
ACHIEVE/CHEMICAL PRINCIPLES ACCESS 1TERM
7th Edition
ISBN: 9781319399849
Author: ATKINS
Publisher: MAC HIGHER
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 2, Problem 2C.3BST
Interpretation Introduction
Interpretation:
Formal charges on respective atom in below structure have to be determined.
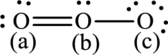
Concept Introduction:
The formal charge is the charge assigned on each atom irrespective of its electronegativity so that electrons are equally shared between the atoms. The formal charge on each atom in the Lewis structure can be calculated from the equation written as follows:
Here,
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Synthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.
Synthesize N-Methylcyclohexylamine from cyclohexanol using the necessary organic or inorganic reagents. Draw the structures of the compounds.
If possible, please provide the formula of the compound 3,3-dimethylbut-2-enal.
Chapter 2 Solutions
ACHIEVE/CHEMICAL PRINCIPLES ACCESS 1TERM
Ch. 2 - Prob. 2A.1ASTCh. 2 - Prob. 2A.1BSTCh. 2 - Prob. 2A.2ASTCh. 2 - Prob. 2A.2BSTCh. 2 - Prob. 2A.3ASTCh. 2 - Prob. 2A.3BSTCh. 2 - Prob. 2A.4ASTCh. 2 - Prob. 2A.4BSTCh. 2 - Prob. 2A.1ECh. 2 - Prob. 2A.2E
Ch. 2 - Prob. 2A.3ECh. 2 - Prob. 2A.4ECh. 2 - Prob. 2A.5ECh. 2 - Prob. 2A.6ECh. 2 - Prob. 2A.7ECh. 2 - Prob. 2A.8ECh. 2 - Prob. 2A.9ECh. 2 - Prob. 2A.10ECh. 2 - Prob. 2A.11ECh. 2 - Prob. 2A.12ECh. 2 - Prob. 2A.13ECh. 2 - Prob. 2A.14ECh. 2 - Prob. 2A.15ECh. 2 - Prob. 2A.16ECh. 2 - Prob. 2A.17ECh. 2 - Prob. 2A.18ECh. 2 - Prob. 2A.19ECh. 2 - Prob. 2A.20ECh. 2 - Prob. 2A.21ECh. 2 - Prob. 2A.22ECh. 2 - Prob. 2A.23ECh. 2 - Prob. 2A.24ECh. 2 - Prob. 2A.25ECh. 2 - Prob. 2A.26ECh. 2 - Prob. 2A.27ECh. 2 - Prob. 2A.28ECh. 2 - Prob. 2A.29ECh. 2 - Prob. 2A.30ECh. 2 - Prob. 2B.1ASTCh. 2 - Prob. 2B.1BSTCh. 2 - Prob. 2B.2ASTCh. 2 - Prob. 2B.2BSTCh. 2 - Prob. 2B.3ASTCh. 2 - Prob. 2B.3BSTCh. 2 - Prob. 2B.4ASTCh. 2 - Prob. 2B.4BSTCh. 2 - Prob. 2B.5ASTCh. 2 - Prob. 2B.5BSTCh. 2 - Prob. 2B.1ECh. 2 - Prob. 2B.2ECh. 2 - Prob. 2B.3ECh. 2 - Prob. 2B.4ECh. 2 - Prob. 2B.5ECh. 2 - Prob. 2B.6ECh. 2 - Prob. 2B.7ECh. 2 - Prob. 2B.8ECh. 2 - Prob. 2B.9ECh. 2 - Prob. 2B.10ECh. 2 - Prob. 2B.11ECh. 2 - Prob. 2B.12ECh. 2 - Prob. 2B.13ECh. 2 - Prob. 2B.14ECh. 2 - Prob. 2B.15ECh. 2 - Prob. 2B.16ECh. 2 - Prob. 2B.17ECh. 2 - Prob. 2B.18ECh. 2 - Prob. 2B.19ECh. 2 - Prob. 2B.20ECh. 2 - Prob. 2B.21ECh. 2 - Prob. 2B.22ECh. 2 - Prob. 2B.23ECh. 2 - Prob. 2B.24ECh. 2 - Prob. 2C.1ASTCh. 2 - Prob. 2C.1BSTCh. 2 - Prob. 2C.2ASTCh. 2 - Prob. 2C.2BSTCh. 2 - Prob. 2C.3ASTCh. 2 - Prob. 2C.3BSTCh. 2 - Prob. 2C.1ECh. 2 - Prob. 2C.2ECh. 2 - Prob. 2C.3ECh. 2 - Prob. 2C.4ECh. 2 - Prob. 2C.5ECh. 2 - Prob. 2C.6ECh. 2 - Prob. 2C.7ECh. 2 - Prob. 2C.8ECh. 2 - Prob. 2C.9ECh. 2 - Prob. 2C.10ECh. 2 - Prob. 2C.11ECh. 2 - Prob. 2C.12ECh. 2 - Prob. 2C.13ECh. 2 - Prob. 2C.14ECh. 2 - Prob. 2C.15ECh. 2 - Prob. 2C.16ECh. 2 - Prob. 2C.17ECh. 2 - Prob. 2C.18ECh. 2 - Prob. 2D.1ASTCh. 2 - Prob. 2D.1BSTCh. 2 - Prob. 2D.2ASTCh. 2 - Prob. 2D.2BSTCh. 2 - Prob. 2D.1ECh. 2 - Prob. 2D.2ECh. 2 - Prob. 2D.3ECh. 2 - Prob. 2D.4ECh. 2 - Prob. 2D.5ECh. 2 - Prob. 2D.6ECh. 2 - Prob. 2D.7ECh. 2 - Prob. 2D.8ECh. 2 - Prob. 2D.9ECh. 2 - Prob. 2D.10ECh. 2 - Prob. 2D.11ECh. 2 - Prob. 2D.12ECh. 2 - Prob. 2D.13ECh. 2 - Prob. 2D.14ECh. 2 - Prob. 2D.15ECh. 2 - Prob. 2D.16ECh. 2 - Prob. 2D.17ECh. 2 - Prob. 2D.18ECh. 2 - Prob. 2D.19ECh. 2 - Prob. 2D.20ECh. 2 - Prob. 2E.1ASTCh. 2 - Prob. 2E.1BSTCh. 2 - Prob. 2E.2ASTCh. 2 - Prob. 2E.2BSTCh. 2 - Prob. 2E.3ASTCh. 2 - Prob. 2E.3BSTCh. 2 - Prob. 2E.4ASTCh. 2 - Prob. 2E.4BSTCh. 2 - Prob. 2E.5ASTCh. 2 - Prob. 2E.5BSTCh. 2 - Prob. 2E.1ECh. 2 - Prob. 2E.2ECh. 2 - Prob. 2E.3ECh. 2 - Prob. 2E.4ECh. 2 - Prob. 2E.5ECh. 2 - Prob. 2E.6ECh. 2 - Prob. 2E.7ECh. 2 - Prob. 2E.8ECh. 2 - Prob. 2E.9ECh. 2 - Prob. 2E.10ECh. 2 - Prob. 2E.11ECh. 2 - Prob. 2E.12ECh. 2 - Prob. 2E.13ECh. 2 - Prob. 2E.14ECh. 2 - Prob. 2E.15ECh. 2 - Prob. 2E.16ECh. 2 - Prob. 2E.17ECh. 2 - Prob. 2E.18ECh. 2 - Prob. 2E.19ECh. 2 - Prob. 2E.20ECh. 2 - Prob. 2E.21ECh. 2 - Prob. 2E.22ECh. 2 - Prob. 2E.23ECh. 2 - Prob. 2E.24ECh. 2 - Prob. 2E.25ECh. 2 - Prob. 2E.26ECh. 2 - Prob. 2E.27ECh. 2 - Prob. 2E.28ECh. 2 - Prob. 2E.29ECh. 2 - Prob. 2E.30ECh. 2 - Prob. 2F.1ASTCh. 2 - Prob. 2F.1BSTCh. 2 - Prob. 2F.2ASTCh. 2 - Prob. 2F.2BSTCh. 2 - Prob. 2F.3ASTCh. 2 - Prob. 2F.3BSTCh. 2 - Prob. 2F.4ASTCh. 2 - Prob. 2F.4BSTCh. 2 - Prob. 2F.1ECh. 2 - Prob. 2F.2ECh. 2 - Prob. 2F.3ECh. 2 - Prob. 2F.4ECh. 2 - Prob. 2F.5ECh. 2 - Prob. 2F.6ECh. 2 - Prob. 2F.7ECh. 2 - Prob. 2F.8ECh. 2 - Prob. 2F.9ECh. 2 - Prob. 2F.10ECh. 2 - Prob. 2F.11ECh. 2 - Prob. 2F.12ECh. 2 - Prob. 2F.13ECh. 2 - Prob. 2F.14ECh. 2 - Prob. 2F.15ECh. 2 - Prob. 2F.16ECh. 2 - Prob. 2F.17ECh. 2 - Prob. 2F.18ECh. 2 - Prob. 2F.19ECh. 2 - Prob. 2F.20ECh. 2 - Prob. 2F.21ECh. 2 - Prob. 2G.1ASTCh. 2 - Prob. 2G.1BSTCh. 2 - Prob. 2G.2ASTCh. 2 - Prob. 2G.2BSTCh. 2 - Prob. 2G.1ECh. 2 - Prob. 2G.2ECh. 2 - Prob. 2G.3ECh. 2 - Prob. 2G.4ECh. 2 - Prob. 2G.5ECh. 2 - Prob. 2G.6ECh. 2 - Prob. 2G.7ECh. 2 - Prob. 2G.8ECh. 2 - Prob. 2G.9ECh. 2 - Prob. 2G.11ECh. 2 - Prob. 2G.12ECh. 2 - Prob. 2G.13ECh. 2 - Prob. 2G.14ECh. 2 - Prob. 2G.15ECh. 2 - Prob. 2G.16ECh. 2 - Prob. 2G.17ECh. 2 - Prob. 2G.18ECh. 2 - Prob. 2G.19ECh. 2 - Prob. 2G.20ECh. 2 - Prob. 2G.21ECh. 2 - Prob. 2G.22ECh. 2 - Prob. 2.1ECh. 2 - Prob. 2.2ECh. 2 - Prob. 2.3ECh. 2 - Prob. 2.4ECh. 2 - Prob. 2.5ECh. 2 - Prob. 2.6ECh. 2 - Prob. 2.7ECh. 2 - Prob. 2.8ECh. 2 - Prob. 2.9ECh. 2 - Prob. 2.10ECh. 2 - Prob. 2.11ECh. 2 - Prob. 2.12ECh. 2 - Prob. 2.13ECh. 2 - Prob. 2.14ECh. 2 - Prob. 2.17ECh. 2 - Prob. 2.19ECh. 2 - Prob. 2.22ECh. 2 - Prob. 2.23ECh. 2 - Prob. 2.24ECh. 2 - Prob. 2.25ECh. 2 - Prob. 2.26ECh. 2 - Prob. 2.27ECh. 2 - Prob. 2.28ECh. 2 - Prob. 2.29ECh. 2 - Prob. 2.30ECh. 2 - Prob. 2.31ECh. 2 - Prob. 2.32ECh. 2 - Prob. 2.33ECh. 2 - Prob. 2.34ECh. 2 - Prob. 2.35ECh. 2 - Prob. 2.36ECh. 2 - Prob. 2.37ECh. 2 - Prob. 2.39ECh. 2 - Prob. 2.40ECh. 2 - Prob. 2.41ECh. 2 - Prob. 2.42ECh. 2 - Prob. 2.43ECh. 2 - Prob. 2.44ECh. 2 - Prob. 2.45ECh. 2 - Prob. 2.46ECh. 2 - Prob. 2.47ECh. 2 - Prob. 2.48ECh. 2 - Prob. 2.49ECh. 2 - Prob. 2.50ECh. 2 - Prob. 2.51ECh. 2 - Prob. 2.52ECh. 2 - Prob. 2.53ECh. 2 - Prob. 2.54ECh. 2 - Prob. 2.55ECh. 2 - Prob. 2.56ECh. 2 - Prob. 2.57ECh. 2 - Prob. 2.58ECh. 2 - Prob. 2.59ECh. 2 - Prob. 2.60ECh. 2 - Prob. 2.61ECh. 2 - Prob. 2.62ECh. 2 - Prob. 2.63ECh. 2 - Prob. 2.64E
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Synthesize 1,4-dibromobenzene from acetanilide (N-phenylacetamide) using the necessary organic or inorganic reagents. Draw the structures of the compounds.arrow_forwardIndicate the products obtained by mixing (3-oxo-3-phenylpropyl)triphenylphosphonium bromide with sodium hydride.arrow_forwardWe mix N-ethyl-2-hexanamine with excess methyl iodide and followed by heating with aqueous Ag2O. Indicate the major products obtained.arrow_forward
- Indicate the products obtained by mixing acetophenone with iodine and NaOH.arrow_forwardIndicate the products obtained by mixing 2-Propanone and ethyllithium and performing a subsequent acid hydrolysis.arrow_forwardIndicate the products obtained if (E)-2-butenal and 3-oxo-butanenitrile are mixed with sodium ethoxide in ethanol.arrow_forward
- Question 3 (4 points), Draw a full arrow-pushing mechanism for the following reaction Please draw all structures clearly. Note that this intramolecular cyclization is analogous to the mechanism for halohydrin formation. COH Br + HBr Brarrow_forwardIndicate the products obtained if 2,2-dimethylpropanal and acetaldehyde are mixed with sodium ethoxide in ethanol.arrow_forwardIndicate the products obtained if 2,2-dimethylpropanal and acetaldehyde are reacted with sodium ethoxide in ethanol.arrow_forward
- 2,2-Dimethylpropanal and acetaldehyde are reacted with sodium ethoxide in ethanol. Indicate the products obtained.arrow_forwardAdd conditions above and below the arrow that turn the reactant below into the product below in a single transformationADS fint anditions 百 Abl res condinese NC ง Add on condtions 1.0 B H,N.arrow_forward3. Provide all the steps and reagents for this synthesis. OHarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
INTRODUCTION TO MOLECULAR QUANTUM MECHANICS -Valence bond theory - 1; Author: AGK Chemistry;https://www.youtube.com/watch?v=U8kPBPqDIwM;License: Standard YouTube License, CC-BY