
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
5th Edition
ISBN: 9781260170405
Author: SMITH
Publisher: MCG
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22, Problem 22.44P
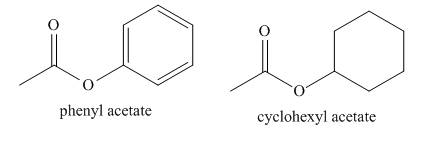
(a) Propose an explanation for the difference in the frequency of the carbonyl absorptions of phenyl acetate
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using the graphs could you help me explain the answers. I assumed that both graphs are proportional to the inverse of time, I think. Could you please help me.
Synthesis of Dibenzalacetone
[References]
Draw structures for the carbonyl electrophile and enolate nucleophile that react to give the enone below.
Question 1
1 pt
Question 2
1 pt
Question 3
1 pt
H
Question 4
1 pt
Question 5
1 pt
Question 6
1 pt
Question 7
1pt
Question 8
1 pt
Progress:
7/8 items
Que Feb 24 at
You do not have to consider stereochemistry.
. Draw the enolate ion in its carbanion form.
• Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner.
⚫ Separate multiple reactants using the + sign from the drop-down menu.
?
4
Shown below is the mechanism presented for the formation of biasplatin in reference 1 from the Background and Experiment document. The amounts used of each reactant are shown. Either draw or describe a better alternative to this mechanism. (Note that the first step represents two steps combined and the proton loss is not even shown; fixing these is not the desired improvement.) (Hints: The first step is correct, the second step is not; and the amount of the anhydride is in large excess to serve a purpose.)
Chapter 22 Solutions
Package: Loose Leaf for Organic Chemistry with Biological Topics with Connect Access Card
Ch. 22 - Prob. 22.1PCh. 22 - Draw the three possible resonance structures for...Ch. 22 - Prob. 22.3PCh. 22 - Give an IUPAC or common name for each compound. a....Ch. 22 - Problem 22.5 Draw the structure corresponding to...Ch. 22 - Problem 22.6 Explain why the boiling point of is...Ch. 22 - Problem 22.7 How would the compounds in each pair...Ch. 22 - Problem 22.8 Deduce the structures of compounds ...Ch. 22 - Problem 22.9 Without reading ahead in Chapter 22,...Ch. 22 - Rank the compounds in each group in order of...
Ch. 22 - Prob. 22.11PCh. 22 - Prob. 22.12PCh. 22 - Prob. 22.13PCh. 22 - Prob. 22.14PCh. 22 - Problem 22.15 Draw the products of each...Ch. 22 - Problem 22.16 Draw the products of each reaction.
...Ch. 22 - Prob. 22.17PCh. 22 - Problem 22.18 Draw a stepwise mechanism for the...Ch. 22 - Prob. 22.19PCh. 22 - Problem 22.20 Fenofibrate is a...Ch. 22 - Problem 22.21 What product is formed when the...Ch. 22 - How would you synthesize olestra from sucrose?
Ch. 22 - Problem 22.23 What is the composition of the soap...Ch. 22 - Problem 22.24 Draw a stepwise mechanism for the...Ch. 22 - Prob. 22.25PCh. 22 - Problem 22.26 Some penicillins cannot be...Ch. 22 - Prob. 22.27PCh. 22 - Prob. 22.28PCh. 22 - Prob. 22.29PCh. 22 - Problem 22.30 Glucosamine is a dietry supplement...Ch. 22 - Draw the products of each reaction. a. c. b.Ch. 22 - Draw a tautomer of each compound.
a. b. c.
Ch. 22 - Draw the product of each reaction. a. b.Ch. 22 - Draw the product of each reaction. a. b.Ch. 22 - Prob. 22.35PCh. 22 - Problem 22.36 Outline two different ways that can...Ch. 22 - 22.37 Rank the following compounds in order of...Ch. 22 - Prob. 22.38PCh. 22 - Prob. 22.39PCh. 22 - 22.40 Give the IUPAC or common name for each...Ch. 22 - 22.41 Give the structure corresponding to each...Ch. 22 - Prob. 22.42PCh. 22 - 22.43 Explain why is a stronger acid and a weaker...Ch. 22 - (a) Propose an explanation for the difference in...Ch. 22 - Draw the product formed when phenylacetic acid is...Ch. 22 - Draw the product formed when phenylacetonitrile ...Ch. 22 - 22.47 Draw the organic products formed in each...Ch. 22 - Prob. 22.48PCh. 22 - Prob. 22.49PCh. 22 - Prob. 22.50PCh. 22 - Prob. 22.51PCh. 22 - Prob. 22.52PCh. 22 - Prob. 22.53PCh. 22 - 22.54 Draw a stepwise mechanism f or the following...Ch. 22 - 22.55 When acetic acid () is treated with a trace...Ch. 22 - Prob. 22.56PCh. 22 - Prob. 22.57PCh. 22 - Draw a stepwise mechanism for the following...Ch. 22 - Prob. 22.59PCh. 22 - Prob. 22.60PCh. 22 - Prob. 22.61PCh. 22 - Draw a stepwise mechanism for the following...Ch. 22 - 22.63 Acid-catalyzed hydrolysis of forms compound...Ch. 22 - 22.64 What carboxylic acid and alcohol are needed...Ch. 22 - Problem 22.65 Devise a synthesis of each compound...Ch. 22 - Prob. 22.66PCh. 22 - Prob. 22.67PCh. 22 - Prob. 22.68PCh. 22 - Prob. 22.69PCh. 22 - 22.70 What polyester or poly amide can be prepared...Ch. 22 - 22.71 What two monomers are needed to prepare each...Ch. 22 - Prob. 22.72PCh. 22 - 22.73 How can IR spectroscopy be used to...Ch. 22 - 22.74 Rank the following compounds in order of...Ch. 22 - 22.75 Identify the structures of each compound...Ch. 22 - 22.76 Identify the structures of A and B, isomers...Ch. 22 - Prob. 22.77PCh. 22 - 22.78 Identify the structure of compound C...Ch. 22 - 22.79 Identify the structures of D and E, isomers...Ch. 22 - 22.80 With reference to amides A and B, the...Ch. 22 - Prob. 22.81PCh. 22 - Prob. 22.82PCh. 22 - Prob. 22.83PCh. 22 - Draw a stepwise mechanism for the following...Ch. 22 - Draw a stepwise mechanism for the following...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Hi I need help on the question provided in the image.arrow_forwardDraw a reasonable mechanism for the following reaction:arrow_forwardDraw the mechanism for the following reaction: CH3 CH3 Et-OH Et Edit the reaction by drawing all steps in the appropriate boxes and connecting them with reaction arrows. Add charges where needed. Electron-flow arrows should start on the electron(s) of an atom or a bond and should end on an atom, bond, or location where a new bond should be created. H± EXP. L CONT. י Α [1] осн CH3 а CH3 :Ö Et H 0 N о S 0 Br Et-ÖH | P LL Farrow_forward
- 20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward20.00 mL of 0.025 M HCl is titrated with 0.035 M KOH. What volume of KOH is needed?arrow_forward20.00 mL of 0.150 M NaOH is titrated with 37.75 mL of HCl. What is the molarity of the HCl?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY