
How much steam at
To Calculate: The amount of steam condensed to melt 1 gram of ice resulting in ice water to remains at
Answer to Problem 54A
Explanation of Solution
Given:
Mass of ice to be melt is 1 gram.
Temperature of steam is
Temperature of ice is
Formula used:
Heat required:
Where,
m is mass of substance.
c is the specific heat.
The formula to calculate required heat when there is a phase change:
m is the mass of substance.
L is latent heat.
Calculation:
Let the mass of steam to be condensed is
Heat lost when
(Where,
Then,
Heat lost when
Heat lost when Heat lost when
(where,
According to thermodynamics,
Conclusion:
The amount of steam at 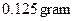
Chapter 23 Solutions
Conceptual Physics: The High School Physics Program
Additional Science Textbook Solutions
Microbiology: An Introduction
Organic Chemistry (8th Edition)
Anatomy & Physiology (6th Edition)
Human Biology: Concepts and Current Issues (8th Edition)
Chemistry & Chemical Reactivity
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
- In the circuit shown below & = 66.0 V, R5 = 4.00, R3 = 2.00, R₂ = 2.20 ₪, I5 = 11.41 A, I₁ = 10.17 A, and i̟ = 6.88 A. Find the current through R2 and R3, and the values of the resistors R₁ and R. (Due to the nature of this problem, do not use rounded intermediate values-including answers submitted in WebAssign-in your calculations.) 12 = 8.12 8.12 13 R₁₁ = RA = A Based on the known variables, which two junctions should you consider to find the current I3? A 6.9965 61.5123 Ω Which loop will give you an equation with just R4 as the unknown? Did you follow the sign convention for the potential difference across each element in the loop? R₁ www 11 R₂ www R4 www 14 8 15 www R5 www R3arrow_forwardA car traveling at 42 km/h hits a bridge abutment. A passenger in the car moves forward a distance of 53 cm (with respect to the road) while being brought to rest by an inflated air bag. What magnitude of force (assumed constant) acts on the passenger's upper torso, which has a mass of 43 kg? Number i Unitsarrow_forwardThree resistors R₁ = 88.1 Q, R2 = 19.9 £2, R3 = 70.00, and two batteries & ₁ = 40.0 V, and ε2 = 353 V are connected as shown in the diagram below. R₁ www E₁ E2 R₂ ww ww R3 (a) What current flows through R₁, R2, and R3? 11 = 0.454 Did you choose directions for each of the three currents? Given that you have three unknowns to solve for, how many equations, at the least, will you need? A 12 = 1.759 Did you choose directions for each of the three currents? Given that you have three unknowns to solve for, how many equations, at the least, will you need? A 13 2.213 = Did you choose directions for each of the three currents? Given that you have three unknowns to solve for, how many equations, at the least, will you need? A (b) What is the absolute value of the potential difference across R1, R2, and R3? |AVR1 = 40.0 How is the potential difference related to the current and the resistance? V |AVR2 = 35.0 How is the potential difference related to the current and the resistance? V |AVR3 =…arrow_forward
- In the attached image is the circuit for what the net resistance of the circuit connected to the battery? Each resistance in the circuit is equal to 14.00 kΩ. Thanks.arrow_forwardDetermine the equivalent capacitance for the group of capacitors in the drawing. Assume that all capacitors be the same where C = 24.0 µF. Thank you.arrow_forwardIn the figure below, what is the net resistance of the circuit connected to the battery? Assume that all resistances in the circuit is equal to 14.00 kΩ. Thank you.arrow_forward
- Due to the nature of this problem, do not use rounded intermediate values-including answers submitted in WebAssign-in your calculations. 3 4 Find the currents flowing in the circuit in the figure below. (Assume the resistances are R₁ =6, R₂ = 20, R₂ = 10 N, R₁ = 8, r₁ = 0.75 0, r2=0.50, 3 × A × A I, = 3.78 12 13 = 2.28 = 1.5 × A R₁ b a R₁₂ w C 1, 12 13 R₂ E3 12 V E₁ 18 V g Ez 3.0 V 12 Ea شرة R₁ e 24 V d = 0.25 0, and 4 = 0.5 0.)arrow_forwardIn the circuit shown below Ɛ = 66.0 V, R5 = 4.00 £2, R3 = 2.00 N, R₂ = 2.20 N, I5 = 11.41 A, I = 10.17 A, and d I₁ = 6.88 A. Find the current through R2 and R3, and the values of the resistors R₁ and R. (Due to the nature of this problem, do not use rounded intermediate values—including answers submitted in WebAssign-in your calculations.) 12 = 8.12 A RA = -1.24 Based on the known variables, which two junctions should you consider to find the current I3? A 9.59 Which loop will give you an equation with just R₁ as the unknown? Did you follow the sign convention for the potential difference across each element in the loop? 6.49 Which loop will give you an equation with just R as the unknown? Did you follow the sign convention for the potential difference across each element in the loop? N R₁ ww R₂ www R4 ww 14 15 www R5 www R3arrow_forwardCertain types of particle detectors can be used to reconstruct the tracks left by unstable, fast-moving sub-atomic particles. Assume that a track with a length of L=2.97 mm in the laboratory frame of reference has been observed. Further assume that you determined from other detector data that the particle moved at a speed of L=0.910 ⚫ c, also in the laboratory frame of reference. c denotes the speed of light in vacuum. What proper lifetime would you determine for this particle from the data given? T= 4.0 Sarrow_forward
- generated worksheetarrow_forwardWhile cruising down University Boulevard you are stopped by a cop who states that you ran a red traffic light. Because you don't want to pay the stiff fine, you are attempting a physics defense. You claim that due to the relativistic Doppler effect, the red color of the light λ=616 nm appeared green '=531 nm to you. The cop makes a quick calculation of his own and rejects your defense. How fast, in terms of your speed u divided by the speed of light in vacuum c, would you have to drive to justify your claim? Note that the speed u is taken to be a positive quantity. U 4.0 Carrow_forward220 V is supplied to 800 primary turns of an autotransformer. What will the outputvoltage be across 200 secondary turns? 2. A filament transformer has a turns ratio of 1:20. What current must be supplied to theprimary windings if 5 A is required by the filament? 3. The filament transformer in the previous question is supplied with 150 V to theprimary side. What is the secondary voltage? 4. 440 V is supplied to 1000 primary turns of an autotransformer. If the desired outputvoltage is 100 V how many secondary turns must be tapped?arrow_forward
 College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON
University Physics (14th Edition)PhysicsISBN:9780133969290Author:Hugh D. Young, Roger A. FreedmanPublisher:PEARSON Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press
Introduction To Quantum MechanicsPhysicsISBN:9781107189638Author:Griffiths, David J., Schroeter, Darrell F.Publisher:Cambridge University Press Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley
Lecture- Tutorials for Introductory AstronomyPhysicsISBN:9780321820464Author:Edward E. Prather, Tim P. Slater, Jeff P. Adams, Gina BrissendenPublisher:Addison-Wesley College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON
College Physics: A Strategic Approach (4th Editio...PhysicsISBN:9780134609034Author:Randall D. Knight (Professor Emeritus), Brian Jones, Stuart FieldPublisher:PEARSON





