
(a)
Interpretation:
Whether the given compound can undergo a Friedel–Crafts reaction, it is to be determined.
Concept introduction:
The Friedel–Crafts reaction is the electrophilic aromatic substitution reaction. In The Friedel–Crafts alkylation, the
Answer to Problem 23.51P
The given compound can undergo a Friedel–Crafts reaction as the aromatic ring is activated by two methyl groups.
Explanation of Solution
The given aromatic compound is:
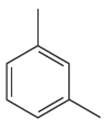
In this aromatic compound, the benzene ring has two methyl groups attached. The alkyl groups are electron donating inductively, thus increases the electron density around the ring and activates it. As the ring is electron rich and activated it can undergo Friedel–Crafts reaction.
It is determined that a given compound can undergo Friedel–Crafts reaction based on the activation of aromatic ring.
(b)
Interpretation:
Whether the given compound can undergo a Friedel–Crafts reaction, it is to be determined.
Concept introduction:
The Friedel–Crafts reaction is the electrophilic aromatic substitution reaction. In The Friedel–Crafts alkylation, the aromatic compound reacts with alkyl halide in presence of
Answer to Problem 23.51P
The given compound cannot undergo a Friedel–Crafts reaction as the aromatic ring is deactivated by two acetyl groups.
Explanation of Solution
The given aromatic compound is:
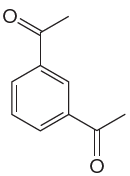
In this aromatic compound, the benzene ring has two acetyl (carbonyl) groups attached. The carbonyl groups have electron withdrawing resonance effect, thus decreases the electron density around the ring and deactivates it. As the ring is electron poor and deactivated it cannot undergo Friedel–Crafts reaction.
It is determined that a given compound cannot undergo Friedel–Crafts reaction based on the deactivation of aromatic ring.
(c)
Interpretation:
Whether the given compound can undergo a Friedel–Crafts reaction, it is to be determined.
Concept introduction:
The Friedel–Crafts reaction is the electrophilic aromatic substitution reaction. In The Friedel–Crafts alkylation, the aromatic compound reacts with alkyl halide in presence of
Answer to Problem 23.51P
The given compound cannot undergo a Friedel–Crafts reaction as the aromatic ring is deactivated by nitrile groups.
Explanation of Solution
The given aromatic compound is:
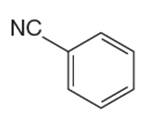
In this aromatic compound, the benzene ring has nitrile group attached. The nitrile group have electron withdrawing resonance effect, thus decreases the electron density around the ring and deactivates it. As the ring is electron poor and deactivated it cannot undergo Friedel–Crafts reaction.
It is determined that a given compound cannot undergo Friedel–Crafts reaction based on the deactivation of aromatic ring.
(d)
Interpretation:
Whether the given compound can undergo a Friedel–Crafts reaction, it is to be determined.
Concept introduction:
The Friedel–Crafts reaction is the electrophilic aromatic substitution reaction. In The Friedel–Crafts alkylation, the aromatic compound reacts with alkyl halide in presence of
Answer to Problem 23.51P
The given compound cannot undergo a Friedel–Crafts reaction as the aromatic ring is deactivated by
Explanation of Solution
The given aromatic compound is:
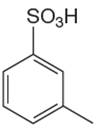
In this aromatic compound, the benzene ring has
It is determined that a given compound cannot undergo Friedel–Crafts reaction based on the deactivation of aromatic ring.
(e)
Interpretation:
Whether the given compound can undergo a Friedel–Crafts reaction, it is to be determined.
Concept introduction:
The Friedel–Crafts reaction is the electrophilic aromatic substitution reaction. In The Friedel–Crafts alkylation, the aromatic compound reacts with alkyl halide in presence of
Answer to Problem 23.51P
The given compound cannot undergo a Friedel–Crafts reaction as the aromatic ring is deactivated by
Explanation of Solution
The given aromatic compound is:
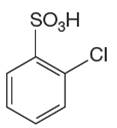
In this aromatic compound, the benzene ring has
It is determined that a given compound cannot undergo Friedel–Crafts reaction based on the deactivation of aromatic ring.
(f)
Interpretation:
Whether the given compound can undergo a Friedel–Crafts reaction, it is to be determined.
Concept introduction:
The Friedel–Crafts reaction is the electrophilic aromatic substitution reaction. In The Friedel–Crafts alkylation, the aromatic compound reacts with alkyl halide in presence of
Answer to Problem 23.51P
The given compound can undergo a Friedel–Crafts reaction as the aromatic ring is activated by one methyl and one methoxy groups.
Explanation of Solution
The given aromatic compound is:
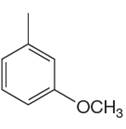
In this aromatic compound, the benzene ring has one methyl and one methoxy groups attached. The alkyl groups are electron donating inductively and methoxy group has electron donating resonance effect, thus increases the electron density around the ring and activates it. As the ring is electron rich and activated it can undergo Friedel–Crafts reaction.
It is determined that a given compound can undergo Friedel–Crafts reaction based on the activation of aromatic ring.
Want to see more full solutions like this?
Chapter 23 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- 36) Complete the following multi-step reactions showing applications of enolate ions arising from ketones, esters, malonic ester, and keto ester, etc. (30 pts) (1) A NaOH, H₂O+ heat A NaOEt EtO OEt (11) EOH, H+ H. B LDA, H₂O+ -78°C B (i) NaOMe, Et-Br (ii) H₂O+, heat EtOOC (III) COOEt B A (i) NaOEt LiAlH 4-bromo-2-butene H₂O+ (ii) H3O+, heat Write the mechanism for Aldol Condensation (I A or B), and Claisen Condensation (II A).arrow_forward31) Complete two sets of reactions involving (R)-4-methyl-pent-2-ol producing racemic mixture of tertiary alcohols (D) and ketone derivative (C). Illustrate the mechanism of B and C or D. (25 pts) O OH 0 K2Cr2O7 Ph-CH2-Br, Mg, H2SO4 THF, H3O* (A) (D) Racemic mixture TsCl, Py (B) KCN, DMSO Ph-CH2-Br, Mg, THF, H3O+ (C) Mechanism for reactions B and C:arrow_forwardManoharan Mariappan, Ph.D., Dept. of Natur. Sci., NFC, Tallahassee, FL 33) Synthesize the aromatic compound containing para-substituted carbonyl compound starting from benzene. Illustrate the mechanism for reaction A. 1) NU (25 pts) A FeCl B (i) HNO3, H2SO4 (II) Sn, HCl(aq) NH₂ NO₂-D NH₂ (i) MeCO2Me, heat C (ii) K2Cr2O7/H2SO4 D (ii) SOCl2 (iii) 2 Et-NH2 Mechanism for reaction for the nitration of alkyl benzene (B-i): Characterize the product compound arising from the reaction D by IR and IH NMR spectral data: IR data (cm): 'H NMR data: Draw the structure and assign the chemical shift with the spin-splitting.arrow_forward
- Write structural formulas for the major products by doing addition reactions 1. You must add H2 as Pt is catalyst it does not take part in reactions only speed up the process H₂ CH2=CH-CH3 Pt 2. Add HCI break it into H and Cl CH3 HCI 3. Add Br2 only CC14 is catalyst CH3-CH=CH2 B12 CCl4 4. Add water to this and draw major product, H2SO4 is catalyst you have add water H20 in both the reaction below H₂SO4 CH3-CH=CH2 CH3 H2SO4/H₂O CH3-C=CH2 reflux ?arrow_forwardPlan the synthesis of the following compound using the starting material provided and any other reagents needed as long as carbon based reagents have 3 carbons or less. Either the retrosynthesis or the forward synthesis (mechanisms are not required but will be graded if provided) will be accepted if all necessary reagents and intermediates are shown (solvents and temperature requirements are not needed unless specifically involved in the reaction, i.e. DMSO in the Swern oxidation or heat in the KMnO4 oxidation). H Harrow_forwardHint These are benzene substitution reactions. ALCI3 and UV light are catalyst no part in reactions and triangle A means heating. A. Add ethyl for Et in benzene ring alkylation reaction EtCl = CH3CH2CL 1) EtC1 / AlCl3 / A ? B: Add Br to benzene ring ( substitution) 2) Br₂ / uv light ? C Add (CH3)2 CHCH2 in benzene ring ( substitution) (CH3)2CHCH,C1 / AICI, ?arrow_forward
- Draw the mechanism to make the alcohol 2-hexanol. Draw the Mechanism to make the alcohol 1-hexanol.arrow_forwardDraw the mechanism for the formation of diol by starting with 1-pentanal in... basic conditions then acidic conditions then draw the mechanism for the formation of a carboxylic acid from your product.arrow_forwardIdentify each chiral carbon as either R or S. Identify the overall carbohydrates as L or Darrow_forward
- Ethers can be formed via acid-catalyzed acetal formation. Draw the mechanism for the molecule below and ethanol.arrow_forwardHOCH, H HO CH-OH OH H OH 11 CH₂OH F II OH H H 0 + H OHarrow_forwardDraw the mechanism for the formation of diol by starting with one pen and all in... basic conditions then acidic conditions then draw the mechanism for the formation of a carboxylic acid from your product.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





