
a)
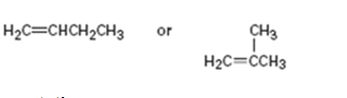
Interpretation:
The names of the
Concept introduction:
The longest carbon chain containing the double bond to be chosen. Based on the name of the parent compound - the alkene name ends with the suffix -ene. The chain is to be numbered from the end that gives the lowest number to the carbon in double bond. Substituents are to be numbered according to their positions in the chain and listed alphabetically. The position of the double bond is indicated by giving the number of the first alkene carbon before the name of the parent. The stability of alkenes can be explained using the concept of hyperconjucation or based on the bond strengths. Further a compound with more number of sp3-sp2 bonds is more stable.
To name:
The alkene given and to state which is more stable.
b)
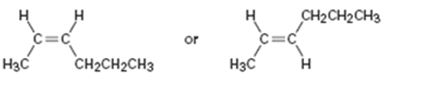
Interpretation:
The names of the alkenes given are to be given. Among the two compounds which is more stable also is to be stated.
Concept introduction:
The longest carbon chain containing the double bond to be chosen. Based on the name of the parent compound - the alkene name ends with the suffix -ene. The chain is to be numbered from the end that gives the lowest number to the carbon in double bond. Substituents are to be numbered according to their positions in the chain and listed alphabetically. The position of the double bond is indicated by giving the number of the first alkene carbon before the name of the parent. Among cis and trans isomers, the cis isomer is less stable than the trans isomer, because of the steric strain between the two larger substituents on the same side of the double bond.
To name:
The alkene given and to state which is more stable.
c)
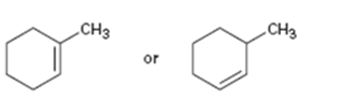
Interpretation:
The names of the alkenes given are to be given. Among the two compounds which is more stable also is to be stated.
Concept introduction:
The maximum number of carbons in the ring is counted. Based on the name of the parent cycloalkane - the cycloalkene is named with the suffix -ene. The cycloalkane is numbered such that the double bond is in between C1 & C2 and the first substituent has the lowest number possible. Usually the position of a double bond is not shown in the name because it is always between C1 & C2. The stability of cycloalkenes can be explained using the concept of hyper conjucation or based on the bond strengths.
To name:
The alkene given and to state which is more stable.
Trending nowThis is a popular solution!

Chapter 7 Solutions
ORGANIC CHEMISTRY-EBOOK>I<
- What is the reaction mechanism for this?arrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s). Be sure to account for all bond-breaking and bond-making steps. + Drawing Arrows CH3ONA, CH3OH heat : Br:O Na → H H Br Na + H H H H H :0: .H + Undo Reset Done Q CH3 Drag To Pan +arrow_forwardWhat is the reaction mechanism for this?arrow_forward
- 20.19 Predict the structure of the major 1,2-addition product formed by reaction of one mole of Cl₂ with 3-methylenecyclohexene. Also predict the structure of the 1,4-addition product formed under these conditions. 20.20 Which of the two molecules shown do you expect to be the major product formed by 1,2-addition of HCI to cyclopentadiene? Explain. Cyclopentadiene + HC 3-Chlorocyclopentene (racemic) or 4-Chlorocyclopentene (racemic)arrow_forward20.35 Propose structural formulas for compounds A and B and specify the configuration of compound B. EtO₂C 250°C C14H2004 CO₂Et 1. Oso, then NaHSO3 2. HIO4 C14H2006 A Barrow_forward20.21 Predict the major product formed by 1,4-addition of HCI to cyclopentadiene. 20.22 Draw structural formulas for the two constitutional isomers with the molecular for- mula C₂H,Br, formed by adding one mole of Br, to cyclopentadiene.arrow_forward
- Add substituents to draw the conformer below (sighting down the indicated bond), then rotate the back carbon to provide the conformation that will be capable of an E2 elimination. R/S stereochemistry is graded. + I I H CH3 Ph Досн Br OCH 3 Drawing Q H Atoms, Bonds and Rings Charges Tap a node to see suggestions. H H H H H Undo Reset Remove Done Rotatearrow_forward20.17 Predict the structure of the major product formed by 1,2-addition of HBr to 3-methylenecyclohexene. 3-Methylenecyclohexene 20.18 Predict the major product formed by 1,4-addition of HBr to 3-methylenecyclohexene.arrow_forward+ Draw a vicinal alkyl bromide that would produce the following alkene in an E2 elimination. Use a dash or wedge bond to indicate stereochemistry on asymmetric centers, where applicable. Ignore any inorganic byproducts. Br Drawing Strong Base H Q Atoms, Bonds Charges and Rings Draw or tap a new bond to see suggestions. Remove Done 語 Reset Undo + Drag To Panarrow_forward
