
(a)
Interpretation:
The expected products obtained from the given reaction have to be predicted.
Concept introduction:
Esterification reaction and intermolecular esterification reactions:
General scheme for esterification reaction:
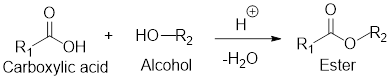
If the carboxylic acid and alcohol
Example for intermolecular esterification reaction:
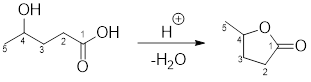
(b)
Interpretation:
The expected products obtained from the given reaction have to be predicted.
Concept introduction:
Transesterification and intermolecular transesterification reactions:
In the presence of an acid, ester and alcohol forms another new ester which is known as transester and a new alcohol will also be formed. This type of reaction is called transesterification reaction.
General scheme for transesterification reaction:
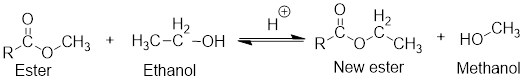
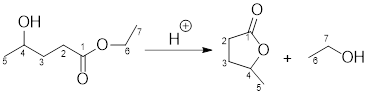
If the ester and alcohol are present within the same compound, then cyclic ester is formed which is known as lactone and the reaction is known as intermolecular transesterification reaction.
Example for intermolecular transesterification reaction:
Want to see the full answer?
Check out a sample textbook solution
Chapter 11 Solutions
Pearson eText for Essential Organic Chemistry -- Instant Access (Pearson+)

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

