
Concept explainers
(a)
Interpretation: The theoretical yield of
Concept introduction:
Number of moles of a substance,
From its given mass is,
Theoretical yield is the maximum product yield that can be expected based on the masses of the reactants and the reaction stoichiometry.
2-Iodobenzoic acid is a crystaline solid and it can be prepared from 2-aminobenzoic acid treated with
The reaction can be represnted as follows,
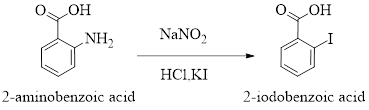
(a)
Answer to Problem 104IL
Theoretical yield of benzoic acid is
Explanation of Solution
From the given data,
Let’s calculate the number of moles of 2-amino benzoic acid:
From the given:
Let’s calculate the number of moles of
From the given:
Let’s calculate the number of moles of
From the equation , we can see that 1 mole of 2- amino benzoic acid gives 1 mole of 1-iodo benzoic acid .
For this conversion required 1 mole of
Theoretically produced
Molar mass of 2-iodobenzoic acid =
Therefore, theoretical yield of 2-iodobenzoic acid is
(b)
Interpretation: To Check the possibilty of isomers for 2-iodobenzoic acid
Concept introduction:
2-Iodobenzoic acid is a crystaline solid and it can be prepared from 2-aminobenzoic acid treated with
The reaction can be represnted as follows,
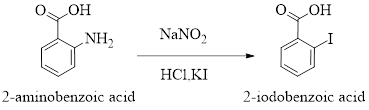
Two compounds that have the same molecular formula, but have different structural formulas are called isomers.
(b)
Answer to Problem 104IL
No other possible isomers for 2-Iodobenzoic acid.
Explanation of Solution
No other isomers of 2-iodo benzoic acid are possible in this reaction because the
(c)
Interpretation: The molar mass of the product formed in the given reaction has to be determined. And check whether the calculated molar mass is in reasonable agreement with the theoretical molar mass or not.
Concept introduction:
2-Iodobenzoic acid is a crystaline solid and it can be prepared from 2-aminobenzoic acid treated with
The reaction can be represnted as follows,
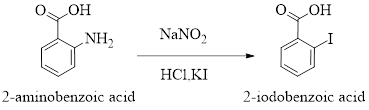
Concentration of solutions can be expressed in various terms; molarity is one such concentration expressing term.
Molarity (M) of a solution is the number of gram moles of a solute present in one liter of the solution.
A solution containing one gram mole or
Amount of substance (mol) can be determined by using the equation,
The molar mass of an element or compound is the mass in grams of 1 mole of that substance, and it is expressed in the unit of grams per mol (g/mol).
Theoretical yield is the maximum product yield that can be expected based on the masses of the reactants and the reaction stoichiometry.
(c)
Answer to Problem 104IL
Molar mass of 2-Iodobenzoic acid
There is only
Explanation of Solution
From the given,
Let’s calculate the number of mole of
Let’s calculate molar mass of 2-iodo benzoic acid.
Theoretical mass 2-Iodobenzoic acid is
Therefore, there is only
We can say that it is in reasonable agreement with theoretical molar mass.
Want to see more full solutions like this?
Chapter 23 Solutions
CHEMISTRY+CHEM...HYBRID ED.(LL)>CUSTOM<
- Draw the mechanism for the following reaction: CH3 CH3 Et-OH Et Edit the reaction by drawing all steps in the appropriate boxes and connecting them with reaction arrows. Add charges where needed. Electron-flow arrows should start on the electron(s) of an atom or a bond and should end on an atom, bond, or location where a new bond should be created. H± EXP. L CONT. י Α [1] осн CH3 а CH3 :Ö Et H 0 N о S 0 Br Et-ÖH | P LL Farrow_forward20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward20.00 mL of 0.025 M HCl is titrated with 0.035 M KOH. What volume of KOH is needed?arrow_forward
- 20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward20.00 mL of 0.025 M HCl is titrated with 0.035 M KOH. What volume of KOH is needed?arrow_forward20.00 mL of 0.150 M HCl is titrated with 37.75 mL of NaOH. What is the molarity of the NaOH?arrow_forward
- Calculate the pH of 0.450 M KOH.arrow_forwardWhich does NOT describe a mole? A. a unit used to count particles directly, B. Avogadro’s number of molecules of a compound, C. the number of atoms in exactly 12 g of pure C-12, D. the SI unit for the amount of a substancearrow_forward5 What would the complete ionic reaction be if aqueous solutions of potassium sulfate and barium acetate were mixed? ed of Select one: O a 2 K SO4 + Ba2 +2 C₂H3O21 K+SO4 + Ba2+ + 2 C2H3O21 K+SO42 + Ba2 +2 C2H3O2 BaSO4 +2 K+ + 2 C2H3O estion Ob. O c. Od. 2 K SO4 +Ba2 +2 C₂H₂O₂ BaSO4 + K+ + 2 C2H3O BaSO4 + K + 2 C2H301 →Ba² +SO42 +2 KC2H3O s pagearrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





