
OWLv2 with Student Solutions Manual eBook, 4 terms (24 months) Printed Access Card for McMurry's Organic Chemistry, 9th
9th Edition
ISBN: 9781305671874
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 31.1, Problem 2P
Order the following monomers with respect to their expected reactivity toward anionic
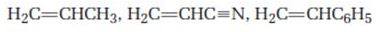
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Can you please explain this problem to me and expand it so I can understand the full Lewis dot structure? Thanks!
Can you please explain this problem to me and expand it so I can understand the full Lewis dot structure? Thanks!
Can you please explain this problem to me and expand it so I can understand the full Lewis dot structure? Thanks!
Chapter 31 Solutions
OWLv2 with Student Solutions Manual eBook, 4 terms (24 months) Printed Access Card for McMurry's Organic Chemistry, 9th
Ch. 31.1 - Order the following monomers with respect to their...Ch. 31.1 - Order the following monomers with respect to their...Ch. 31.1 - Prob. 3PCh. 31.2 - Prob. 4PCh. 31.2 - Prob. 5PCh. 31.3 - Prob. 6PCh. 31.3 - Prob. 7PCh. 31.4 - Prob. 8PCh. 31.4 - Show the mechanism of the nucleophilic addition...Ch. 31.5 - Prob. 10P
Ch. 31.6 - Prob. 11PCh. 31.6 - Prob. 12PCh. 31.SE - Prob. 13VCCh. 31.SE - Prob. 14VCCh. 31.SE - Prob. 15MPCh. 31.SE - The polyurethane foam used for home insulation...Ch. 31.SE - Prob. 17MPCh. 31.SE - Prob. 18MPCh. 31.SE - Prob. 19MPCh. 31.SE - Identify the monomer units from which each of the...Ch. 31.SE - Prob. 21APCh. 31.SE - Draw the structure of Kodel, a polyester prepared...Ch. 31.SE - Show the structure of the polymer that results...Ch. 31.SE - Prob. 24APCh. 31.SE - Prob. 25APCh. 31.SE - 1, 3-Cyclopentadiene undergoes thermal...Ch. 31.SE - Prob. 27APCh. 31.SE - Prob. 28APCh. 31.SE - Prob. 29APCh. 31.SE - Prob. 30APCh. 31.SE - Prob. 31APCh. 31.SE - Prob. 32APCh. 31.SE - Prob. 33APCh. 31.SE - Prob. 34APCh. 31.SE - Prob. 35AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Please answer the questions in the photos and please revise any wrong answers. Thank youarrow_forward(Please be sure that 7 carbons are available in the structure )Based on the 1H NMR, 13C NMR, DEPT 135 NMR and DEPT 90 NMR, provide a reasoning step and arrive at the final structure of an unknown organic compound containing 7 carbons. Dept 135 shows peak to be positive at 128.62 and 13.63 Dept 135 shows peak to be negative at 130.28, 64.32, 30.62 and 19.10.arrow_forward-lease help me answer the questions in the photo.arrow_forward
- For the reaction below, the concentrations at equilibrium are [SO₂] = 0.50 M, [0] = 0.45 M, and [SO3] = 1.7 M. What is the value of the equilibrium constant, K? 2SO2(g) + O2(g) 2SO3(g) Report your answer using two significant figures. Provide your answer below:arrow_forwardI need help with this question. Step by step solution, please!arrow_forwardZn(OH)2(s) Zn(OH)+ Ksp = 3 X 10-16 B₁ = 1 x 104 Zn(OH)2(aq) B₂ = 2 x 1010 Zn(OH)3 ẞ3-8 x 1013 Zn(OH) B4-3 x 1015arrow_forward
- Help me understand this by showing step by step solution.arrow_forwardscratch paper, and the integrated rate table provided in class. our scratch work for this test. Content attribution 3/40 FEEDBACK QUESTION 3 - 4 POINTS Complete the equation that relates the rate of consumption of H+ and the rate of formation of Br2 for the given reaction. 5Br (aq) + BrO3 (aq) + 6H (aq) →3Br2(aq) + 3H2O(l) • Your answers should be whole numbers or fractions without any decimal places. Provide your answer below: Search 尚 5 fn 40 * 00 99+ 2 9 144 a [arrow_forward(a) Write down the structure of EDTA molecule and show the complex structure with Pb2+ . (b) When do you need to perform back titration? (c) Ni2+ can be analyzed by a back titration using standard Zn2+ at pH 5.5 with xylenol orange indicator. A solution containing 25.00 mL of Ni2+ in dilute HCl is treated with 25.00 mL of 0.05283 M Na2EDTA. The solution is neutralized with NaOH, and the pH is adjusted to 5.5 with acetate buffer. The solution turns yellow when a few drops of indicator are added. Titration with 0.02299 M Zn2+ requires 17.61 mL to reach the red end point. What is the molarity of Ni2+ in the unknown?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
CBSE Class 12 Chemistry || Polymers || Full Chapter || By Shiksha House; Author: Best for NEET;https://www.youtube.com/watch?v=OxdJlS0xZ0Y;License: Standard YouTube License, CC-BY