
Concept explainers
Rank the following compounds in order of increasing reactivity towards nucleophilic attack.
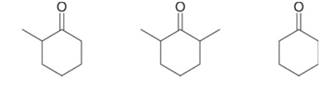
Interpretation: The given compounds are to be arranged in order of increasing reactivity towards nucleophilic attack.
Concept introduction: Both aldehydes and ketones undergo nucleophilic addition reaction. The relative reactivity of carbonyl group is inversely proportional to the number of
Answer to Problem 21.1P
The arrangement of given compounds in the increasing reactivity towards nucleophilic attack is,
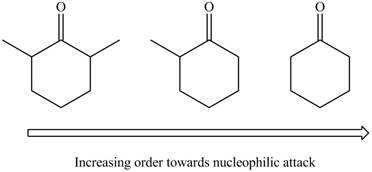
Explanation of Solution
Both aldehydes and ketones undergo nucleophilic addition reaction. In this reaction, the nucleophilic attack takes place at carbonyl carbon. The relative reactivity of carbonyl group is inversely proportional to the number of
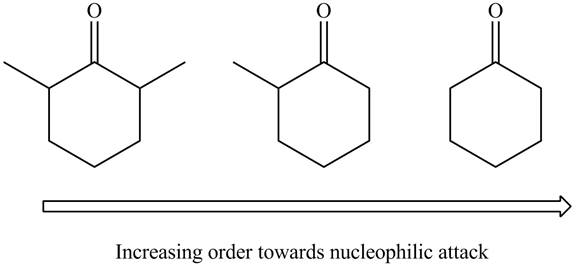
Figure 1
The arrangement of given compounds in the increasing reactivity towards nucleophilic attack is shown in Figure 1.
Want to see more full solutions like this?
Chapter 21 Solutions
Organic Chemistry
Additional Science Textbook Solutions
College Physics: A Strategic Approach (3rd Edition)
Human Biology: Concepts and Current Issues (8th Edition)
Loose Leaf For Integrated Principles Of Zoology
Applications and Investigations in Earth Science (9th Edition)
Laboratory Experiments in Microbiology (12th Edition) (What's New in Microbiology)
- Identify the compound with the longest carbon - nitrogen bond. O CH3CH2CH=NH O CH3CH2NH2 CH3CH2C=N CH3CH=NCH 3 The length of all the carbon-nitrogen bonds are the samearrow_forwardIdentify any polar covalent bonds in epichlorohydrin with S+ and 8- symbols in the appropriate locations. Choose the correct answer below. Η H's+ 6Η Η Η Η Η Ηδ Η Ο Ο HH +Η Η +Η Η Η -8+ CIarrow_forwardH H:O::::H H H HH H::O:D:D:H HH HH H:O:D:D:H .. HH H:O:D:D:H H H Select the correct Lewis dot structure for the following compound: CH3CH2OHarrow_forward
- Rank the following compounds in order of decreasing boiling point. ннннн -С-С-Н . н-с- ННННН H ΗΤΗ НННН TTTĪ н-с-с-с-с-о-н НННН НН C' Н н-с-с-с-с-н НН || Ш НННН H-C-C-C-C-N-H ННННН IVarrow_forwardRank the following compounds in order of decreasing dipole moment. |>||>||| ||>|||>| |>|||>|| |||>||>| O ||>>||| H F H F H c=c || H c=c F F IIIarrow_forwardchoose the description that best describes the geometry for the following charged species ch3-arrow_forward
- Why isn't the ketone in this compound converted to an acetal or hemiacetal by the alcohol and acid?arrow_forwardWhat is the approximate bond angle around the nitrogen atom? HNH H Harrow_forwardOH 1. NaOCH2CH3 Q 2. CH3CH2Br (1 equiv) H3O+ Select to Draw 1. NaOCH2 CH3 2. CH3Br (1 equiv) heat Select to Edit Select to Drawarrow_forward
- Complete and balance the following half-reaction in acidic solution. Be sure to include the proper phases for all species within the reaction. S₂O₃²⁻(aq) → S₄O₆²⁻(aq)arrow_forwardQ Select to Edit NH3 (CH3)2CHCI (1 equiv) AICI 3 Select to Draw cat. H2SO4 SO3 (1 equiv) HO SOCl2 pyridine Select to Edit >arrow_forwardComplete and balance the following half-reaction in basic solution. Be sure to include the proper phases for all species within the reaction. Zn(s) → Zn(OH)₄²⁻(aq)arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

