
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 43P
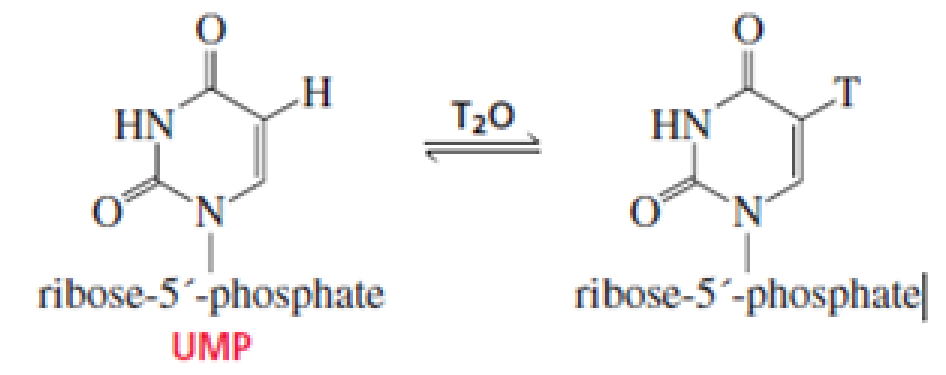
When UMP is dissolved in T2O, exchange of T for H occurs at the 5 position. Propose a mechanism for the exchange. (T is 3H, a hydrogen with two neutrons.)
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
#1. Retro-Electrochemical Reaction: A ring has been made, but the light is causing the molecule to un-
cyclize. Undo the ring into all possible molecules. (2pts, no partial credit)
hv
Don't used Ai solution
I have a question about this problem involving mechanisms and drawing curved arrows for acids and bases. I know we need to identify the nucleophile and electrophile, but are there different types of reactions? For instance, what about Grignard reagents and other types that I might not be familiar with? Can you help me with this? I want to identify the names of the mechanisms for problems 1-14, such as Gilman reagents and others. Are they all the same? Also, could you rewrite it so I can better understand? The handwriting is pretty cluttered. Additionally, I need to label the nucleophile and electrophile, but my main concern is whether those reactions differ, like the "Brønsted-Lowry acid-base mechanism, Lewis acid-base mechanism, acid-catalyzed mechanisms, acid-catalyzed reactions, base-catalyzed reactions, nucleophilic substitution mechanisms (SN1 and SN2), elimination reactions (E1 and E2), organometallic mechanisms, and so forth."
Chapter 18 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 18.1 - Prob. 1PCh. 18.2 - If H218O were used to hydrolyze lysozyme, which...Ch. 18.3 - Which of the following amino acid side chains can...Ch. 18.3 - Arginine and lysine side chains fit into trypsins...Ch. 18.4 - Which of the following amino acid side chains can...Ch. 18.4 - Prob. 6PCh. 18.5 - Prob. 7PCh. 18.5 - Draw the mechanism for the hydroxide-ion-catalyzed...Ch. 18.5 - What advantage does the enzyme gain by forming an...Ch. 18.7 - Prob. 10P
Ch. 18.7 - Prob. 11PCh. 18.8 - How many conjugated double bonds are there in a....Ch. 18.8 - Instead of adding to the 4a-position and...Ch. 18.8 - In succinate dehydrogenase, FAD is covalently...Ch. 18.8 - Prob. 15PCh. 18.9 - Acetolactate synthase is another TPP-requiring...Ch. 18.9 - Acetolactate synthase can also transfer the acyl...Ch. 18.9 - Prob. 18PCh. 18.9 - Prob. 19PCh. 18.10 - Prob. 21PCh. 18.11 - Prob. 23PCh. 18.11 - Which compound is more easily decarboxylated?Ch. 18.11 - Explain why the ability of PLP to catalyze an...Ch. 18.11 - Explain why the ability of PLP to catalyze an...Ch. 18.12 - What groups are interchanged in the following...Ch. 18.13 - Why is the coenzyme called tetrahydrofolate?Ch. 18.13 - What amino acid is formed by the following...Ch. 18.13 - How do the structures of tetrahydrofolate and...Ch. 18.13 - What is the source of the methyl group in...Ch. 18 - Prob. 32PCh. 18 - Prob. 33PCh. 18 - From what vitamins are the following coenzymes...Ch. 18 - Prob. 35PCh. 18 - For each of the following reaction, name both the...Ch. 18 - Explain why serine proteases do not catalyze...Ch. 18 - Prob. 38PCh. 18 - For each of the following enzyme catalyzed...Ch. 18 - Trisephosphate isomerase (TIM) catalyzes the...Ch. 18 - Prob. 41PCh. 18 - What acyl groups have we seen transferred by...Ch. 18 - When UMP is dissolved in T2O, exchange of T for H...Ch. 18 - Prob. 44PCh. 18 - When transaminated, the three branched-chain amino...Ch. 18 - Aldolase shows no activity if it is incubated with...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 2. 200 LOD For an unknown compound with a molecular ion of 101 m/z: a. Use the molecular ion to propose at least two molecular formulas. (show your work) b. What is the DU for each of your possible formulas? (show your work) C. Solve the structure and assign each of the following spectra. 8 6 4 2 (ppm) 150 100 50 ō (ppm) 4000 3000 2000 1500 1000 500 HAVENUMBERI-11arrow_forwardComplete the spectroscopy with structurearrow_forwardComplete the spectroscopy with structurearrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

DIGESTER-35 | VITAMINS AND THEIR RELATED COENZYMES| GPAT | NIPER | PHARMACIST| DI; Author: GPAT DISCUSSION CENTER;https://www.youtube.com/watch?v=CGrdNYmho0s;License: Standard YouTube License, CC-BY