
(a)
Interpretation: The molecular formulas of the compound represented by data points A, D, and E needs to be determined.
Concept Introduction: Empirical formula of a compound shows the simplest ratio of atoms present in the compound, on the other hand, the molecular formula shows the actual number of moles of atoms present in the compound.
(a)
Explanation of Solution
The empirical formula of the compound is given
Since the empirical formula is
Or,
Therefore, the molecular formula of the compound is
Now, from the given mass of carbon, the number of moles of carbon in the compound will be:
It confirms that there are 2 mol of carbon atoms in the compound. Thus, confirms the molecular formula equals to
Now, the molar mass corresponding point D is 150 g/mol. Using the empirical mass, the molecular formula can be determined as follows:
Or,
Therefore, the molecular formula of the compound is
Also, the point corresponding mass of carbon equals to 60 g. Now, from the given mass of carbon, the number of moles of carbon in the compound will be:
It confirms that there are 5 mol of carbon atoms in the compound. Thus, confirms the molecular formula equals to
Molar mass corresponding to point E is 180 g/mol. Using the empirical mass, the molecular formula can be determined as follows:
Or,
Therefore, the molecular formula of the compound is
Also, the point corresponds mass of carbon equals to 70 g. Now, from the given mass of carbon, the number of moles of carbon in the compound will be:
It confirms that there are 6 mol of carbon atoms in the compound. Thus, confirms the molecular formula equals to
Therefore, the molecular formula of compounds A, D, and E is
(b)
Interpretation: The slope of the line needs to be determined, and whether the value is consistent with the empirical formula or not needs to be determined.
Concept Introduction: An equation for a straight-line graph can be represented as follows:
Here, m is the slope of the graph and c is the intercept.
(b)
Explanation of Solution
Now, the given plot is a straight line; thus, the equation for the graph will be
Here, the intercept of the graph is zero; thus, the equation can be rewritten as follows:
Now, taking any one point say A, the molar mass of the compound is 60 g/mol, and the mass of the carbon atom is 25 g. Thus,
The value of m will be:
Thus, the slope of the graph is 2.5. Now, this is the ratio of the molar mass of the compound to the mass of carbon in the compound. From the slope, the mass percentage of carbon can be calculated as follows:
Yes, the value is constant with the empirical formula of the compound as the empirical formula is given
(c)
Interpretation: The value of x and y for the two other valid data points that fall on the same line between A and D needs to be determined.
Concept Introduction: From a graph, the new points on the line can be easily determined by checking drawing vertical and horizontal lines from that points to check where the values lie on the x and y-axis.
(c)
Explanation of Solution
From the graph, it is clear that the two points that fall on the same line between A and D will have the value of x and y as follows:
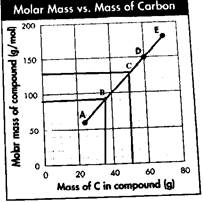
For B: The value of x, which is the mass of the carbon atom, will be 35 g, and the value of y, which is the molar mass of the compound, will be 90 g/mol. Therefore, the point on the graph will be B
For C: The value of x, which is the mass of the carbon atom, will be 50 g and the value of y, which is the molar mass of the compound, will be 130 g/mol. Therefore, the point on the graph will be C
Chapter 10 Solutions
Chemistry 2012 Student Edition (hard Cover) Grade 11
- NMR spectrum of ethyl acetate has signals whose chemical shifts are indicated below. Which hydrogen or set of hydrogens corresponds to the signal at 4.1 ppm? Select the single best answer. The H O HỌC—C—0—CH, CH, 2 A ethyl acetate H NMR: 1.3 ppm, 2.0 ppm, 4.1 ppm Check OA B OC ch B C Save For Later Submit Ass © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center |arrow_forwardHow many signals do you expect in the H NMR spectrum for this molecule? Br Br Write the answer below. Also, in each of the drawing areas below is a copy of the molecule, with Hs shown. In each copy, one of the H atoms is colored red. Highlight in red all other H atoms that would contribute to the same signal as the H already highlighted red Note for advanced students: In this question, any multiplet is counted as one signal. 1 Number of signals in the 'H NMR spectrum. For the molecule in the top drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. Check For the molecule in the bottom drawing area, highlight in red any other H atoms that will contribute to the same signal as the H atom already highlighted red. If no other H atoms will contribute, check the box at right. O ✓ No additional Hs to color in top molecule ง No additional Hs to color in bottom…arrow_forwardin the kinetics experiment, what were the values calculated? Select all that apply.a) equilibrium constantb) pHc) order of reactiond) rate contstantarrow_forward
- true or false, given that a 20.00 mL sample of NaOH took 24.15 mL of 0.141 M HCI to reach the endpoint in a titration, the concentration of the NaOH is 1.17 M.arrow_forwardin the bromothymol blue experiment, pKa was measured. A closely related compound has a Ka of 2.10 x 10-5. What is the pKa?a) 7.1b) 4.7c) 2.0arrow_forwardcalculate the equilibrium concentration of H2 given that K= 0.017 at a constant temperature for this reaction. The inital concentration of HBr is 0.050 M.2HBr(g) ↔ H2(g) + Br2(g)a) 4.48 x 10-2 M b) 5.17 x 10-3 Mc) 1.03 x 10-2 Md) 1.70 x 10-2 Marrow_forward
- true or falsegiven these two equilibria with their equilibrium constants:H2(g) + CI2(l) ↔ 2HCI(g) K= 0.006 CI2(l) ↔ CI2(g) K= 0.30The equilibrium contstant for the following reaction is 1.8H2(g) + CI2 ↔ 2HCI(g)arrow_forwardI2(g) + CI2(g) ↔ 2ICIK for this reaction is 81.9. Find the equilibrium concentration of I2 if the inital concentration of I2 and CI2 are 0.010 Marrow_forwardtrue or false,the equilibrium constant for this reaction is 0.50.PCI5(g) ↔ PCI3(g) + CI2(g)Based on the above, the equilibrium constant for the following reaction is 0.25.2PCI5(g) ↔. 2PCI3(g) + 2CI2(g)arrow_forward
- true or false, using the following equilibrium, if carbon dioxide is added the equilibrium will shift toward the productsC(s) + CO2(g) ↔ 2CO(g)arrow_forward2S2O2/3- (aq) + I2 (aq) ---> S4O2/6- (aq) +2I- (aq) Experiment I2 (M) S2O3- (M) Initital Rate (M/s) 1 0.01 0.01 0.0004 2 0.01 0.02 0.0004 3 0.02 0.01 0.0008 Calculate the overall order for this reaction using the table data a) 3b) 0c) 2d) 1arrow_forwardthe decomposition of N2O5 is the first order with a half-life of 1.98 minutes. If the inital concentration of N2O5 is 0.200 M, what is the concentration after 6 minutes?a) 0.612 Mb) 0.035 Mc) 0.024 Md) 0.100 Marrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





