
Concept explainers
(a)
Interpretation:
Lewis structure of periodate ion
Concept Introduction:
Lewis structure represents covalent bonds and describes valence electrons configuration of atoms. The covalent bonds are depicted by lines and unshared electron pairs by pairs of dots. The sequence to write Lewis structure of some molecule is given as follows:
- The central atom is identified and various other atoms are arranged around it. This central atom so chosen is often the least electronegative.
- Total valence electrons are estimated for each atom.
- A single bond is first placed between each atom pair.
- The electrons left can be allocated as unshared electron pairs or as multiple bonds around
symbol of element to satisfy the octet (or duplet) for each atom. - Add charge on overall structure in case of polyatomic cation or anion.
(a)
Explanation of Solution
The molecule
The symbol for oxygen is
The symbol for iodine is
One negative charge on molecule is added up as one valence electron in total count.
Thus total valence electrons are sum of the valence electrons for each atom in
The skeleton structure in
To complete the valence electrons of iodine, it forms double bond with three oxygen atoms.
Hence, the Lewis structure for
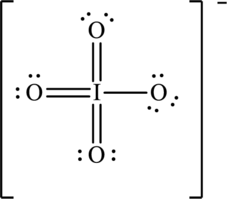
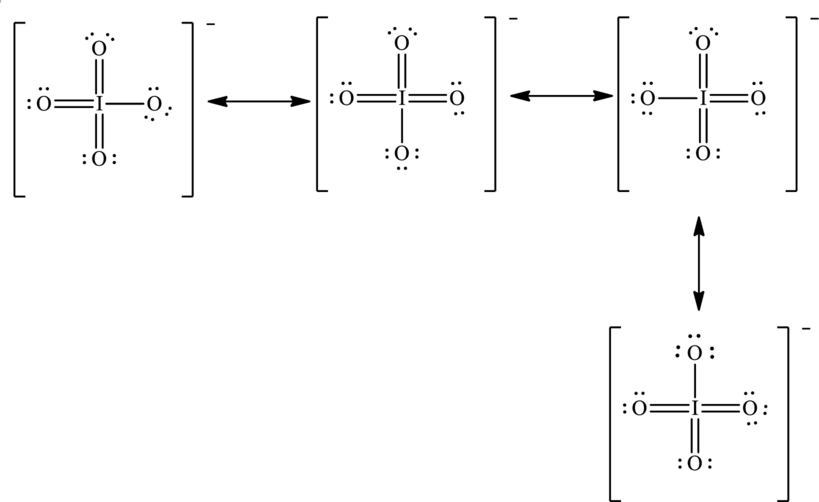
To complete the octet of iodine atom the negative charge is delocalized on each oxygen atom and since there are four oxygen atoms, therefore, four resonance structures will be formed and possible resonance structures are as follows:
(b)
Interpretation:
Lewis structure of hydrogen phosphate ion
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
Hydrogen phosphate ion
The symbol for oxygen is
The symbol for hydrogen is
The symbol for phosphorus is
Two negative charges on molecule are added up as one valence electron in total count.
Thus total valence electrons are sum of the valence electrons for each atom in
The skeleton structure in
To complete the valence electrons of phosphorous, it forms double bond with one oxygen atom.
Hence, 20 electrons are allocated as 10 lone pairs on remaining oxygen atoms to complete their octet. The Lewis structure is as follows:
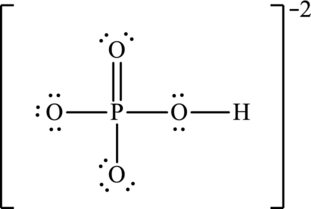
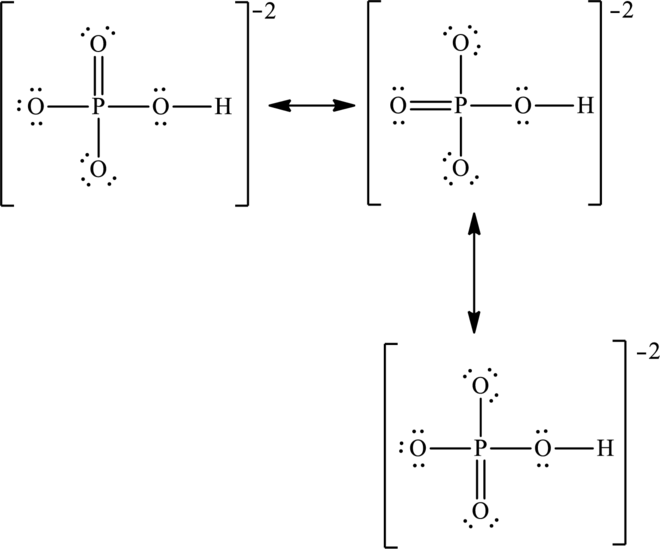
The negative charge is delocalized on three oxygen atoms and therefore three resonance structures will be formed and possible resonance structures are as follows:
(c)
Interpretation:
Lewis structure of chloric acid
Concept Introduction:
Refer to part (a).
(c)
Explanation of Solution
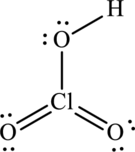
Chloric acid
The symbol for oxygen is
The symbol for hydrogen is
The symbol for chlorine is
Thus total valence electrons are sum of the valence electrons for each atom in
The skeleton structure in
To complete the valence electrons of chlorine, it forms double bond with two oxygen atoms.
Hence, 14 electrons are allocated as 6 lone pairs on remaining oxygen atoms and 1 lone pair on chlorine to complete their respective octet. The Lewis structure is as follows:
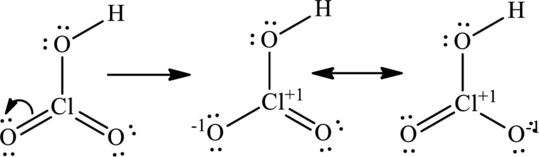
The lone pair on oxygen atom that is attached to chlorine participates in resonance with that and produces
(d)
Interpretation:
Lewis structure of arsenate ion
Concept Introduction:
Refer to part (a).
(d)
Explanation of Solution
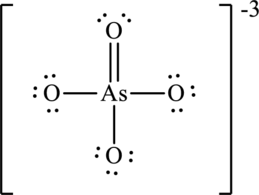
Arsenate ion
The symbol for oxygen is
The symbol for arsenic is
The symbol for chlorine is
Three negative charges on molecule is added up as three valence electrons in total count. Thus total valence electrons are sum of the valence electrons for each atom in arsenate ion
The skeleton structure in arsenate ion
To complete the valence electrons of arsenic, it forms double bond with one oxygen atom.
Hence, 22 electrons are allocated as 11 lone pairs on remaining oxygen atoms to complete their octet. The Lewis structure is as follows:
The negative charge is delocalized on each oxygen atom and since there are four oxygen atoms, therefore, four resonance structures will be formed and possible resonance structures are as,
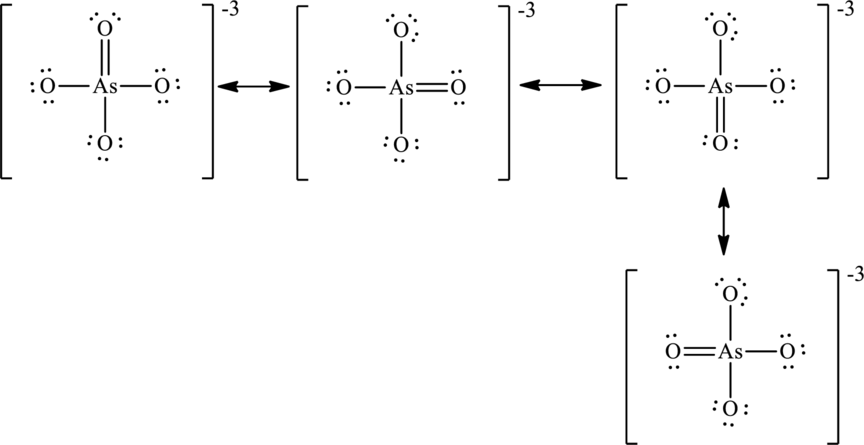
Want to see more full solutions like this?
Chapter 2 Solutions
CHEMICAL PRINCIPLES (LL) W/ACCESS
- I need help with the followingarrow_forwardFor Raman spectroscopy/imaging, which statement is not true regarding its disadvantages? a) Limited spatial resolution. b) Short integration time. c) A one-dimensional technique. d) Weak signal, only 1 in 108 incident photons is Raman scattered. e) Fluorescence interference.arrow_forwardUsing a cell of known pathlength b = 1.25115 x 10-3 cm, a water absorption spectrum was measured. The band at 1645 cm-1, assigned to the O-H bending, showed an absorbance, A, of 1.40. a) Assuming that water density is 1.00 g/mL, calculate the water molar concentration c (hint: M= mole/L) b) Calculate the molar absorptivity, a, of the 1645 cm-1 band c) The transmitted light, I, can be written as I= Ioexp(-xb), where x is the absorption coefficient (sometimes designated as alpha), Io is the input light, and b is the cell pathlength. Prove that x= (ln10)*x*c. (Please provide a full derivation of the equation for x from the equation for I). d) Calculate x for the 1645 cm-1 bandarrow_forward
- For CARS, which statement is not true regarding its advantages? a) Contrast signal based on vibrational characteristics, no need for fluorescent tagging. b) Stronger signals than spontaneous Raman. c) Suffers from fluorescence interference, because CARS signal is at high frequency. d) Faster, more efficient imaging for real-time analysis. e) Higher resolution than spontaneous Raman microscopy.arrow_forwardDraw the major product of the Claisen condensation reaction between two molecules of this ester. Ignore inorganic byproducts. Incorrect, 5 attempts remaining 1. NaOCH3/CH3OH 2. Acidic workup Select to Draw O Incorrect, 5 attempts remaining The total number of carbons in the parent chain is incorrect. Review the reaction conditions including starting materials and/or intermediate structures and recount the number of carbon atoms in the parent chain of your structure. OKarrow_forwardUsing a cell of known pathlength b = 1.25115 x 10-3 cm, a water absorption spectrum was measured. The band at 1645 cm-1, assigned to the O-H bending, showed an absorbance, A, of 1.40. a) Assuming that water density is 1.00 g/mL, calculate the water molar concentration c (hint: M= mole/L) b) Calculate the molar absorptivity, a, of the 1645 cm-1 band c) The transmitted light, I, can be written as I= Ioexp(-xb), where x is the absorption coefficient (sometimes designated as alpha), Io is the input light, and b is the cell pathlength. Prove that x= (ln10)*x*c d) Calculate x for the 1645 cm-1 bandarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

