
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
5th Edition
ISBN: 9781260170405
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24, Problem 24.41P
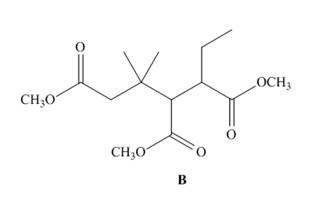
Even though B contains three ester groups, a single Dieckmann product results when B is treated with
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Check the box under each structure in the table that is an enantiomer of the molecule shown below. If none of them are, check the none of the above box under
the table.
Molecule 1
Molecule 2
Molecule 3
----|||
Molecule 4
Molecule 5
Molecule 6
none of the above
mm..
Use the vapor-liquid equilibrium data at 1.0 atm. for methanol-water (Table 2-8 ) for the following:
If the methanol vapor mole fraction is 0.600, what is the methanol liquid mole fraction?
Is there an azeotrope in the methanol-water system at a pressure of 1.0 atmospheres?
If water liquid mole fraction is 0.350, what is the water vapor mole fraction?
What are the K values of methanol and of water at a methanol mole fraction in the liquid of 0.200?
What is the relative volatility αM-W at a methanol mole fraction in the liquid of 0.200?
Check the box under each structure in the table that is an enantiomer of the molecule shown below. If none of them are, check the none of the above box under
the table.
||
|II*****
Molecule 1
|
Molecule 4
none of the above
Molecule 2
Molecule 3
Х
mm...
C
---|||
***
Molecule 5
Molecule 6
Chapter 24 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
Ch. 24 - Prob. 24.1PCh. 24 - Prob. 24.2PCh. 24 - Problem 24.3
What unsaturated carbonyl compound is...Ch. 24 - Prob. 24.4PCh. 24 - Prob. 24.5PCh. 24 - Prob. 24.6PCh. 24 - Problem 24.7
Draw the products formed in each...Ch. 24 - Problem 24.8
Draw the products formed in the...Ch. 24 - Prob. 24.9PCh. 24 - Prob. 24.10P
Ch. 24 - Prob. 24.11PCh. 24 - Prob. 24.12PCh. 24 - Prob. 24.13PCh. 24 - Prob. 24.14PCh. 24 - Prob. 24.15PCh. 24 - Problem 24.16
What ester is formed when each...Ch. 24 - Prob. 24.17PCh. 24 - Prob. 24.18PCh. 24 -
Draw the products of each reaction.
a. b.
Ch. 24 - Problem 24.20
Two steps in a synthesis of the...Ch. 24 - Prob. 24.21PCh. 24 - Problem 24.22
Which of the following compounds can...Ch. 24 - Prob. 24.23PCh. 24 - Problem 24.24
What starting materials are needed...Ch. 24 - Problem 24.25
Draw the products when each pair of...Ch. 24 - Prob. 24.26PCh. 24 - Problem 24.27
What starting materials are needed...Ch. 24 - Prob. 24.28PCh. 24 - 24.29 What steps are needed to convert A to B?
Ch. 24 - Prob. 24.30PCh. 24 - 24.31 Draw the product formed in each directed...Ch. 24 - Prob. 24.32PCh. 24 - 24.33 What starting materials are needed to...Ch. 24 - Prob. 24.34PCh. 24 - Prob. 24.35PCh. 24 - 24.36 Identify the structures of C and D in the...Ch. 24 - Prob. 24.37PCh. 24 - Prob. 24.38PCh. 24 - 24.39 Draw the product formed from a Claisen...Ch. 24 - Prob. 24.40PCh. 24 - 24.41 Even though B contains three ester groups, a...Ch. 24 - Prob. 24.42PCh. 24 - Prob. 24.43PCh. 24 - 24.44 Vetivone is isolated from vetiver, a...Ch. 24 - Draw the product of each Robinson annulation from...Ch. 24 - Prob. 24.46PCh. 24 - 24.47 Draw the organic products formed in each...Ch. 24 - 24.48 Fill in the lettered reagents needed for...Ch. 24 - Prob. 24.49PCh. 24 - Prob. 24.50PCh. 24 - Prob. 24.51PCh. 24 - 24.52 Draw a stepwise mechanism for the following...Ch. 24 - Prob. 24.53PCh. 24 - Prob. 24.54PCh. 24 - Prob. 24.55PCh. 24 - Prob. 24.56PCh. 24 - Prob. 24.57PCh. 24 - Prob. 24.58PCh. 24 - Prob. 24.59PCh. 24 - 24.60 Devise a synthesis of each compound from the...Ch. 24 - 24.61 Devise a synthesis of each compound from...Ch. 24 - 24.62 Devise a synthesis of each compound from ,...Ch. 24 - Prob. 24.63PCh. 24 - Prob. 24.64PCh. 24 - 24.65 Answer the following questions about...Ch. 24 - Prob. 24.66PCh. 24 - Prob. 24.67PCh. 24 - Prob. 24.68PCh. 24 - 24.69 Devise a stepwise mechanism for the...Ch. 24 - 24.70 Draw a stepwise mechanism for the following...Ch. 24 - Prob. 24.71PCh. 24 - Prob. 24.72PCh. 24 - Prob. 24.73P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- is SiBr4 Silicon (IV) tetra Bromine? is KClO2 potassium dihypochlorite ?arrow_forward"יוון HO" Br CI Check the box under each structure in the table that is an enantiomer of the molecule shown below. If none of them are, check the none of the above box under the table. Molecule 1 Molecule 2 Molecule 3 Br Br Br HO OH H CI OH ✓ Molecule 4 Molecule 5 Molecule 6 CI Br יייון H Br OH OH CI Br ☐ none of the above × Garrow_forwardUS2 Would this be Uranium (II) diSulfide?arrow_forward
- nomenclature for PU(SO4)3arrow_forwardLi2CrO4 is this Lithium (II) Chromatearrow_forwardCheck the box under each structure in the table that is an enantiomer of the molecule shown below. If none of them are, check the none of the above box under the table. NH ** Molecule 1 NH Molecule 4 none of the above Х Molecule 3 Molecule 2 H N wwwwww.. HN Molecule 5 Molecule 6 HN R mw... N H ☐arrow_forward
- Nomenclature P4S3 Would this be tetraphsophorus tri sulfide?arrow_forwardDon't used Ai solutionarrow_forwardBenzene-toluene equilibrium is often approximated as αBT = 2.34. Generate the y-x diagram for this relative volatility. Also, generate the equilibrium data using Raoult’s law, and compare your results to these.arrow_forward
- Given the most probable macrostate: s/k (K) Populations 300 4 200 8 100 16 0 32 Indicate how to demonstrate that the population of the levels is consistent with the Boltzmann distribution.arrow_forwardRank the following components in order of decreasing volatility: butane, n-pentane, iso-pentene (e.g., 3-methyl-1-butene), isoprene, pentanol? Briefly explain your answer.arrow_forwardViscosity of a liquid related to the activation energy.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License