
(a)
Interpretation:
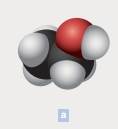
The chemical formula of each of the following is to be written. In parts a, the black spheres represent carbon atoms; the red, oxygen atoms; and the white, hydrogen atoms.
Concept introduction:
The particles possessing by chemical compounds are usually represented by chemical formulas. Besides, the chemical formula of a substance is having the
Answer to Problem 1PE
The chemical formula of the compound is C2H6O
Explanation of Solution
The chemical compounds are made up of polyatomic ions and the ratio of positive to negative ions present in the substance is expressed as formula of ionic compound. In the above example, the black spheres represent carbon atoms; the red, oxygen atoms; and the white represents hydrogen atoms. From the diagram, we come to know that,
Number of carbon atoms = 2
Number of hydrogen atoms = 6
Number of oxygen atom = 1
Thus, the molecular formula is C2H6O
Thus, the chemical formula of compound (a) is written.
(b)
Interpretation:
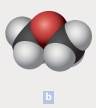
The chemical formula of each of the following is to be written. In parts b, the black spheres represent carbon atoms; the red, oxygen atoms; and the white, hydrogen atoms.
Concept introduction:
The particles possessing by chemical compounds are usually represented by chemical formulas. Besides, the chemical formula of a substance is having the symbols of element using which the chemical compound is made. Generally, in a chemical formula, the subscript number after a symbol denotes the number of atoms of the element in the formula unit of the substance. If the compound is having only one atom of an element of interest, the subscript is omitted.
Answer to Problem 1PE
The chemical formula of the compound is C2H6O
Explanation of Solution
The chemical compounds are made up of polyatomic ions and the ratio of positive to negative ions present in the substance is expressed as formula of ionic compound. In the above example, the black spheres represent carbon atoms; the red, oxygen atoms; and the white represents hydrogen atoms. From the diagram, we come to know that,
Number of carbon atoms = 2
Number of hydrogen atoms = 6
Number of oxygen atom = 1
Thus, the molecular formula is C2H6O
Thus, the chemical formula of compound (b) is written.
(c)
Interpretation:
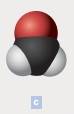
The chemical formula of each of the following is to be written. In parts c, the black spheres represent carbon atoms; the red, oxygen atoms; and the white, hydrogen atoms.
Concept introduction:
The particles possessing by chemical compounds are usually represented by chemical formulas. Besides, the chemical formula of a substance is having the symbols of element using which the chemical compound is made. Generally, in a chemical formula, the subscript number after a symbol denotes the number of atoms of the element in the formula unit of the substance. If the compound is having only one atom of an element of interest, the subscript is omitted.
Answer to Problem 1PE
The chemical formula of the compound is CH2O
Explanation of Solution
The chemical compounds are made up of polyatomic ions and the ratio of positive to negative ions present in the substance is expressed as formula of ionic compound. In the above example, the black spheres represent carbon atoms; the red, oxygen atoms; and the white represents hydrogen atoms. From the diagram, we come to know that,
Number of carbon atoms = 1
Number of hydrogen atoms = 2
Number of oxygen atom = 1
Thus, the molecular formula is CH2O
Thus, the chemical formula of compound (c) is written.
(d)
Interpretation:
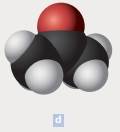
The chemical formula of each of the following is to be written. In parts d, the black spheres represent carbon atoms; the red, oxygen atoms; and the white, hydrogen atoms.
Concept introduction:
The particles possessing by chemical compounds are usually represented by chemical formulas. Besides, the chemical formula of a substance is having the symbols of element using which the chemical compound is made. Generally, in a chemical formula, the subscript number after a symbol denotes the number of atoms of the element in the formula unit of the substance. If the compound is having only one atom of an element of interest, the subscript is omitted.
Answer to Problem 1PE
The chemical formula of the compound is C3H6O
Explanation of Solution
The chemical compounds are made up of polyatomic ions and the ratio of positive to negative ions present in the substance is expressed as formula of ionic compound. In the above example, the black spheres represent carbon atoms; the red, oxygen atoms; and the white represents hydrogen atoms. From the diagram, we come to know that,
Number of carbon atoms = 3
Number of hydrogen atoms = 6
Number of oxygen atom = 1
Thus, the molecular formula is C3H6O
Thus, the chemical formula of compound (d) is written.
(e)
Interpretation:
The chemical formula of each of the following is to be written. In parts e, the compound made up of molecules with one nitrogen atom and three hydrogen atoms
Concept introduction:
The particles possessing by chemical compounds are usually represented by chemical formulas. Besides, the chemical formula of a substance is having the symbols of element using which the chemical compound is made. Generally, in a chemical formula, the subscript number after a symbol denotes the number of atoms of the element in the formula unit of the substance. If the compound is having only one atom of an element of interest, the subscript is omitted.
Answer to Problem 1PE
The chemical formula of the compound is NH3
Explanation of Solution
The chemical compounds are made up of polyatomic ions and the ratio of positive to negative ions present in the substance is expressed as formula of ionic compound. In the above example, the black spheres represent carbon atoms; the red, oxygen atoms; and the white represents hydrogen atoms. From the diagram, we come to know that,
Number of nitrogen atom = 1
Number of hydrogen atoms = 3
Thus, the molecular formula is NH3
Thus, the chemical formula of compound (e) is written.
(f)
Interpretation:
The chemical formula of each of the following is to be written. In parts f, the compound made up of a crystal with two particles coming from aluminium atoms for every three particles coming from oxygen atoms.
Concept introduction:
The particles possessing by chemical compounds are usually represented by chemical formulas. Besides, the chemical formula of a substance is having the symbols of element using which the chemical compound is made. Generally, in a chemical formula, the subscript number after a symbol denotes the number of atoms of the element in the formula unit of the substance. If the compound is having only one atom of an element of interest, the subscript is omitted.
Answer to Problem 1PE
The chemical formula of the compound is Al2O3
Explanation of Solution
The chemical compounds are made up of polyatomic ions and the ratio of positive to negative ions present in the substance is expressed as formula of ionic compound. In the above example, the black spheres represent carbon atoms; the red, oxygen atoms; and the white represents hydrogen atoms. From the diagram, we come to know that,
Number of aluminium atoms = 2
Number of oxygen atoms = 3
Thus, the molecular formula is Al2O3
Thus, the chemical formula of compound (f) is written.
Want to see more full solutions like this?
Chapter 6 Solutions
Introductory Chemistry: An Active Learning Approach
- Describe the principle of resonance and give a set of Lewis Structures to illustrate your explanation.arrow_forwardDon't used hand raitingarrow_forwardIt is not unexpected that the methoxyl substituent on a cyclohexane ring prefers to adopt the equatorial conformation. OMe H A G₂ = +0.6 kcal/mol OMe What is unexpected is that the closely related 2-methoxytetrahydropyran prefers the axial conformation: H H OMe OMe A Gp=-0.6 kcal/mol Methoxy: CH3O group Please be specific and clearly write the reason why this is observed. This effect that provides stabilization of the axial OCH 3 group in this molecule is called the anomeric effect. [Recall in the way of example, the staggered conformer of ethane is more stable than eclipsed owing to bonding MO interacting with anti-bonding MO...]arrow_forward
- 206 Pb 82 Express your answers as integers. Enter your answers separated by a comma. ▸ View Available Hint(s) VAΣ ΜΕ ΑΣΦ Np, N₁ = 82,126 Submit Previous Answers ? protons, neutronsarrow_forwardPlease draw the inverted chair forms of the products for the two equilibrium reactions shown below. Circle the equilibrium reaction that would have a AG = 0, i.e., the relative energy of the reactant (to the left of the equilibrium arrows) equals the relative energy of the product? [No requirement to show or do calculations.] CH3 CH3 HH CH3 1 -CH3arrow_forward5. Please consider the Newman projection of tartaric acid drawn below as an eclipsed conformer (1). Please draw the most stable conformer and two intermediate energy conformers noting that staggered conformers are lower in energy than eclipsed forms even if the staggered conformers have gauche relationships between groups. [Draw the substituents H and OH on the front carbons and H, OH and CO₂H on the back carbons based on staggered forms. -CO₂H is larger than -OH.] OH COH ICOOH COOH COOH 1 2 COOH COOH 3 4 Staggered Staggered Staggered (most stable) Indicate the number of each conformer above (1, 2, 3 and 4) that corresponds to the relative energies below. Ref=0 Rotation 6. (60 points) a. Are compounds 1 and 2 below enantiomers, diastereomers or identical? OH OH HO HO LOH HO HO OH 2 OH OH b. Please complete the zig-zag conformation of the compound (3R,4S)-3,4-dichloro-2,5-dimethylhexane by writing the respective atoms in the boxes. 3.arrow_forward
- The plutonium isotope with 144 neutrons Enter the chemical symbol of the isotope.arrow_forwardThe mass ratio of sodium to fluorine in sodium fluoride is 1.21:1. A sample of sodium fluoride produced 26.1 gg of sodium upon decomposition. How much fluorine was formed?arrow_forward32S 16 Enter your answers numerically separated by a comma. Np. Nn = 跖 ΟΙ ΑΣΦ Submit Request Answer ? protons, neutronsarrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





