
Chemistry Atoms First2e
2nd Edition
ISBN: 9781947172647
Author: OpenStax
Publisher: OpenStax College
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 21, Problem 7E
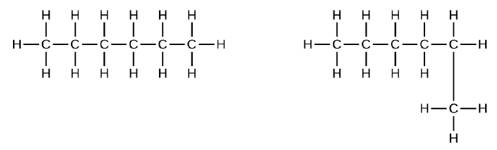
Explain why these two molecules are not isomers:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Can I please get the graph 1: Concentration vs. Density?
Order the following series of compounds from highest to lowest reactivity to electrophilic aromatic substitution, explaining your answer: 2-nitrophenol, p-Toluidine, N-(4-methylphenyl)acetamide, 4-methylbenzonitrile, 4-(trifluoromethyl)benzonitrile.
Ordene la siguiente serie de compuestos de mayor a menor reactividad a la sustitución aromática electrofílica, explicando su respuesta: ácido bencenosulfónico, fluorobenceno, etilbenceno, clorobenceno, terc-butilbenceno, acetofenona.
Chapter 21 Solutions
Chemistry Atoms First2e
Ch. 21 - Write the chemical formula and Lewis structure of...Ch. 21 - What is the difference between the hybridization...Ch. 21 - On a microscopic level, how does the reaction of...Ch. 21 - On a microscopic level, how does the reaction of...Ch. 21 - Explain why unbranched alkenes can form geometric...Ch. 21 - Explain why these two molecules are not isomers:Ch. 21 - Explain why these two molecules are not isomers:Ch. 21 - How does the carbon-atom hybridization change when...Ch. 21 - Write the Lewis structure and molecular formula...Ch. 21 - Write the chemical formula, condensed formula, and...
Ch. 21 - Give the complete IUPAC name for each of the...Ch. 21 - Give the complete IUPAC name for each of the...Ch. 21 - Butane is used as a fuel in disposable lighters....Ch. 21 - Write Lewis structures and name the five...Ch. 21 - Write Lewis structures for the Cis -trans isomers...Ch. 21 - Write structures for the three isomers of the...Ch. 21 - Isooctane is the common name of the isomer of...Ch. 21 - Write Lewis structures and IUPAC names for the...Ch. 21 - Write Lewis structures and IUPAC names for all...Ch. 21 - Name and write the structures of all isomers of...Ch. 21 - Write the structures for all the isomers of the...Ch. 21 - Write Lewis structures and describe the molecular...Ch. 21 - Benzene is one of the compounds used as an octane...Ch. 21 - Teflon is prepared by the polymerization of...Ch. 21 - Write two complete, balanced equations for each of...Ch. 21 - Write two complete, balanced equations for each of...Ch. 21 - What mass of 2-bromopropane could be prepared from...Ch. 21 - Acetylene is a very weak acid; however, it will...Ch. 21 - Ethylene can be produced by the pyrolysis of...Ch. 21 - Why do the compounds hexane, hexanol, and hexane...Ch. 21 - Write condensed formulas and provide IUPAC names...Ch. 21 - Give the complete IUPAC name for each of the...Ch. 21 - Give the complete IUPAC name and the common name...Ch. 21 - Write the condensed structures of both isomers...Ch. 21 - Write the condensed structures of all isomers with...Ch. 21 - Draw the condensed formulas for each of the...Ch. 21 - MTBE, Methyl tert -butyl ether, CH3OC(CH3)3, is...Ch. 21 - Write two complete balanced equations for each of...Ch. 21 - Write two complete balanced equations for each of...Ch. 21 - Order the following molecules from least to most...Ch. 21 - Predict the products of oxidizing the molecules...Ch. 21 - Predict the products of reducing the following...Ch. 21 - Explain why it is not possible to possible a...Ch. 21 - How does hybridization of the substituted carbon...Ch. 21 - Fatty acids are carboxylic acids that have long...Ch. 21 - Write a condensed structural formula, such as...Ch. 21 - Write a condensed structural formula, such as...Ch. 21 - The foul odor of rancid butter is caused by...Ch. 21 - Write the two-resonance structures for the acetate...Ch. 21 - Write two complete, balanced equations for each of...Ch. 21 - Write two complete balanced equations for each of...Ch. 21 - Yields in organic reactions are sometimes low....Ch. 21 - Alcohols A, B and C all have the composition C4H...Ch. 21 - Write the Lewis structures of both isomers with...Ch. 21 - What is the molecular structure about the nitrogen...Ch. 21 - Write the two resonance structures for the...Ch. 21 - Draw Lewis structures for pyridine and its...Ch. 21 - Write the Lewis structures of all isomers with the...Ch. 21 - Write two complete balanced equations for the...Ch. 21 - Write two complete, balanced equations for each of...Ch. 21 - Identify any carbon atoms that change...Ch. 21 - Identify any carbon atoms that change...Ch. 21 - Identify any carbon atoms that change...
Additional Science Textbook Solutions
Find more solutions based on key concepts
What is the difference between cellular respiration and external respiration?
Human Physiology: An Integrated Approach (8th Edition)
1. Why is the quantum-mechanical model of the atom important for understanding chemistry?
Chemistry: Structure and Properties (2nd Edition)
Match the following examples of mutagens. Column A Column B ___a. A mutagen that is incorporated into DNA in pl...
Microbiology: An Introduction
1. If an object is not moving, does that mean that there are no forces acting on it? Explain.
College Physics: A Strategic Approach (3rd Edition)
MAKE CONNECTIONS Review the description of meiosis (see Figure 10.8) and Mendels laws of segregation and indepe...
Campbell Biology in Focus (2nd Edition)
Why is an endospore called a resting structure? Of what advantage is an endospore to a bacterial cell?
Microbiology: An Introduction
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Can I please get all final concentrations please!arrow_forwardState the detailed mechanism of the reaction of benzene with isopropanol in sulfuric acid.arrow_forwardDo not apply the calculations, based on the approximation of the stationary state, to make them perform correctly. Basta discard the 3 responses that you encounter that are obviously erroneous if you apply the formula to determine the speed of a reaction. For the decomposition reaction of N2O5(g): 2 N2O5(g) · 4 NO2(g) + O2(g), the following mechanism has been proposed: N2O5 -> NO2 + NO3_(K1) NO2 + NO3 →> N2O5 (k-1) → NO2 + NO3 → NO2 + O2 + NO (K2) NO + N2O5 → NO2 + NO2 + NO2 (K3) Give the expression for the acceptable rate. (A). d[N₂O] dt = -1 2k,k₂[N205] k₁+k₂ d[N₂O5] (B). dt =-k₁[N₂O₂] + k₁[NO2][NO3] - k₂[NO2]³ (C). d[N₂O] dt =-k₁[N₂O] + k₁[N205] - K3 [NO] [N205] (D). d[N2O5] =-k₁[NO] - K3[NO] [N₂05] dtarrow_forward
- A 0.10 M solution of acetic acid (CH3COOH, Ka = 1.8 x 10^-5) is titrated with a 0.0250 M solution of magnesium hydroxide (Mg(OH)2). If 10.0 mL of the acid solution is titrated with 20.0 mL of the base solution, what is the pH of the resulting solution?arrow_forwardFor the decomposition reaction of N2O5(g): 2 N2O5(g) → 4 NO2(g) + O2(g), the following mechanism has been proposed: N2O5 NO2 + NO3 (K1) | NO2 + NO3 → N2O5 (k-1) | NO2 + NO3 NO2 + O2 + NO (k2) | NO + N2O51 NO2 + NO2 + NO2 (K3) → Give the expression for the acceptable rate. → → (A). d[N205] dt == 2k,k₂[N₂O₂] k₁+k₁₂ (B). d[N2O5] =-k₁[N₂O] + k₁[NO₂] [NO3] - k₂[NO₂]³ dt (C). d[N2O5] =-k₁[N₂O] + k [NO] - k₂[NO] [NO] d[N2O5] (D). = dt = -k₁[N2O5] - k¸[NO][N₂05] dt Do not apply the calculations, based on the approximation of the stationary state, to make them perform correctly. Basta discard the 3 responses that you encounter that are obviously erroneous if you apply the formula to determine the speed of a reaction.arrow_forwardFor the decomposition reaction of N2O5(g): 2 N2O5(g) → 4 NO2(g) + O2(g), the following mechanism has been proposed: N2O5 NO2 + NO3 (K1) | NO2 + NO3 → N2O5 (k-1) | NO2 + NO3 NO2 + O2 + NO (k2) | NO + N2O51 NO2 + NO2 + NO2 (K3) → Give the expression for the acceptable rate. → → (A). d[N205] dt == 2k,k₂[N₂O₂] k₁+k₁₂ (B). d[N2O5] =-k₁[N₂O] + k₁[NO₂] [NO3] - k₂[NO₂]³ dt (C). d[N2O5] =-k₁[N₂O] + k [NO] - k₂[NO] [NO] d[N2O5] (D). = dt = -k₁[N2O5] - k¸[NO][N₂05] dt Do not apply the calculations, based on the approximation of the stationary state, to make them perform correctly. Basta discard the 3 responses that you encounter that are obviously erroneous if you apply the formula to determine the speed of a reaction.arrow_forward
- R lactam or lactone considering as weak acid or weak base and whyarrow_forward81. a. Propose a mechanism for the following reaction: OH CH2=CHCHC=N b. What is the product of the following reaction? HO H₂O N=CCH2CH2CH OH HO CH3CCH=CH2 H₂O C=N 82. Unlike a phosphonium ylide that reacts with an aldehyde or a ketone to form an alkene a sulfonium uliaarrow_forwardFor each reaction below, decide if the first stable organic product that forms in solution will create a new CC bond, and check the appropriate box. Next, for each reaction to which you answered "Yes" to in the table, draw this product in the drawing area below. Note for advanced students: for this problem, don't worry if you think this product will continue to react under the current conditions - just focus on the first stable product you expect to form in solution. ? NH2 MgBr Will the first product that forms in this reaction create a new CC bond? ○ Yes ○ No MgBr ? Will the first product that forms in this reaction create a new CC bond? O Yes O No Click and drag to start drawing a structure. :☐ G x c olo Ar HEarrow_forward
- Predicting As the lead product manager at OrganometALEKS Industries, you are trying to decide if the following reaction will make a molecule with a new C - C bond as its major product: H₂N O H 1. ? 2. H3O+ If this reaction will work, draw the major organic product or products you would expect in the drawing area below. If there's more than one major product, you can draw them in any arrangement you like. Be sure you use wedge and dash bonds if necessary, for example to distinguish between major products with different stereochemistry. 0 If the major products of this reaction won't have a new CC bond, just check the box under the drawing area and leave it blank. فا Explanation Check Click and drag to start drawing a structure.arrow_forwardHighlight the chirality (or stereogenic) center(s) in the given compound. A compound may have one or more stereogenic centers. OH OH OH OH OH OHarrow_forwardUsing wedge-and-dash bonds, modify the bonds on the chiral carbon in the molecule below so the molecule has R stereochemical configuration. NH H Br X टेarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY