
General, Organic, and Biological Chemistry Seventh Edition
7th Edition
ISBN: 9781305767867
Author: H. Stephan Stoker
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 24.2, Problem 5QQ
Interpretation Introduction
Interpretation: To identify the number of carbon atoms and the
Concept introduction: Glycolysis is the
The carbonyl compound contains a double bond between carbon
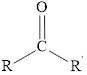
Here,
The chemical structure of carboxylic acid is as follows:
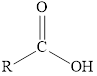
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What are the IUPAC Names of all the compounds in the picture?
1) a) Give the dominant Intermolecular Force (IMF) in a sample of each of the following
compounds. Please show your work. (8) SF2, CH,OH, C₂H₂
b) Based on your answers given above, list the compounds in order of their Boiling Point
from low to high. (8)
19.78 Write the products of the following sequences of reactions. Refer to your reaction road-
maps to see how the combined reactions allow you to "navigate" between the different
functional groups. Note that you will need your old Chapters 6-11 and Chapters 15-18
roadmaps along with your new Chapter 19 roadmap for these.
(a)
1. BHS
2. H₂O₂
3. H₂CrO4
4. SOCI₂
(b)
1. Cl₂/hv
2. KOLBU
3. H₂O, catalytic H₂SO4
4. H₂CrO4
Reaction
Roadmap
An alkene 5. EtOH
6.0.5 Equiv. NaOEt/EtOH
7. Mild H₂O
An alkane
1.0
2. (CH3)₂S
3. H₂CrO
(d)
(c)
4. Excess EtOH, catalytic H₂SO
OH
4. Mild H₂O*
5.0.5 Equiv. NaOEt/EtOH
An alkene 6. Mild H₂O*
A carboxylic
acid
7. Mild H₂O*
1. SOC₁₂
2. EtOH
3.0.5 Equiv. NaOEt/E:OH
5.1.0 Equiv. NaOEt
6.
NH₂
(e)
1. 0.5 Equiv. NaOEt/EtOH
2. Mild H₂O*
Br
(f)
i
H
An aldehyde
1. Catalytic NaOE/EtOH
2. H₂O*, heat
3. (CH,CH₂)₂Culi
4. Mild H₂O*
5.1.0 Equiv. LDA
Br
An ester
4. NaOH, H₂O
5. Mild H₂O*
6. Heat
7.
MgBr
8. Mild H₂O*
7. Mild H₂O+
Chapter 24 Solutions
General, Organic, and Biological Chemistry Seventh Edition
Ch. 24.1 - The primary site within the human body where...Ch. 24.1 - What is the first site within the human body where...Ch. 24.1 - What effect does gastric juice in the stomach have...Ch. 24.1 - Prob. 4QQCh. 24.1 - Which of the following substances is needed for...Ch. 24.2 - Prob. 1QQCh. 24.2 - Prob. 2QQCh. 24.2 - Prob. 3QQCh. 24.2 - Prob. 4QQCh. 24.2 - Prob. 5QQ
Ch. 24.2 - Prob. 6QQCh. 24.2 - Prob. 7QQCh. 24.3 - Prob. 1QQCh. 24.3 - Prob. 2QQCh. 24.3 - Prob. 3QQCh. 24.3 - Prob. 4QQCh. 24.3 - Accumulation of which of the following substances...Ch. 24.4 - Prob. 1QQCh. 24.4 - The net yield of ATP for the complete oxidation of...Ch. 24.4 - Prob. 3QQCh. 24.5 - Prob. 1QQCh. 24.5 - Prob. 2QQCh. 24.5 - Prob. 3QQCh. 24.6 - Prob. 1QQCh. 24.6 - Prob. 2QQCh. 24.6 - Prob. 3QQCh. 24.6 - Which of the following statements about ATP...Ch. 24.6 - Prob. 5QQCh. 24.7 - Prob. 1QQCh. 24.7 - Prob. 2QQCh. 24.8 - Prob. 1QQCh. 24.8 - Prob. 2QQCh. 24.8 - Prob. 3QQCh. 24.9 - Which of the following hormones promotes the...Ch. 24.9 - Which of the following pairs of hormones increases...Ch. 24.10 - Prob. 1QQCh. 24.10 - Prob. 2QQCh. 24.10 - Prob. 3QQCh. 24 - Where does carbohydrate digestion begin in the...Ch. 24 - Very little digestion of carbohydrates occurs in...Ch. 24 - Prob. 24.3EPCh. 24 - Prob. 24.4EPCh. 24 - Prob. 24.5EPCh. 24 - Prob. 24.6EPCh. 24 - Prob. 24.7EPCh. 24 - Prob. 24.8EPCh. 24 - Prob. 24.9EPCh. 24 - Prob. 24.10EPCh. 24 - Prob. 24.11EPCh. 24 - Prob. 24.12EPCh. 24 - Prob. 24.13EPCh. 24 - Prob. 24.14EPCh. 24 - Prob. 24.15EPCh. 24 - Prob. 24.16EPCh. 24 - Prob. 24.17EPCh. 24 - Prob. 24.18EPCh. 24 - Prob. 24.19EPCh. 24 - Prob. 24.20EPCh. 24 - Prob. 24.21EPCh. 24 - Prob. 24.22EPCh. 24 - Prob. 24.23EPCh. 24 - Prob. 24.24EPCh. 24 - Prob. 24.25EPCh. 24 - Prob. 24.26EPCh. 24 - Prob. 24.27EPCh. 24 - Prob. 24.28EPCh. 24 - Prob. 24.29EPCh. 24 - Prob. 24.30EPCh. 24 - Prob. 24.31EPCh. 24 - Prob. 24.32EPCh. 24 - Prob. 24.33EPCh. 24 - Prob. 24.34EPCh. 24 - Prob. 24.35EPCh. 24 - Prob. 24.36EPCh. 24 - Prob. 24.37EPCh. 24 - Prob. 24.38EPCh. 24 - Prob. 24.39EPCh. 24 - Prob. 24.40EPCh. 24 - Prob. 24.41EPCh. 24 - Prob. 24.42EPCh. 24 - Prob. 24.43EPCh. 24 - Prob. 24.44EPCh. 24 - Prob. 24.45EPCh. 24 - Prob. 24.46EPCh. 24 - Prob. 24.47EPCh. 24 - Prob. 24.48EPCh. 24 - Prob. 24.49EPCh. 24 - Prob. 24.50EPCh. 24 - Prob. 24.51EPCh. 24 - Prob. 24.52EPCh. 24 - Prob. 24.53EPCh. 24 - Prob. 24.54EPCh. 24 - Prob. 24.55EPCh. 24 - Prob. 24.56EPCh. 24 - Prob. 24.57EPCh. 24 - Prob. 24.58EPCh. 24 - Prob. 24.59EPCh. 24 - Prob. 24.60EPCh. 24 - Prob. 24.61EPCh. 24 - Prob. 24.62EPCh. 24 - Prob. 24.63EPCh. 24 - Prob. 24.64EPCh. 24 - Prob. 24.65EPCh. 24 - The liver, but not the brain or muscle cells, has...Ch. 24 - Prob. 24.67EPCh. 24 - Prob. 24.68EPCh. 24 - Prob. 24.69EPCh. 24 - Prob. 24.70EPCh. 24 - Prob. 24.71EPCh. 24 - Prob. 24.72EPCh. 24 - Prob. 24.73EPCh. 24 - Prob. 24.74EPCh. 24 - Prob. 24.75EPCh. 24 - Prob. 24.76EPCh. 24 - Prob. 24.77EPCh. 24 - Prob. 24.78EPCh. 24 - Prob. 24.79EPCh. 24 - Prob. 24.80EPCh. 24 - Prob. 24.81EPCh. 24 - Prob. 24.82EPCh. 24 - Prob. 24.83EPCh. 24 - Prob. 24.84EPCh. 24 - Prob. 24.85EPCh. 24 - Prob. 24.86EPCh. 24 - Prob. 24.87EPCh. 24 - Prob. 24.88EPCh. 24 - Prob. 24.89EPCh. 24 - Prob. 24.90EPCh. 24 - Prob. 24.91EPCh. 24 - Prob. 24.92EPCh. 24 - Prob. 24.93EPCh. 24 - Prob. 24.94EPCh. 24 - Prob. 24.95EPCh. 24 - Prob. 24.96EPCh. 24 - Prob. 24.97EPCh. 24 - Prob. 24.98EPCh. 24 - Prob. 24.99EPCh. 24 - Prob. 24.100EPCh. 24 - Prob. 24.101EPCh. 24 - Prob. 24.102EPCh. 24 - Prob. 24.103EPCh. 24 - Prob. 24.104EPCh. 24 - Prob. 24.105EPCh. 24 - Prob. 24.106EPCh. 24 - Prob. 24.107EPCh. 24 - Prob. 24.108EPCh. 24 - Prob. 24.109EPCh. 24 - Prob. 24.110EPCh. 24 - Prob. 24.111EPCh. 24 - Prob. 24.112EPCh. 24 - Prob. 24.113EPCh. 24 - Prob. 24.114EPCh. 24 - Prob. 24.115EPCh. 24 - Compare the biological functions of glucagon and...Ch. 24 - Prob. 24.117EPCh. 24 - Prob. 24.118EP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Li+ is a hard acid. With this in mind, which if the following compounds should be most soluble in water? Group of answer choices LiBr LiI LiF LiClarrow_forwardQ4: Write organic product(s) of the following reactions and show the curved-arrow mechanism of the reactions. Br MeOH OSO2CH3 MeOHarrow_forwardProvide the correct IUPAC name for the compound shown here. Reset cis- 5- trans- ☑ 4-6- 2- 1- 3- di iso tert- tri cyclo sec- oct but hept prop hex pent yl yne ene anearrow_forward
- Q6: Predict the major product(s) for the following reactions. Note the mechanism (SN1, SN2, E1 or E2) the reaction proceeds through. If no reaction takes place, indicate why. Pay attention to stereochemistry. NaCN DMF Br σ Ilm... Br H Br H H NaCN CH3OH KOtBu tBuOH NaBr H₂O LDA Et2O (CH3)2CHOH KCN DMSO NaOH H₂O, A LDA LDA Systemarrow_forwardQ7: For the following reactions, indicate the reaction conditions that would provide the indicated product in a high yield. Note the major reaction pathway that would take place (SN1, SN2, E1, or E2) Note: There may be other products that are not shown. There maybe more than one plausible pathway. Br H3C OH H3C CI ... H3C SCH2CH3 CI i SCH2CH3 ཨ་ Br System Settarrow_forwardQ2: Rank the compounds in each of the following groups in order of decreasing rate of solvolysis in aqueous acetone. OSO2CF3 OSO2CH3 OH a. b. CI Brarrow_forward
- ох 4-tert-butyl oxy cyclohex-1-ene Incorrect, 1 attempt remaining The systematic name of this compound classifies the -OR group as a substituent of the hydrocarbon, which is considered the principal functional group. The ether substituent is named with the suffix 'oxy'. The general format for the systematic name of a hydrocarbon is: [prefix/substituent] + [parent] + [functional group suffix] Substituents are listed in alphabetical order. Molecules with a chiral center will indicate the absolute configuration at the beginning of its name with the R and S notation.arrow_forward5. Compressibility (6 points total). The isothermal compressibility is a measure of how hard/easy it is to compress an object (how squishy is it?) at constant temperature. It is др defined as Br=-()=-(200²)T' (a) You might wonder why there is a negative sign in this formula. What does it mean when this quantity is positive and what does it mean when this quantity is negative? (b) Derive the formula for the isothermal compressibility of an ideal gas (it is very simple!) (c) Explain under what conditions for the ideal gas the compressibility is higher or lower, and why that makes sense.arrow_forward19. (3 pts) in Chapter 7 we will see a reaction of halocyclohexanes that requires that the halogen occupy an axial position with this in mind, would you expect cis-1-bromo-3-methylcyclohexane or trans-1-bromo-3-methylcyclohexane to be more reactive in this reaction? Briefly explain your choice using structures to support your answer. Mere-eries-cecleone) The tran-i-browse-3-methylcyclohexionearrow_forward
- Please help me calculate the undiluted samples ppm concentration. My calculations were 280.11 ppm. Please see if I did my math correctly using the following standard curve. Link: https://mnscu-my.sharepoint.com/:x:/g/personal/vi2163ss_go_minnstate_edu/EVSJL_W0qrxMkUjK2J3xMUEBHDu0UM1vPKQ-bc9HTcYXDQ?e=hVuPC4arrow_forwardProvide an IUPAC name for each of the compounds shown. (Specify (E)/(Z) stereochemistry, if relevant, for straight chain alkenes only. Pay attention to commas, dashes, etc.) H₁₂C C(CH3)3 C=C H3C CH3 CH3CH2CH CI CH3 Submit Answer Retry Entire Group 2 more group attempts remaining Previous Nextarrow_forwardArrange the following compounds / ions in increasing nucleophilicity (least to most nucleophilic) CH3NH2 CH3C=C: CH3COO 1 2 3 5 Multiple Choice 1 point 1, 2, 3 2, 1, 3 3, 1, 2 2, 3, 1 The other answers are not correct 0000arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning