
Concept explainers
Fill in the blanks in each line of the following table. The first line is already completed as an example.
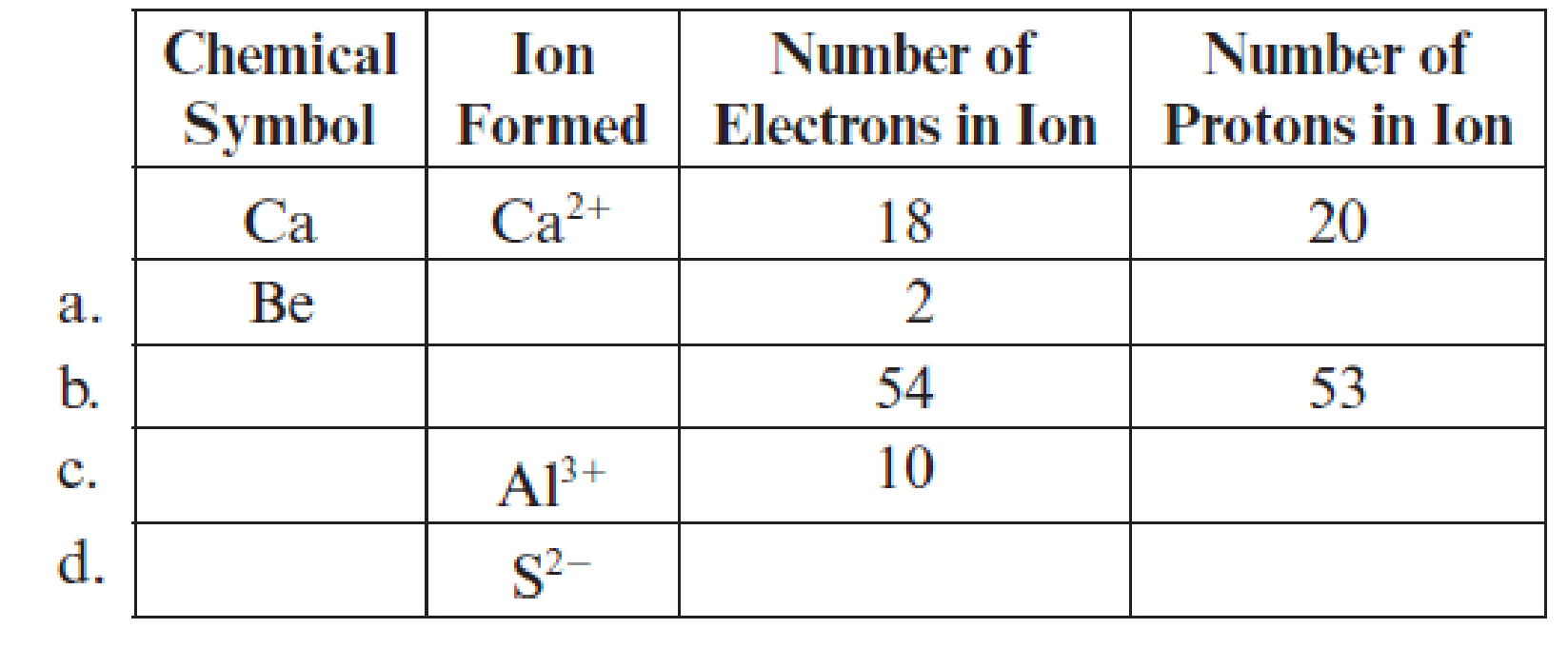
(a)
Interpretation:
Filling of each blank in the following table has to be done:
Concept Introduction:
Atoms are composed of three types of particles called subatomic particles. They are as follows:
- Protons: Positively charged particles in an atom.
- Neutrons: Neutral charged particles in an atom.
- Electrons: Negatively charged particles in an atom.
The neutral atom has equal number of protons and electrons. Gaining or loosing of electrons of an atom forms ion.
Negative charged ions are formed by gaining one or more electrons and it has more electrons than protons.
Positive charged ions are formed by losing one or more electrons and it has more protons than electrons.
Answer to Problem 4.29EP
Complete table is shown below:
Explanation of Solution
The chemical symbol of the element is
Two electrons are lesser than the total number protons thatmeans it lost two electrons and the charge of the beryllium ion is
Hence, the symbol of ion is
(b)
Interpretation:
Filling of each blank in the following table has to be done:
Concept Introduction:
Atoms are composed of three types of particles called subatomic particles. They are as follows:
- Protons: Positively charged particles in an atom.
- Neutrons: Neutral charged particles in an atom.
- Electrons: Negatively charged particles in an atom.
The neutral atom has equal number of protons and electrons. Gaining or loosing of electrons of an atom form ion.
Negative charged ions are formed by gaining one or more electrons and it has more electrons than protons.
Positive charged ions are formed by losing one or more electrons and it has more protons than electrons.
Answer to Problem 4.29EP
Complete table is shown below:
Explanation of Solution
The element has
Hence, the number of protons are
(c)
Interpretation:
Filling of each blank in the following table has to be done:
Concept Introduction:
Atoms are composed of three types of particles called subatomic particles. They are as follows:
- Protons: Positively charged particles in an atom.
- Neutrons: Neutral charged particles in an atom.
- Electrons: Negatively charged particles in an atom.
The neutral atom has equal number of protons and electrons. Gaining or loosing of electrons of an atom forms ion.
Negative charged ions are formed by gaining one or more electrons and it has more electrons than protons.
Positive charged ions are formed by losing one or more electrons and it has more protons than electrons.
Answer to Problem 4.29EP
Complete table is shown below:
Explanation of Solution
The ion
Hence, ion
(d)
Interpretation:
Filling of each blank in the following table has to be done:
Concept Introduction:
Atoms are composed of three types of particles called subatomic particles. They are as follows:
- Protons: Positively charged particles in an atom.
- Neutrons: Neutral charged particles in an atom.
- Electrons: Negatively charged particles in an atom.
The neutral atom has equal number of protons and electrons. Gaining or loosing of electrons of an atom forms ion.
Negative charged ions are formed by gaining one or more electrons and it has more electrons than protons.
Positive charged ions are formed by losing one or more electrons and it has more protons than electrons.
Answer to Problem 4.29EP
Complete table is shown below:
Explanation of Solution
The given ion is
The charge of sulfur atom is
Hence, ion is
Want to see more full solutions like this?
Chapter 4 Solutions
Study Guide with Selected Solutions for Stoker's General, Organic, and Biological Chemistry, 7th
- Part C IN H N. Br₂ (2 equiv.) AlBr3 Draw the molecule on the canvas by choosing buttons from the Tools (for bonds and + e (×) H± 12D T EXP. L CONT. דarrow_forward9. OA. Rank the expected boiling points of the compounds shown below from highest to lowest. Place your answer appropriately in the box. Only the answer in the box will be graded. (3) points) OH OH بر بد بدید 2 3arrow_forwardThere is an instrument in Johnson 334 that measures total-reflectance x-ray fluorescence (TXRF) to do elemental analysis (i.e., determine what elements are present in a sample). A researcher is preparing a to measure calcium content in a series of well water samples by TXRF with an internal standard of vanadium (atomic symbol: V). She has prepared a series of standard solutions to ensure a linear instrument response over the expected Ca concentration range of 40-80 ppm. The concentrations of Ca and V (ppm) and the instrument response (peak area, arbitrary units) are shown below. Also included is a sample spectrum. Equation 1 describes the response factor, K, relating the analyte signal (SA) and the standard signal (SIS) to their respective concentrations (CA and CIS). Ca, ppm V, ppm SCa, arb. units SV, arb. units 20.0 10.0 14375.11 14261.02 40.0 10.0 36182.15 17997.10 60.0 10.0 39275.74 12988.01 80.0 10.0 57530.75 14268.54 100.0…arrow_forward
- A mixture of 0.568 M H₂O, 0.438 M Cl₂O, and 0.710 M HClO are enclosed in a vessel at 25 °C. H₂O(g) + C₁₂O(g) = 2 HOCl(g) K = 0.0900 at 25°C с Calculate the equilibrium concentrations of each gas at 25 °C. [H₂O]= [C₁₂O]= [HOCI]= M Σ Marrow_forwardWhat units (if any) does the response factor (K) have? Does the response factor (K) depend upon how the concentration is expressed (e.g. molarity, ppm, ppb, etc.)?arrow_forwardProvide the structure, circle or draw, of the monomeric unit found in the biological polymeric materials given below. HO OH amylose OH OH 행 3 HO cellulose OH OH OH Ho HOarrow_forward
- OA. For the structure shown, rank the bond lengths (labeled a, b and c) from shortest to longest. Place your answer in the box. Only the answer in the box will be graded. (2 points) H -CH3 THe b Нarrow_forwardDon't used hand raitingarrow_forwardQuizzes - Gen Organic & Biological Che... ☆ myd21.lcc.edu + O G screenshot on mac - Google Search savings hulu youtube google disney+ HBO zlib Homework Hel...s | bartleby cell bio book Yuzu Reader: Chemistry G periodic table - Google Search b Home | bartleby 0:33:26 remaining CHEM 120 Chapter 5_Quiz 3 Page 1: 1 > 2 > 3 > 6 ¦ 5 > 4 > 7 ¦ 1 1 10 8 ¦ 9 a ¦ -- Quiz Information silicon-27 A doctor gives a patient 0.01 mC i of beta radiation. How many beta particles would the patient receive in I minute? (1 Ci = 3.7 x 10 10 d/s) Question 5 (1 point) Saved Listen 2.22 x 107 222 x 108 3.7 x 108 2.22 x 108 none of the above Question 6 (1 point) Listen The recommended dosage of 1-131 for a test is 4.2 μCi per kg of body mass. How many millicuries should be given to a 55 kg patient? (1 mCi = 1000 μСi)? 230 mCiarrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning





