
(a)
Interpretation:
The approximate
| Set I | Set II | Set III | |||
| Time, s | [A], M | Time, s | [A], M | Time, s | [A], M |
| 0 | 1.00 | 0 | 1.00 | 0 | 1.00 |
| 25 | 0.78 | 25 | 0.75 | 25 | 0.80 |
| 50 | 0.61 | 50 | 0.50 | 50 | 0.67 |
| 75 | 0.47 | 75 | 0.25 | 75 | 0.57 |
| 100 | 0.37 | 100 | 0.00 | 100 | 0.50 |
| 150 | 0.22 | 150 | 0.40 | ||
| 200 | 0.14 | 200 | 0.33 | ||
| 250 | 0.08 | 250 | 0.29 |
Concept introduction:
- Zero-order reaction- for this, rate of reaction is independent of the concentration.
And the graph of zero order reaction is-

(b)
Interpretation:
The approximate rate of reaction at t= 75 sec for first order reaction is to be determined.
| Set I | Set II | Set III | |||
| Time, s | [A], M | Time, s | [A], M | Time, s | [A], M |
| 0 | 1.00 | 0 | 1.00 | 0 | 1.00 |
| 25 | 0.78 | 25 | 0.75 | 25 | 0.80 |
| 50 | 0.61 | 50 | 0.50 | 50 | 0.67 |
| 75 | 0.47 | 75 | 0.25 | 75 | 0.57 |
| 100 | 0.37 | 100 | 0.00 | 100 | 0.50 |
| 150 | 0.22 | 150 | 0.40 | ||
| 200 | 0.14 | 200 | 0.33 | ||
| 250 | 0.08 | 250 | 0.29 |
Concept introduction:
- The first order reaction is the one in which the rate is directly proportional to the concentration of one of the reactant.
And half-life is
Integral rate law for first order reaction is −
and
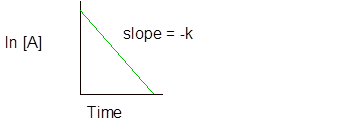
(c)
Interpretation:
The approximate rate of reaction at t= 75 sec for second order reaction is to be determined.
| Set I | Set II | Set III | |||
| Time, s | [A], M | Time, s | [A], M | Time, s | [A], M |
| 0 | 1.00 | 0 | 1.00 | 0 | 1.00 |
| 25 | 0.78 | 25 | 0.75 | 25 | 0.80 |
| 50 | 0.61 | 50 | 0.50 | 50 | 0.67 |
| 75 | 0.47 | 75 | 0.25 | 75 | 0.57 |
| 100 | 0.37 | 100 | 0.00 | 100 | 0.50 |
| 150 | 0.22 | 150 | 0.40 | ||
| 200 | 0.14 | 200 | 0.33 | ||
| 250 | 0.08 | 250 | 0.29 |
Concept introduction:
- The second order reaction is the one in which the rate is directly proportional to the square of the concentration of one of the reactant.
Integral rate law for second order reaction is −
And the graph of second order reaction is-
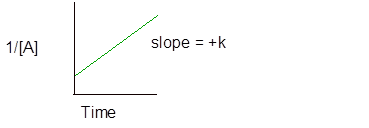
Want to see the full answer?
Check out a sample textbook solution
Chapter 20 Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
- Heating an alcohol in the presence of sulfuric or phosphoric acid will cause a dehydration to occur: the removal of the elements of water from a molecule, forming an alkene. The reaction usually follows an E1 mechanism. The SN1 pathway is suppressed by using a strong acid whose conjugate base is a poor nucleophile. Further, heating the reaction mixture causes a greater increase in the rate of E1 compared to the rate of Sy1. 3rd attempt h Draw curved arrow(s) to show how the alcohol reacts with phosphoric acid. TH © 1 0 0 +1% # 2nd attempt Feedback H Ju See Periodic Table See Hint H Jud See Periodic Table See Hintarrow_forwardPart 2 (0.5 point) 0- Draw the major organic product with the correct geometry. 10 1: 70000 х く 1st attempt Part 1 (0.5 point) Feedback Please draw all four bonds at chiral centers. P See Periodic Table See Hintarrow_forwardHeating an alcohol in the presence of sulfuric or phosphoric acid will cause a dehydration to occur: the removal of the elements of water from a molecule, forming an alkene. The reaction usually follows an E1 mechanism. The SN1 pathway is suppressed by using a strong acid whose conjugate base is a poor nucleophile. Further, heating the reaction mixture causes a greater increase in the rate of E1 compared to the rate of S№1. 2nd attempt 0 See Periodic Table See Hint Draw the organic intermediate from the first step (no byproducts) and draw curved arrow(s) to show how it reacts. TH +11: 1st attempt Feedback H H H C F F See Periodic Table See Hintarrow_forward
- This molecule undergoes an E1 mechanism when stirred in methanol. 3rd attempt CH₂OH CH₂OH 6148 O See Periodic Table. See Hint Draw 3 chemical species including formal charges and lone pairs of electrons. Add the missing curved arrow notation. H N O O SA 3 Br Iarrow_forwardComplete the mechanism for the E1 reaction below by following the directions written above each of the five boxes. Be sure to include lone pair electrons and nonzero formal charges. 1st attempt Y 0 + Provide the missing curved arrow notation. 01: See Periodic Table See Hint H C Br Iarrow_forwardPlease help answer number 2. Thanks in advance.arrow_forward
- How do I explain this? Thank you!arrow_forwardWhen an unknown amine reacts with an unknown acid chloride, an amide with a molecular mass of 163 g/mol (M* = 163 m/z) is formed. In the infrared spectrum, important absorptions appear at 1661, 750 and 690 cm. The 13C NMR and DEPT spectra are provided. Draw the structure of the product as the resonance contributor lacking any formal charges. 13C NMR DEPT 90 200 160 120 80 40 0 200 160 120 80 40 0 DEPT 135 T 200 160 120 80 40 0 Draw the unknown amide. Select Dow Templates More Fragearrow_forwardIdentify the unknown compound from its IR and proton NMR spectra. C4H6O: 'H NMR: 82.43 (1H, t, J = 2 Hz); 8 3.41 (3H, s); 8 4.10 (2H, d, J = 2 Hz) IR: 2125, 3300 cm¹ The C4H6O compound liberates a gas when treated with C2H5 MgBr. Draw the unknown compound. Select Draw с H Templates Morearrow_forward
- Please help with number 6 I got a negative number could that be right?arrow_forward1,4-Dimethyl-1,3-cyclohexadiene can undergo 1,2- or 1,4-addition with hydrogen halides. (a) 1,2-Addition i. Draw the carbocation intermediate(s) formed during the 1,2-addition of hydrobromic acid to 1,4-dimethyl-1,3-cyclohexadiene. ii. What is the major 1,2-addition product formed during the reaction in (i)? (b) 1,4-Addition i. Draw the carbocation intermediate(s) formed during the 1,4-addition of hydrobromic acid to 1,4-dimethyl-1,3-cyclohexadiene. ii. What is the major 1,4-addition product formed from the reaction in (i)? (c) What is the kinetic product from the reaction of one mole of hydrobromic acid with 1,4-dimethyl-1,3-cyclohexadiene? Explain your reasoning. (d) What is the thermodynamic product from the reaction of one mole of hydrobro-mic acid with 1,4-dimethyl-1,3-cyclohexadiene? Explain your reasoning. (e) What major product will result when 1,4-dimethyl-1,3-cyclohexadiene is treated with one mole of hydrobromic acid at - 78 deg * C ? Explain your reasoning.arrow_forwardGive the product of the bimolecular elimination from each of the isomeric halogenated compounds. Reaction A Reaction B. КОВ CH₂ HotBu +B+ ко HOIBU +Br+ Templates More QQQ Select Cv Templates More Cras QQQ One of these compounds undergoes elimination 50x faster than the other. Which one and why? Reaction A because the conformation needed for elimination places the phenyl groups and to each other Reaction A because the conformation needed for elimination places the phenyl groups gauche to each other. ◇ Reaction B because the conformation needed for elimination places the phenyl groups gach to each other. Reaction B because the conformation needed for elimination places the phenyl groups anti to each other.arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,
Physical ChemistryChemistryISBN:9781133958437Author:Ball, David W. (david Warren), BAER, TomasPublisher:Wadsworth Cengage Learning,





