
(a)
Interpretation: Out of the substrates for the below reactions the one pair that will give a higher
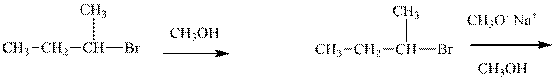
Concept introduction: Carbocation formation is relatively slower than acid-base reactions. Carbocations generated from
Analogous to the case that strong nucleophiles are more favored to react via
(b)
Interpretation: Out of the substrates for the reactions the one pair that will give a higher
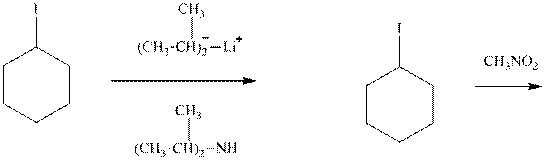
Concept introduction: Carbocation formation is relatively slower than acid-base reactions. Carbocations generated from alkyl halides have two fates; they can be either trapped by nucleophiles to give substitution product or may deprotonated to yield small amount of alkene. Stronger the base more is the probability of elimination over substitution over elimination. Further, if still higher concentration is employed reaction proceeds via bimolecular elimination. On the other hand, weak base waits until the carbocation is formed and the type of elimination with relatively weak base is two-step elimination or
Analogous to the case that strong nucleophiles are more favored to react via
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Organic Chemistry: Structure and Function
- 2. A graph shown below shows first ionization energies for elements from H to Ne. First ionization energy/kJ mol 2500 2000 1500 1000 500 T T T T 1 2 3 5 6 7 8 9 10 Atomic number a) Using arguments of electronic structure, explain why ionization energy of Li is much lower than that of H. (2 points) then dips at O. b) Using the same arguments, explain why ionization energy increases from B to N, and (3 points)arrow_forwardGive the name of this compound, including stereochemistry if relevant: CICH2 CH3 Br CH₂CH=CH2 Write in the product, including stereochemistry where relevant, for these reactions. See end of ch. 8, p. 301-303. 1. 03 a) 2-methyl-2-pentene -> 2. Zn, H* Br2 b) 1-ethylcyclopentene -->arrow_forwardNonearrow_forward
- 3. You may want to read paragraph 1.5 in your textbook before answering this question. Give electron configuration (short-hand notation is fine) for: (5 points) 3+ a) Manganese atom and Mn³+ b) Se atom c) Cu atom and Cu+arrow_forwardPlease correct answer and don't use hand ratingarrow_forwardNonearrow_forward
- Nonearrow_forwardHowever, why are intermolecular forces in metallic and ionic compounds not discussed as extensively? Additionally, what specific types of intermolecular attractions exist in metals and ionic compoundsarrow_forwardWhat is the preparation of 1 Liter of 0.1M NH4Cl buffer at pH 9.0 with solid NH4Cl and 0.1M NaOH. How would I calculate the math to describe this preparation? How would I use Henderson-Hasselbach equation?arrow_forward
- C Predict the major products of this organic reaction. Be sure you use wedge and dash bonds when necessary, for example to distinguish between major products with different stereochemistry. : ☐ + x G C RCO₂H Click and drag to start drawing a structure.arrow_forwardFill in the blanks by selecting the appropriate term from below: For a process that is non-spontaneous and that favors products at equilibrium, we know that a) ΔrG∘ΔrG∘ _________, b) ΔunivSΔunivS _________, c) ΔsysSΔsysS _________, and d) ΔrH∘ΔrH∘ _________.arrow_forwardHighest occupied molecular orbital Lowest unoccupied molecular orbital Label all nodes and regions of highest and lowest electron density for both orbitals.arrow_forward

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

