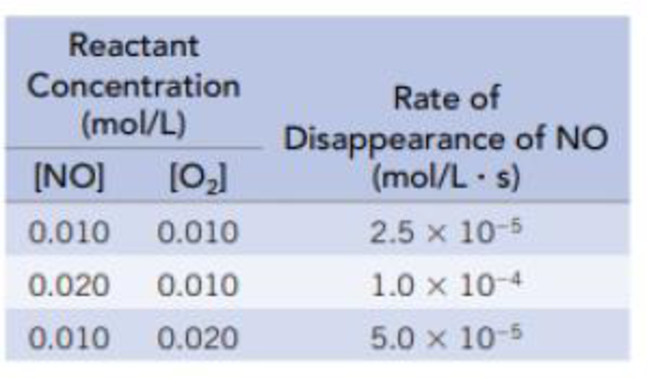
- (a) Determine the order of the reaction for each reactant.
- (b) Write the rate equation for the reaction.
- (c) Calculate the rate constant.
- (d) Calculate the rate (in mol/L · s) at the instant when [NO] = 0.015 mol/L and [O2] = 0.0050 mol/L.
- (e) At the instant when NO is reacting at the rate 1.0 × 10−4 mol/L · s, what is the rate at which O2 is reacting and NO2 is forming?
(a)
Interpretation:
The order of the reaction for each reactant has to be determined
Concept Introduction:
Rate law or rate equation: The relationship between the reactant concentrations and reaction rate is expressed by an equation.
Order of a reaction: The order of a reaction with respect to a particular reactant is the exponent of its concentration term in the rate law expression, and the overall reaction order is the sum of the exponents on all concentration terms.
Rate constant, k: It is a proportionality constant that relates rate and concentration at a given temperature.
Answer to Problem 11PS
The order of
Explanation of Solution
The reaction rate of the chemical reaction is given as,
In order to figure out the reaction equation the order of the reactants needed, which is calculated by comparing any two experiments where the concentration of
(b)
Interpretation:
The rate equation for the reaction has to be written.
Concept Introduction:
Rate law or rate equation: The relationship between the reactant concentrations and reaction rate is expressed by an equation.
Order of a reaction: The order of a reaction with respect to a particular reactant is the exponent of its concentration term in the rate law expression, and the overall reaction order is the sum of the exponents on all concentration terms.
Rate constant, k: It is a proportionality constant that relates rate and concentration at a given temperature.
Answer to Problem 11PS
The rate equation is
Explanation of Solution
The reaction rate is given as,
Hence, Rate equation is
(c)
Interpretation:
The rate constant has to be calculated.
Concept Introduction:
Rate law or rate equation: The relationship between the reactant concentrations and reaction rate is expressed by an equation.
Order of a reaction: The order of a reaction with respect to a particular reactant is the exponent of its concentration term in the rate law expression, and the overall reaction order is the sum of the exponents on all concentration terms.
Rate constant, k: It is a proportionality constant that relates rate and concentration at a given temperature.
Answer to Problem 11PS
The value of rate constant is
Explanation of Solution
The rate constant is calculated as,
The rate constant value is obtained as shown above. By substituting the any one of the concentrations of reactants and the initial rate into the reaction equation obtained at first.
Hence, the value of rate constant is
(d)
Interpretation:
The rate in
Concept Introduction:
Rate law or rate equation: The relationship between the reactant concentrations and reaction rate is expressed by an equation.
Order of a reaction: The order of a reaction with respect to a particular reactant is the exponent of its concentration term in the rate law expression, and the overall reaction order is the sum of the exponents on all concentration terms.
Rate constant, k: It is a proportionality constant that relates rate and concentration at a given temperature.
Answer to Problem 11PS
The instantaneous rate of the reaction is
Explanation of Solution
The rate is calculated as,
The instantaneous rate of the reaction is
(e)
Interpretation:
The rate at which
Concept Introduction:
Rate law or rate equation: The relationship between the reactant concentrations and reaction rate is expressed by an equation.
Order of a reaction: The order of a reaction with respect to a particular reactant is the exponent of its concentration term in the rate law expression, and the overall reaction order is the sum of the exponents on all concentration terms.
Rate constant, k: It is a proportionality constant that relates rate and concentration at a given temperature.
Answer to Problem 11PS
The rate when oxygen reacting is
Explanation of Solution
The rate is calculated as,
The rate when oxygen reacting is
Want to see more full solutions like this?
Chapter 14 Solutions
Chemistry & Chemical Reactivity
Additional Science Textbook Solutions
Microbiology Fundamentals: A Clinical Approach
Fundamentals Of Thermodynamics
Campbell Essential Biology (7th Edition)
Biology: Life on Earth with Physiology (11th Edition)
Physical Science
Organic Chemistry
- What is the pH of the Tris buffer after the addition of 10 mL of 0.01M NaOH? How would I calculate this?arrow_forwardWhy do isopolianions form polymeric species with a defined molecular weight? What does it depend on?arrow_forwardWhat are isopolianions? Describe the structural unit of isopolianions.arrow_forward
- Justify the polymerization of vanadates VO43-, as a function of concentration and pH.arrow_forwardWhat is the preparation of 500 mL of 100mM MOPS buffer (pH=7.5) starting with 1 M MOPS and 1 M NaOH? How would I calculate the math?arrow_forwardIndicate the correct option.a) Isopolianions are formed around metallic atoms in a low oxidation state.b) Non-metals such as N, S, C, Cl, ... give rise to polyacids (oxygenated).c) Both are incorrect.arrow_forward
- 14. Which one of the compounds below is the major organic product obtained from the following series of reactions? Br OH OH CH3O™ Na+ H*, H₂O SN2 HO OH A B C D 0 Earrow_forwardWavelength (nm) I'm not sure what equation I can come up with other than the one generated with my graph. Can you please show me the calculations that were used to find this equation? Give an equation that relates energy to wavelength. Explain how you arrived at your equation. Wavelength Energy (kJ/mol) (nm) 350 341.8 420 284.8 470 254.5 530 225.7 580 206.3 620 192.9 700 170.9 750 159.5 Energy vs. Wavelength (Graph 1) 400 350 y=-0.4367x+470.82 300 250 200 150 100 50 O 0 100 200 300 400 500 600 700 800 Energy (kJ/mol)arrow_forward5. Draw molecular orbital diagrams for superoxide (O2¯), and peroxide (O2²-). A good starting point would be MO diagram for O2 given in your textbook. Then: a) calculate bond orders in superoxide and in peroxide; indicate which species would have a stronger oxygen-oxygen bond; b) indicate which species would be a radical. (4 points)arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





