
Study Guide with Selected Solutions for Stoker's General, Organic, and Biological Chemistry, 7th
7th Edition
ISBN: 9781305081086
Author: STOKER, H. Stephen
Publisher: Brooks Cole
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 4.99EP
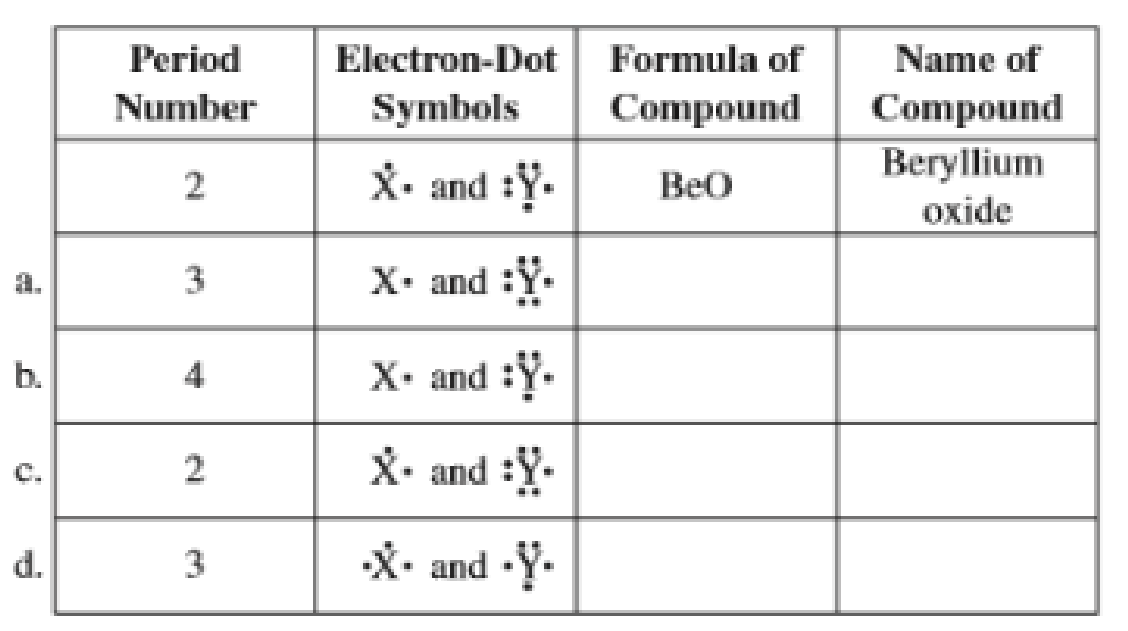
Fill in the blanks in each line of the following table that involves the representative elements X and Y. The first line is already completed as an example.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
In general, ionic compounds consist of metal and non-metal elements. Considering only this
characteristic, which of the following compounds is an ionic compound?
O HF
O NO₂
O WO3
O SO₂
Which of the following compounds is most likely to have parentheses as part of its
chemical formula?
O zinc peroxide
sulfur hexafluoride
O mercury (I) hydrogen phosphate
O aluminum cyanide
Complete the table below by writing the symbols for the cation and anion that make up each ionic compound. The first row has been completed for you.
Chapter 4 Solutions
Study Guide with Selected Solutions for Stoker's General, Organic, and Biological Chemistry, 7th
Ch. 4.1 - Prob. 1QQCh. 4.1 - Prob. 2QQCh. 4.1 - Prob. 3QQCh. 4.2 - How many valence electrons are present in an atom...Ch. 4.2 - Prob. 2QQCh. 4.2 - Prob. 3QQCh. 4.2 - Prob. 4QQCh. 4.2 - Which of the following elements would have the...Ch. 4.3 - Prob. 1QQCh. 4.3 - Prob. 2QQ
Ch. 4.3 - Prob. 3QQCh. 4.4 - In terms of subatomic particles, a Ca2+ ion...Ch. 4.4 - Prob. 2QQCh. 4.4 - Prob. 3QQCh. 4.4 - Prob. 4QQCh. 4.5 - An atom with a 1s22s22p4 electron configuration...Ch. 4.5 - Prob. 2QQCh. 4.5 - Prob. 3QQCh. 4.5 - Prob. 4QQCh. 4.5 - Prob. 5QQCh. 4.6 - Prob. 1QQCh. 4.6 - Prob. 2QQCh. 4.6 - Prob. 3QQCh. 4.7 - What is the chemical formula of the ionic compound...Ch. 4.7 - What is the chemical formula of the ionic compound...Ch. 4.7 - Given that Z2 ions are present in the ionic...Ch. 4.7 - Prob. 4QQCh. 4.8 - Prob. 1QQCh. 4.8 - Which of the following is a correct description of...Ch. 4.9 - Prob. 1QQCh. 4.9 - Prob. 2QQCh. 4.9 - Prob. 3QQCh. 4.9 - Prob. 4QQCh. 4.9 - Prob. 5QQCh. 4.9 - Prob. 6QQCh. 4.9 - The correct name for the binary ionic compound...Ch. 4.9 - Prob. 8QQCh. 4.10 - Prob. 1QQCh. 4.10 - Which of the following statements about polyatomic...Ch. 4.10 - The nitrate, sulfate, and phosphate ions have,...Ch. 4.10 - Prob. 4QQCh. 4.11 - Prob. 1QQCh. 4.11 - Prob. 2QQCh. 4.11 - Prob. 3QQCh. 4.11 - Prob. 4QQCh. 4.11 - Prob. 5QQCh. 4.11 - What is the chemical formula for the compound...Ch. 4 - Contrast the two general types of chemical bonds...Ch. 4 - Contrast the two general types of chemical...Ch. 4 - How many valence electrons do atoms with the...Ch. 4 - How many valence electrons do atoms with the...Ch. 4 - Prob. 4.5EPCh. 4 - Prob. 4.6EPCh. 4 - Write the complete electron configuration for each...Ch. 4 - Write the complete electron configuration for each...Ch. 4 - Prob. 4.9EPCh. 4 - For each of the following pairs of representative...Ch. 4 - How many of the highlighted elements in the...Ch. 4 - How many of the highlighted elements in the...Ch. 4 - Draw Lewis symbols for atoms of each of the...Ch. 4 - Draw Lewis symbols for atoms of each of the...Ch. 4 - Each of the following Lewis symbols represents a...Ch. 4 - Each of the following Lewis symbols represents a...Ch. 4 - Prob. 4.17EPCh. 4 - Prob. 4.18EPCh. 4 - What is the chemical property of the noble gases...Ch. 4 - Prob. 4.20EPCh. 4 - Prob. 4.21EPCh. 4 - Prob. 4.22EPCh. 4 - Give the chemical symbol for each of the following...Ch. 4 - Give the chemical symbol for each of the following...Ch. 4 - What would be the chemical symbol for an ion with...Ch. 4 - What would be the chemical symbol for an ion with...Ch. 4 - Fill in the blanks in each line in the following...Ch. 4 - Fill in the blanks in each line in the following...Ch. 4 - Fill in the blanks in each line of the following...Ch. 4 - Fill in the blanks in each line of the following...Ch. 4 - Identify element X by giving its chemical symbol,...Ch. 4 - Prob. 4.32EPCh. 4 - Prob. 4.33EPCh. 4 - Prob. 4.34EPCh. 4 - Prob. 4.35EPCh. 4 - Draw Lewis symbols for the following ions. a. O2...Ch. 4 - What is the charge on the monatomic ion formed by...Ch. 4 - What is the charge on the monatomic ion formed by...Ch. 4 - Indicate the number of electrons lost or gained...Ch. 4 - Indicate the number of electrons lost or gained...Ch. 4 - Which noble gas has an electron configuration...Ch. 4 - Prob. 4.42EPCh. 4 - Which noble gas is isoelectronic with each of the...Ch. 4 - Which noble gas is isoelectronic with each of the...Ch. 4 - Prob. 4.45EPCh. 4 - Indicate whether or not each of the following...Ch. 4 - Prob. 4.47EPCh. 4 - Prob. 4.48EPCh. 4 - Prob. 4.49EPCh. 4 - Write the electron configuration of the following....Ch. 4 - How many valence electrons are present in each of...Ch. 4 - Prob. 4.52EPCh. 4 - Using Lewis structures, show how ionic compounds...Ch. 4 - Using Lewis structures, show how ionic compounds...Ch. 4 - The following Lewis symbols for ions have the...Ch. 4 - Prob. 4.56EPCh. 4 - Prob. 4.57EPCh. 4 - Prob. 4.58EPCh. 4 - Prob. 4.59EPCh. 4 - Prob. 4.60EPCh. 4 - The component elements for four binary ionic...Ch. 4 - Prob. 4.62EPCh. 4 - Write the complete chemical formula (symbol and...Ch. 4 - Write the complete chemical formula (symbol and...Ch. 4 - Write the chemical formula for the ionic compound...Ch. 4 - Prob. 4.66EPCh. 4 - Prob. 4.67EPCh. 4 - What is the chemical formula of the ionic compound...Ch. 4 - A representative element (X) forms an ion with a 2...Ch. 4 - A representative element (Z) forms an ion with a...Ch. 4 - Prob. 4.71EPCh. 4 - The following questions pertain to the ionic...Ch. 4 - Prob. 4.73EPCh. 4 - Prob. 4.74EPCh. 4 - Prob. 4.75EPCh. 4 - Prob. 4.76EPCh. 4 - Prob. 4.77EPCh. 4 - Prob. 4.78EPCh. 4 - Prob. 4.79EPCh. 4 - Which of the following binary compounds would be...Ch. 4 - Name the following binary ionic compounds, each of...Ch. 4 - Name the following binary ionic compounds, each of...Ch. 4 - Calculate the charge on the metal ion in the...Ch. 4 - Calculate the charge on the metal ion in the...Ch. 4 - Prob. 4.85EPCh. 4 - Prob. 4.86EPCh. 4 - Prob. 4.87EPCh. 4 - Prob. 4.88EPCh. 4 - Name each of the following binary ionic compounds....Ch. 4 - Name each of the following binary ionic compounds....Ch. 4 - Prob. 4.91EPCh. 4 - Name each compound in the following pairs of...Ch. 4 - Prob. 4.93EPCh. 4 - Write chemical formulas for the following binary...Ch. 4 - Prob. 4.95EPCh. 4 - Write chemical formulas for the following binary...Ch. 4 - Prob. 4.97EPCh. 4 - Prob. 4.98EPCh. 4 - Fill in the blanks in each line of the following...Ch. 4 - Prob. 4.100EPCh. 4 - Prob. 4.101EPCh. 4 - Prob. 4.102EPCh. 4 - Prob. 4.103EPCh. 4 - How many oxygen atoms are present in each of the...Ch. 4 - Prob. 4.105EPCh. 4 - Prob. 4.106EPCh. 4 - Prob. 4.107EPCh. 4 - Prob. 4.108EPCh. 4 - Prob. 4.109EPCh. 4 - Prob. 4.110EPCh. 4 - How many ions are present per formula unit in each...Ch. 4 - Prob. 4.112EPCh. 4 - Name the following compounds, all of which contain...Ch. 4 - Prob. 4.114EPCh. 4 - Prob. 4.115EPCh. 4 - Prob. 4.116EPCh. 4 - Prob. 4.117EPCh. 4 - Write formulas for the following compounds, all of...Ch. 4 - Write chemical formulas for the following...Ch. 4 - Write chemical formulas for the following...Ch. 4 - Prob. 4.121EPCh. 4 - Prob. 4.122EPCh. 4 - Prob. 4.123EPCh. 4 - Prob. 4.124EP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1. Hydrogen is a Group I element and each hydrogen will contribute I valence electron. Carbon is a Group (Roman numeral) element and each carbon will contribute ___ (number) electrons. Every oxygen atom in a compound will contribute valence electrons. Chloromethane has the molecular formula . Its skeleton is and the number of valence electrons may be determined as follows. There are three hydrogen atoms, each of which contributes 1 valence electron; the single carbon contributes 4 electrons; and the single chlorine atom contributes 7 electrons, making a total of 14 . A convenient tabular form of this calculation isarrow_forwardFill in the blanks in each line in the following table. The first line is already completed as an example.arrow_forwardWrite the chemical formula of each of the following: a The compound made up of a crystal with one particle coming from a nickel atom for every two particles coming from chlorine atoms. b The compound made up of a crystal with two particles coming from silver atoms for every one particle coming from an oxygen atom. c The compound made up of molecules with six carbon atoms, 12 hydrogen atoms, and six oxygen atoms. d The compound made up of molecules with two hydrogen atoms, one sulfur atom, and four oxygen atoms.arrow_forward
- Which of the following is true about an individual atom? Explain. a. An individual atom should be considered to be a solid. b.An individual atom should be considered to be a liquid. c. An individual atom should be considered to be a gas. d. The state of the atom depends on which element it is. e. An individual atom cannot be considered to be a solid, liquid, or gas. Justify your choice, and for choices you did not pick, explain what is wrong with them.arrow_forwardWrite the chemical formula of each of the following: a The compound made up of a crystal with two particles coming from chromium atoms for every three particles coming from oxygen atoms. b The compound made up of a crystal with one particle coming from a barium atom for every two particles coming from chlorine atoms. c The compound made up of molecules with 12 carbon atoms, 22 hydrogen atoms, and 11 oxygen atoms. d The compound made up of molecules with three hydrogen atoms, one phosphorus atom, and four oxygen atoms.arrow_forwardTell what is wrong with each of the following molecular formulas and write a correct formula: a. H3PO3 phosphorous acid b. SICI4 silicon tetrachloride c. SOO sulfur dioxide d. 2HO hydrogen peroxide-two hydrogen atoms and two oxygen atomsarrow_forward
- For each of the following atomic numbers, use the periodic (able to write the formula (including the charge) for the simple ion that the element is most likely to form. a. 53 c. 55 e. 9 b. 38 d. 88 f. 1.13arrow_forwardThe isotope of an unknown element, X, has a mass number of 79. The most stable ion of the isotope has 36 electrons and forms a binary compound with sodium, having a formula of Na2X. Which of the following statements is(are) true? For the false statements, correct them. a. The binary compound formed between X and fluorine will be a covalent compound. b. The isotope of X contains 38 protons. c. The isotope of X contains 41 neutrons. d. The identity of X is strontium, Sr.arrow_forwardWhat are bus? How are ions formed from atoms? Do isolated atoms form ions spontaneously? To what do the termscationandanionrefer? In terms of subatomic particles, how is an ion related to the atom from which it is formed? Does the nucleus of an atom change when the atom is converted into an ion? How can the periodic table be used to predict what ion an element’s atoms will form?arrow_forward
- Name each of the following binary ionic compounds. a. AuCl b. KCl c. AgCl d. CuCl2arrow_forward7.101 Lead selenide nanocrystals may provide a breakthrough in the engineering of solar panels to be efficient enough to be an economical source of electricity. Selenium is generally considered a nonmetal while lead is considered a metal. Is this distinction enough to suggest that this compound should be ionic? Explain your answer.arrow_forwardWhat is the charge on the monatomic ion formed by each of the following elements? a. 3Li b. 15P c. 16S d. 13Alarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Atomic Number, Atomic Mass, and the Atomic Structure | How to Pass ChemistryThe Nucleus: Crash Course Chemistry #1; Author: Crash Course;https://www.youtube.com/watch?v=FSyAehMdpyI;License: Standard YouTube License, CC-BY