
Organic Chemistry (8th Edition)
8th Edition
ISBN: 9780134042282
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 25, Problem 31P
Interpretation Introduction
Interpretation:
The structure of dipalmitoylphosphatidylcholine has to be given.
Expert Solution & Answer
Answer to Problem 31P
The structure of dipalmitoylphosphatidylcholine is given below:
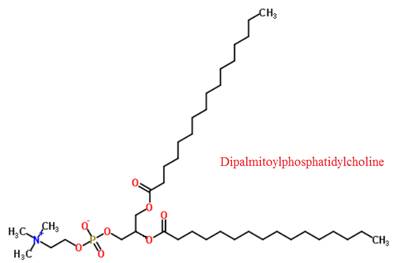
Explanation of Solution
Dipalmitoylphosphatidylcholine is a surfactant found in the lining of the lungs. This lining prevents the lungs from collapsing when the lung volume is low. The injuries caused by inhaling particles can be avoided by this surfactant.
The structure of dipalmitoylphosphatidylcholine is given below:
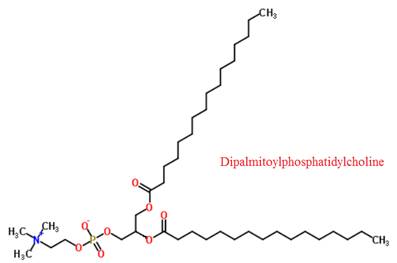
Conclusion
The structure of dipalmitoylphosphatidylcholine was given.
Want to see more full solutions like this?
Subscribe now to access step-by-step solutions to millions of textbook problems written by subject matter experts!
Students have asked these similar questions
3. Name this ether correctly.
H₁C
H3C
CH3
CH3
4. Show the best way to make the ether in #3 by a
Williamson Ether Synthesis.
Start from an alcohol or phenol.
5. Draw the structure of an example of a sulfide.
1. Which one(s) of these can be oxidized with CrO3 ?
(could be more than one)
a) triphenylmethanol
b) 2-pentanol
c) Ethyl alcohol
d)
CH3
2. Write in all the product(s) of this reaction. Label them
as "major" or "minor".
2-methyl-2-hexanol
H2SO4, heat
3) Determine if the pairs are constitutional isomers, enantiomers, diastereomers, or mesocompounds.
(4 points)
Chapter 25 Solutions
Organic Chemistry (8th Edition)
Ch. 25.1 - Prob. 1PCh. 25.3 - Which has a higher melting point, glyceryl...Ch. 25.3 - Draw the structure of an optically inactive fat...Ch. 25.3 - Draw the structure of an optically active fat...Ch. 25.5 - Do the identities of R1 and R2 in phosphatidic...Ch. 25.5 - Membranes contain proteins, Integral membrane...Ch. 25.5 - Prob. 8PCh. 25.5 - The membrane phospholipids in deer have a higher...Ch. 25.6 - Treating PGC2 with a strong base such as sodium...Ch. 25.7 - Mark off the isoprene units in menthol, -selinene,...
Ch. 25.7 - Prob. 13PCh. 25.7 - Prob. 14PCh. 25.8 - Propose mechanisms for the Claisen condensation...Ch. 25.8 - Prob. 16PCh. 25.8 - Propose a mechanism for the conversion of...Ch. 25.8 - Propose a mechanism for the biosynthesis of...Ch. 25.8 - Propose a mechanism for the conversion of the E...Ch. 25.8 - The fluoro-substitued geranyl pyrophosphate shown...Ch. 25.8 - Prob. 22PCh. 25.8 - Prob. 23PCh. 25.9 - Draw the individual 1,2-hydride and 1,2-methyl...Ch. 25.10 - Prob. 26PCh. 25.10 - Prob. 27PCh. 25.10 - The acid component of a cholesterol ester is a...Ch. 25.10 - Prob. 29PCh. 25.10 - Prob. 30PCh. 25 - Prob. 31PCh. 25 - An optically active fat, when completely...Ch. 25 - Prob. 33PCh. 25 - a. How many different triacylglycerols are there...Ch. 25 - Cardiolipins are found in heart muscles. Draw the...Ch. 25 - Nutmeg contains a simple, fully saturated...Ch. 25 - Draw the product that is obtained from the...Ch. 25 - Prob. 39PCh. 25 - Prob. 40PCh. 25 - Propose a mechanism for the biosynthesis of...Ch. 25 - 5-Androstene-3.17-dione is isomerized to...Ch. 25 - Prob. 44PCh. 25 - Eudesmol is a sesquiterpene found in eucalyptus....Ch. 25 - Prob. 46PCh. 25 - Prob. 47PCh. 25 - Diethylstilbestrol (DES) was given to pregnant...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- In the decomposition reaction in solution B → C, only species C absorbs UV radiation, but neither B nor the solvent absorbs. If we call At the absorbance measured at any time, A0 the absorbance at the beginning of the reaction, and A∞ the absorbance at the end of the reaction, which of the expressions is valid? We assume that Beer's law is fulfilled.arrow_forward> You are trying to decide if there is a single reagent you can add that will make the following synthesis possible without any other major side products: 1. ☑ CI 2. H3O+ O Draw the missing reagent X you think will make this synthesis work in the drawing area below. If there is no reagent that will make your desired product in good yield or without complications, just check the box under the drawing area and leave it blank. Click and drag to start drawing a structure. Explanation Check ? DO 18 Ar B © 2025 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibilityarrow_forwardDon't use ai to answer I will report you answerarrow_forward
- Consider a solution of 0.00304 moles of 4-nitrobenzoic acid (pKa = 3.442) dissolved in 25 mL water and titrated with 0.0991 M NaOH. Calculate the pH at the equivalence pointarrow_forwardWhat is the name of the following compound? SiMe3arrow_forwardK Draw the starting structure that would lead to the major product shown under the provided conditions. Drawing 1. NaNH2 2. PhCH2Br 4 57°F Sunny Q Searcharrow_forward
- 7 Draw the starting alkyl bromide that would produce this alkyne under these conditions. F Drawing 1. NaNH2, A 2. H3O+ £ 4 Temps to rise Tomorrow Q Search H2arrow_forward7 Comment on the general features of the predicted (extremely simplified) ¹H- NMR spectrum of lycopene that is provided below. 00 6 57 PPM 3 2 1 0arrow_forwardIndicate the compound formula: dimethyl iodide (propyl) sulfonium.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning


Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning