
Concept explainers
a) Bromoacetone
Interpretation:
The structure for bromoacetone is to be shown.
Concept introduction:
To show:
The structure for bromoacetone.
Answer to Problem 54AP
The structure of bromoacetone is
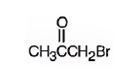
Explanation of Solution
The name indicates that the compound is a ketone with three carbon straight chain with a bromine atom attached to C1.
The structure of bromoacetone is

b) (S)-2-Hydroxypropanal
Interpretation:
The structure for (S)-2-hydroxypropanal is to be shown.
Concept introduction:
To show:
The structure for (S)-2-hydroxypropanal.
Answer to Problem 54AP
The structure of (S)-2-hydroxypropanal is
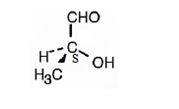
Explanation of Solution
The name indicates that the compound is an aldehyde with three carbon straight chain with a hydroxyl group attached to C2. The molecule is chiral. The three groups, -OH(first highest priority), -CHO (second highest priority) and -CH3(third highest priority) are arranged anticlockwise when viewed from the side away from H (fourth highest priority). Hence it has S stereochemistry.
The structure of (S)-2-hydroxypropanal is

c) 2-Methyl-3-heptanone
Interpretation:
The structure for 2-methyl-3-heptanone is to be shown.
Concept introduction:
Ketones are named by replacing the terminal –e of the parent alkane with –one. The parent chain is the longest one that includes the ketone group and the numbering begins at the end nearer to the carbonyl carbon. If other functional groups are present the double bonded oxygen is considered as a substituent on the parent chain with the prefix –oxo.
To show:
The structure for 2-methyl-3-heptanone.
Answer to Problem 54AP
The structure of 2-methyl-3-heptanone is
Explanation of Solution
The name indicates that the compound is a ketone with seven carbon straight chain having the keto group at position three and a methyl group attached to C2.
The structure of 2-methyl-3-heptanone is
d) (2S,3R)-2,3,4-Trihydroxybutanal
Interpretation:
The structure for (2S,3R)-2,3,4-trihydroxybutanal is to be shown.
Concept introduction:
Aldehydes are named by replacing the terminal –e of the parent alkane with –al. The parent chain is the longest one that includes the -CHO group and the –CHO group is numbered as carbon 1. For cyclic alcohols in which the –CHO group is directly attached to the ring, the suffix –carbaldehyde is used.
To show:
The structure for (2S,3R)-2,3,4-trihydroxybutanal.
Answer to Problem 54AP
The structure of (2S,3R)-2,3,4-trihydroxybutanal is
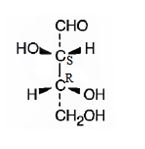
Explanation of Solution
The name indicates that the compound is an aldehyde with four carbon straight chain and has three hydroxyl groups attached to C2, C3 and C4.
The molecule is chiral. The C2 is attached to the three groups, -OH(first highest priority), -CHO (second highest priority) and –C3 (third highest priority) arranged anticlockwise when viewed from the side away from H (fourth highest priority). Hence it has S stereochemistry.
The C3 is attached to the three groups, -OH(first highest priority), –C2 (second highest priority) and –CH2OH--(third highest priority) arranged clockwise when viewed from the side away from H (fourth highest priority). Hence it has R stereochemistry.
The structure of (2S,3R)-2,3,4-trihydroxybutanal is

e) 2,2,4,4-Tetramethyl-3-pentanone
Interpretation:
The structure for 2,2,4,4-tetramethyl-3-pentanone is to be shown.
Concept introduction:
Ketones are named by replacing the terminal –e of the parent alkane with –one. The parent chain is the longest one that includes the ketone group and the numbering begins at the end nearer to the carbonyl carbon. If other functional groups are present the double bonded oxygen is considered as a substituent on the parent chain with the prefix –oxo.
To show:
The structure for 2,2,4,4-tetramethyl-3-pentanone.
Answer to Problem 54AP
The structure of 2,2,4,4-tetramethyl-3-pentanone is
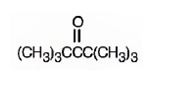
Explanation of Solution
The name indicates that the compound is a ketone with five carbon straight chain with a keto group at position three attached to four methyl groups, two on C2 and other two on C4.
The structure of 2,2,4,4-Tetramethyl-3-pentanone is
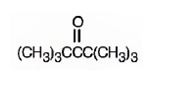
f) 4-methyl-3-penten-2-one
Interpretation:
The structure for 4-methyl-3-penten-2-one is to be shown.
Concept introduction:
Ketones are named by replacing the terminal –e of the parent alkane with –one. The parent chain is the longest one that includes the ketone group and the numbering begins at the end nearer to the carbonyl carbon. If other functional groups are present the double bonded oxygen is considered as a substituent on the parent chain with the prefix –oxo.
To show:
The structure for 4-methyl-3-penten-2-one.
Answer to Problem 54AP
The structure of 4-methyl-3-penten-2-one is
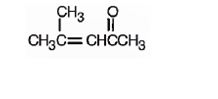
Explanation of Solution
The name indicates that the compound is a ketone containing a five carbon straight chain, having a keto group at position two and a double bond between C3 and C4 with a methyl group on C4.
The structure of 4-methyl-3-penten-2-one is

g) Butanedial
Interpretation:
The structure for butanedial is to be shown.
Concept introduction:
Aldehydes are named by replacing the terminal –e of the parent alkane with –al. The parent chain is the longest one that includes the -CHO group and the –CHO group is numbered as carbon 1. For cyclic alcohols in which the –CHO group is directly attached to the ring, the suffix –carbaldehyde is used.
To show:
The structure for butanedial.
Answer to Problem 54AP
The structure of butanedial is
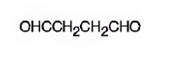
Explanation of Solution
The name of the compound indicates that it has a four carbon straight chain with two aldehyde groups at both ends.
The structure of butanedial is

h) 3-Phenyl-2-propenal
Interpretation:
The structure for 3-phenyl-2-propenal is to be shown.
Concept introduction:
Aldehydes are named by replacing the terminal –e of the parent alkane with –al. The parent chain is the longest one that includes the -CHO group and the –CHO group is numbered as carbon 1. For cyclic alcohols in which the –CHO group is directly attached to the ring, the suffix –carbaldehyde is used.
To show:
The structure for 3-phenyl-2-propenal.
Answer to Problem 54AP
The structure of 3-phenyl-2-propenal is
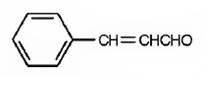
Explanation of Solution
The name of the compound indicates that the compound is a three carbon aldehyde with a double bond between C2 & C3 and has a phenyl group attached to C3.
The structure of 3-phenyl-2-propenal is

i) 6,6-Dimethyl-2,4-cyclohexadienone
Interpretation:
The structure for 6,6-dimethyl-2,4-cyclohexadienone is to be shown.
Concept introduction:
Ketones are named by replacing the terminal –e of the parent alkane with –one. The parent chain is the longest one that includes the ketone group and the numbering begins at the end nearer to the carbonyl carbon. If other functional groups are present the double bonded oxygen is considered as a substituent on the parent chain with the prefix –oxo.
To show:
The structure for 6,6-dimethyl-2,4-cyclohexadienone.
Answer to Problem 54AP
The structure of 6,6-dimethyl-2,4-cyclohexadienone is
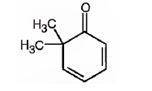
Explanation of Solution
The name of the compound indicates that it is a cyclic ketone with a cyclohexadiene ring containing two double bonds, one between C2 & C3 and other between C4 & C5. It also has two methyl groups on C6.
The structure of 6,6-dimethyl-2,4-cyclohexadienone is

j) p-Nitroacetophenone
Interpretation:
The structure for p-nitroacetophenone is to be shown.
Concept introduction:
Ketones are named by replacing the terminal –e of the parent alkane with –one. The parent chain is the longest one that includes the ketone group and the numbering begins at the end nearer to the carbonyl carbon. If other functional groups are present the double bonded oxygen is considered as a substituent on the parent chain with the prefix –oxo. Some common names like acetophenone are retained by IUPAC.
To show:
The structure for p-nitroacetophenone.
Answer to Problem 54AP
The structure of p-nitroacetophenone is
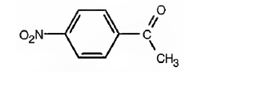
Explanation of Solution
The name of the compound indicates that it contains an actyl and nitro groups attached to a benzene ring in para relationship.
The structure of p-nitroacetophenone is

Want to see more full solutions like this?
Chapter 19 Solutions
Study Guide with Student Solutions Manual for McMurry's Organic Chemistry, 9th
- Determine if the following salt is neutral, acidic or basic. If acidic or basic, write the appropriate equilibrium equation for the acid or base that exists when the salt is dissolved in aqueous solution. If neutral, simply write only NR. Be sure to include the proper phases for all species within the reaction LiNO3arrow_forwardAn unknown weak acid with a concentration of 0.410 M has a pH of 5.600. What is the Ka of the weak acid?arrow_forward(racemic) 19.84 Using your reaction roadmaps as a guide, show how to convert 2-oxepanone and ethanol into 1-cyclopentenecarbaldehyde. You must use 2-oxepanone as the source of all carbon atoms in the target molecule. Show all reagents and all molecules synthesized along the way. & + EtOH H 2-Oxepanone 1-Cyclopentenecarbaldehydearrow_forward
- R₂ R₁ R₁ a R Rg Nu R₂ Rg R₁ R R₁₂ R3 R R Nu enolate forming R₁ R B-Alkylated carbonyl species or amines Cyclic B-Ketoester R₁₁ HOB R R₁B R R₁₂ B-Hydroxy carbonyl R diester R2 R3 R₁ RB OR R₂ 0 aB-Unsaturated carbonyl NaOR Aldol HOR reaction 1) LDA 2) R-X 3) H₂O/H₂O ketone, aldehyde 1) 2°-amine 2) acid chloride 3) H₂O'/H₂O 0 O R₁ R₁ R R₁ R₁₂ Alkylated a-carbon R₁ H.C R₁ H.C Alkylated methyl ketone acetoacetic ester B-Ketoester ester R₁ HO R₂ R B-Dicarbonyl HO Alkylated carboxylic acid malonic ester Write the reagents required to bring about each reaction next to the arrows shown. Next, record any regiochemistry or stereochemistry considerations relevant to the reaction. You should also record any key aspects of the mechanism, such as forma- tion of an important intermediate, as a helpful reminder. You may want to keep track of all reactions that make carbon-carbon bonds, because these help you build large molecules from smaller fragments. This especially applies to the reactions in…arrow_forwardProvide the reasonable steps to achieve the following synthesis.arrow_forwardIdentify which compound is more acidic. Justify your choice.arrow_forward
- Provide the reasonable steps to achieve the following synthesis.arrow_forwardWhen anisole is treated with excess bromine, the reaction gives a product which shows two singlets in 1H NMR. Draw the product.arrow_forward(ii) Draw a reasonable mechanism for the following reaction: CI NaOH heat OH (hint: SNAr Reaction) :arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


