
The wastewater treatment plant at the Ossabaw Paper Company paper mill generates about 24 tonnes of sludge per day. The consistency of the sludge is 35%, meaning that the sludge contains 35 wt% solids and the balance liquids. The mill currently spends $40/tonne to dispose of the sludge in a landfill. The plant environmental engineer has determined that if the sludge consistency could be increased to 75%, the sludge could be incinerated (burned) to generate useful energy and to eliminate the environmental problems associated with landfill disposal.
A flowchart for a preliminary design of the proposed sludge-treatment process follows. For simplicity, we will assume that the liquid in the sludge is just water.
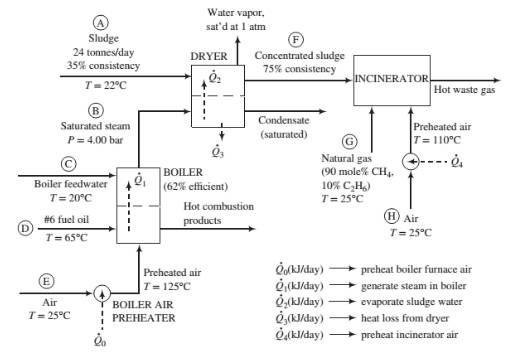 Process description: The sludge from the wastewater treatment plant (Stream (A) passes through a dryer where a portion of the water in the sludge is vaporized. The heat required for the vaporization comes from condensing saturated steam at 4.00 bar (Stream (B)). The steam fed to the dryer is produced in the plant’s oil-fired boiler from feedwater at 20°C (Stream (C)). The heat required to produce the steam is transferred from the boiler furnace, where fuel oil (Stream (D)) is burned with 25% excess air (Stream (E)). The concentrated sludge coming from the dryer (Stream (F)), which has a consistency of 75%, is fed to an incinerator. The heating value of the sludge is insufficient to keep the incinerator temperature high enough for complete combustion, so natural gas (Stream (G)) is used as a supplementary fuel. A stream of outside air at 25°C (Stream (H)) is heated to 110°C and fed to the incinerator along with the concentrated sludge and natural gas. The waste gas from the incinerator is discharged to the atmosphere.
Process description: The sludge from the wastewater treatment plant (Stream (A) passes through a dryer where a portion of the water in the sludge is vaporized. The heat required for the vaporization comes from condensing saturated steam at 4.00 bar (Stream (B)). The steam fed to the dryer is produced in the plant’s oil-fired boiler from feedwater at 20°C (Stream (C)). The heat required to produce the steam is transferred from the boiler furnace, where fuel oil (Stream (D)) is burned with 25% excess air (Stream (E)). The concentrated sludge coming from the dryer (Stream (F)), which has a consistency of 75%, is fed to an incinerator. The heating value of the sludge is insufficient to keep the incinerator temperature high enough for complete combustion, so natural gas (Stream (G)) is used as a supplementary fuel. A stream of outside air at 25°C (Stream (H)) is heated to 110°C and fed to the incinerator along with the concentrated sludge and natural gas. The waste gas from the incinerator is discharged to the atmosphere.
Fuel oil: The oil is a low-sulfur No. 6 fuel oil. Its ultimate (elemental) analysis on a weight basis is 87% C, 10% H, 0.84% S, and the balance oxygen, nitrogen, and nonvolatile ash. The higher heating value of the oil is 3.75 × 104kJ/kg and the heat capacity is Cp= 1.8 kJ/(kg·°C).
Boiler: The boiler has an efficiency of 62%, meaning that 62% of the heating value of the fuel oil burned is used to produce saturated steam at 4.00 bar from boiler feedwater at 20°C. Fuel oil at 65°C and dry air at 125°C are fed to the boiler furnace. The air feed rate is 25% in excess of the amount theoretically required for complete consumption of the fuel.
Sludge: The sludge from the wastewater treatment plant contains 35% w/w solids (S) and the balance liquids (which for the purposes of this problem may be treated as only water) and enters the dryer at 22°C. The sludge includes a number of volatile organic species, some of which may be toxic, and has a terrible odor. The heat capacity of the solids is approximately constant at 2.5 kJ/(kg·°C).
Dryer: The dryer has an efficiency of 55%, meaning that the heat transferred to the sludge, 02, is 55% of the total heat lost by the condensing steam, and the remainder,
Incinerator: The concentrated sludge has a heating value of 19,000 kJ/kg dry solids. For a feed sludge of 75% consistency, the incinerator requires 195 SCM natural gas/tonne wet sludge [ 1 SCM = 1 m3(STP)]. The theoretical air requirement for the sludge is 2.5 SCM air/10.000 kJ of heating value. Air is fed in 100% excess of the amount theoretically required to bum the sludge and the natural gas.
Use material and energy balances to calculate the mass flow rates (tonnes/day) of Streams (B), (C), (D), (E), (F), (G) and (H), and heat flows
- The money saved by implementing this process will be the current cost of disposing of the wastewater plant sludge in a landfill. Two major costs of implementing the process are the installed costs of the new dryer and incinerator. What other costs must be taken into account when determining the economic feasibility of the process? Why might management decide to go ahead with the project even if it proves to be unprofitable?
- What opportunities exist for improving the energy economy of the process? (Hint: Think about the need to preheat the fuel oil and the boiler and incinerator air streams and consider heat exchange possibilities.)
- The driving force for the introduction of this process is to eliminate the environmental cost of sludge disposal. What is that cost—that is, what environmental penalties and risks are associated with using landfills for hazardous waste disposal? What environmental problems might incineration introduce?
Learn your wayIncludes step-by-step video

Chapter 9 Solutions
Elementary Principles of Chemical Processes, Binder Ready Version
Additional Engineering Textbook Solutions
Problem Solving with C++ (10th Edition)
Java: An Introduction to Problem Solving and Programming (8th Edition)
Concepts Of Programming Languages
Management Information Systems: Managing The Digital Firm (16th Edition)
Database Concepts (8th Edition)
Web Development and Design Foundations with HTML5 (8th Edition)
- A semi-truck tire is inflated to 110 psig with nitrogen. What will be the initial gas discharge ratein lbm/s due to a 1/16-inch diameter hole? Assume at temperature of 80℉ and an ambientpressure of 1 atm.arrow_forward# 4 The reaction, AB, is to be carried out isothermally in a continuous flow reactor. The entering volumetric flow rate, vo is 10 L/h and is constant (v=vo). Calculate both the CSTR and PFR volumes necessary to reduce the entering concentration of species A from CAD to CA = 0.01 CAO when the entering molar flow rate of species A is 5 mol/h. (a) This reaction is a second order reaction. The reaction rate constant, k is given as 300 L/mol.h. (b) This reaction is a zeroth order reaction. The reaction rate constant, k is given as 0.05 mol/h.L.arrow_forward#3 Using the initial rates method and the given experimental data below to determine the rate law and the value of the rate constant for the reaction, as shown below. All trials are performed at the same temperature. 2NO + Cl2 → 2NCOCI Trial [NO] (mol/L) [Cl₂] (mol/L) Initial rates (mol/L.s) 1 0.10 0.10 0.00300 2 0.10 0.15 0.00450 3 0.15 0.10 0.00675arrow_forward
- #2 The reaction rate constant at temperature, T₁, is 15 mol/L-s while at the reaction rate constant changed to 7 mol/L-s when temperature changed to T2 at 398 K. What is T₁? Given the activation energy is 600 kJ/mol. Assume at this temperature interval, pre-exponential factor and activation energy are constant.arrow_forward#1 Chloral is consumed at a rate of 10 mol/L-s when reacting with chlorobenzene to form DDT and water in the reaction given below. Determine: i) the rate of disappearance of chlorobenzene. ii) the rate of formation of DDT. CCI CHO (Chloral) + 2C6H5Cl (Chlorobenzene) → (C6H4Cl)2CHCCI 3 (DDT) + H2Oarrow_forward#5 The irreversible liquid phase second order reaction, 2A → B, is carried out in a CSTR. The entering concentration of A, CAD is 2 mol/L, and the exit concentration of A, CA is 0.1 mol/L. The volumetric flow rate, vo, is at 3 L/s and is constant (v=vo). The reaction rate constant, k is 0.03 L/mol's. What is the corresponding reactor volume?arrow_forward
- Problem 9.11 An 80 mm long line MN has its end M 15 mm in front of the V.P. The distance between the ends projector is 50 mm. The front view is parallel to and 20 mm above reference line. Draw the projections of the line and determine its inclination with the V.P. Also, locate the traces. Interpretation Front view of a line is parallel to xy, therefore, 1. The line is parallel to the H.P. 2. The top view of the line has true length. 3. The front view has projected length equal to the distance be- tween the projectors. Construction Refer to Fig. 9.11. 1. Draw a reference line xy. Mark point m' 20 mm above xy and point m 15 mm below xy. 2. Draw a 50 mm long line m'n' parallel to xy. 3. Draw an arc with centre m and radius 80 mm to meet projec- tor from point n' at point n. Join mn to represent the top view. Determine its inclination with xy as the inclination of line MN with the V.P. Here = 51°. 4. Traces Extend line mn to meet xy at point v. Project point v to meet m'n' produced at…arrow_forwardoh 30 20 D и D P 60 60 80arrow_forward⑤ b Δε m ab C 40arrow_forward
- Problem 10.16 An isosceles triangle of base 40 mm and altitude 54 mm has its base in the V.P. The surface of the plane is inclined at 50° to the V.P. and perpendicular to the H.P. Draw its projections. Construction Refer to Fig. 10.17. An isosceles triangle has its base in the V.P., so con- sider that initially the triangle ABC is placed in the V.P. with base AB perpendicular to the H.P. 1. First stage Draw a triangle a'b'c' keeping a'b' perpendicular to xy to represent the front view. Project the corners to xy and obtain ac as the top view. 2. Second stage Reproduce the top view of first stage keeping ab on xy and ac inclined at 50° to xy. Obtain new points a', b' and c' in the front view by joining the points of intersection of the vertical projectors from a, b and c of the second stage with the corresponding horizontal locus lines from a', b' and c' of the first stage. Join a'b'c' to represent the final front view. Here, the front view is an equilateral triangle of side 40 mm. X 54…arrow_forward%9..+ ۱:۱۹ X خطأ عذرا ، الرقم الذي أدخلته خاطئ. يرجى إدخال رقم بطاقة الشحن الصالحة والمحاولة مرة أخرى. رصيد هاتفك قم بمسح الرمز = رقم بطاقة التعبئة 7794839909080 رمز مكون من 13 او 14 رقماً طريقة إعادة التعبئة قم باعادة تعبئة الرصيد إعادة تعبئة الإنترنت إعادة تعبئة الرصيد O >arrow_forwardProblem 10.14 A hexagonal plane of side 30 mm has a corner in the V.P. The surface of the plane is inclined at 45° to the V.P. and perpendicular to the H.P. Draw its projections. Assume that the diagonal through the corner in the V.P. is parallel to the H.P. d' a 2 b b.f C' c.e b 'C' H.P. (a) V.P E HEX 30 e' O' d' a a' b' C' b' X y a b,f c,e d b,f (b) c,earrow_forward
 Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education
Introduction to Chemical Engineering Thermodynami...Chemical EngineeringISBN:9781259696527Author:J.M. Smith Termodinamica en ingenieria quimica, Hendrick C Van Ness, Michael Abbott, Mark SwihartPublisher:McGraw-Hill Education Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...Chemical EngineeringISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
Elements of Chemical Reaction Engineering (5th Ed...Chemical EngineeringISBN:9780133887518Author:H. Scott FoglerPublisher:Prentice Hall
 Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning
Industrial Plastics: Theory and ApplicationsChemical EngineeringISBN:9781285061238Author:Lokensgard, ErikPublisher:Delmar Cengage Learning Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The
Unit Operations of Chemical EngineeringChemical EngineeringISBN:9780072848236Author:Warren McCabe, Julian C. Smith, Peter HarriottPublisher:McGraw-Hill Companies, The





