
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 18, Problem 34P
Give an acceptable name for each
a.
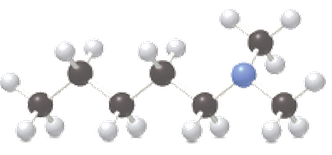
b.
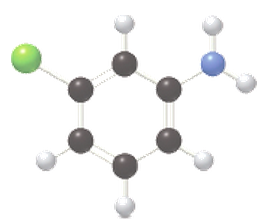
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Draw a tetramer of this alternating copolymer.
H
I
T
H
HH
H
-H
C.
H-
Identify and select all structures below that represent a constitutional isomer(s) of the compound shown above.
H-
H
CIH
H
H
H
HHHH
H
H
0
·H
H–
冊
CH
CHI
HH C-
H-
H
H-
H
H
A.
H
H C
H
H-
-H
HH
H
B.
H-
-H
D.
H
H H
H
•
H
-H
E.
-H
H
H
HICH
T
HHH
F.
H-
Polylactic acid (shown below) is a biodegradable polymer used for food packaging. Identify the monomer(s) used in the production of this polymer using a condensation process.
Chapter 18 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 18.1 - Prob. 18.1PPCh. 18.1 - Prob. 18.1PCh. 18.1 - Prob. 18.2PCh. 18.1 - Prob. 18.3PCh. 18.2 - Prob. 18.2PPCh. 18.2 - Prob. 18.4PCh. 18.2 - Prob. 18.5PCh. 18.3 - Prob. 18.3PPCh. 18.4 - Decaffeinated coffee is produced by extracting the...Ch. 18.4 - Prob. 18.7P
Ch. 18.5 - Prob. 18.8PCh. 18.5 - Naloxone is a drug used to treat overdoses of...Ch. 18.6 - Prob. 18.4PPCh. 18.6 - Prob. 18.10PCh. 18.6 - Prob. 18.11PCh. 18.6 - Name each ammonium salt. a. ( CH3 NH3)+Cl b. [( CH...Ch. 18.6 - Prob. 18.6PPCh. 18.6 - Prob. 18.12PCh. 18.7 - Prob. 18.13PCh. 18.7 - Prob. 18.14PCh. 18.8 - Prob. 18.15PCh. 18.8 - Prob. 18.16PCh. 18.8 - Prob. 18.17PCh. 18.8 - Prob. 18.18PCh. 18.9 - Prob. 18.19PCh. 18.9 - Prob. 18.20PCh. 18.9 - Prob. 18.21PCh. 18.10 - Prob. 18.22PCh. 18 - Prob. 23PCh. 18 - Prob. 24PCh. 18 - Prob. 25PCh. 18 - Prob. 26PCh. 18 - Prob. 27PCh. 18 - Prob. 28PCh. 18 - Prob. 29PCh. 18 - Prob. 30PCh. 18 - Prob. 31PCh. 18 - Prob. 32PCh. 18 - Give an acceptable name for each amine. a. b.Ch. 18 - Give an acceptable name for each amine. a. b.Ch. 18 - Give an acceptable name for each amine. a. b. c....Ch. 18 - Give an acceptable name for each amine. a. CH3(...Ch. 18 - Prob. 37PCh. 18 - Prob. 38PCh. 18 - Prob. 39PCh. 18 - Prob. 40PCh. 18 - Prob. 41PCh. 18 - Prob. 42PCh. 18 - Prob. 43PCh. 18 - Prob. 44PCh. 18 - Which compound in each pair has the higher boiling...Ch. 18 - Which compound in each pair has the higher boiling...Ch. 18 - Draw the hydrogen-bonding interactions that occur...Ch. 18 - Prob. 48PCh. 18 - Prob. 49PCh. 18 - Which compound has the higher water solubility:...Ch. 18 - Prob. 51PCh. 18 - Prob. 52PCh. 18 - Draw the products of each acid-base reaction. a....Ch. 18 - Draw the products of each acid-base reaction. a....Ch. 18 - Prob. 55PCh. 18 - Prob. 56PCh. 18 - What type of nitrogen heterocycle occurs in both...Ch. 18 - Only one of the N atoms in nicotine has a trigonal...Ch. 18 - Prob. 59PCh. 18 - Prob. 60PCh. 18 - Why are aqueous solutions of an alkaloid slightly...Ch. 18 - Prob. 62PCh. 18 - Prob. 63PCh. 18 - Explain why patients with Parkinson’s disease...Ch. 18 - Prob. 65PCh. 18 - Prob. 66PCh. 18 - Prob. 67PCh. 18 - Prob. 68PCh. 18 - Locate the atoms of 2-phenylethylamine in the...Ch. 18 - Locate the atoms of 2-phenylethylamine in the...Ch. 18 - Give an example of an antihistamine. Explain how...Ch. 18 - Give an example of an anti-ulcer drug, and explain...Ch. 18 - Prob. 73PCh. 18 - Prob. 74PCh. 18 - Prob. 75PCh. 18 - Prob. 76PCh. 18 - Prob. 77PCh. 18 - Prob. 78PCh. 18 - Prob. 79PCh. 18 - Why do some antihistamines cause drowsiness while...Ch. 18 - Prob. 81PCh. 18 - Prob. 82PCh. 18 - Compare the structures of morphine and heroin....Ch. 18 - Prob. 84CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the product of the reaction shown below. Ignore small byproducts that would evaporate pleasearrow_forwardPoly(ethylene adipate) is a biodegradable polyester (shown below). Identify the type of polymerization process used in the production of this polymer.arrow_forwardPolymers may be composed of thousands of monomers. draw two repeat units(dimer) of the polymer formed in this reaction. assume there are hydrogen atoms on the two ends of the dimer. ignore inorganic byproducts pleasearrow_forward
- Draw the product of the reaction shown below. Use a dash or wedge bond to indicate stereochemistry of substituents on asymmetric centers, Ignore inorganic byproductsarrow_forwardDraw the product of this reaction please. Ignore inorganic byproductsarrow_forwardOne of the pi molecular orbitals of 1,3-butadiene (CH2=CHCH=CH2) is shown below. Please identify the number of nodal planes perpendicular to the bonding axisarrow_forward
- Draw the monomers required to synthesize this condensation polymer please.arrow_forwardProvide the correct systematic name for the compound shown here. Please take into account the keyboard options belowarrow_forwardcurved arrows are used to illustrate the flow of electrons. using the provided starting and product structures, draw the curved electron-pushing arrows for the following reaction or mechanistic step(s)arrow_forward
- Identify the 'cartoon' drawing of the acceptor orbital in the first mechanistic step of an electrophilic addition reaction of butadiene with HBr. Pleasearrow_forwardH- H H H H H H Identify and select all structures below that represent a constitutional isomer(s) of the compound shown above. H- H H H A. H H H H-C CI H H D. H H H H H H C C -H H C C H H H H B. H CI H H- C C H H H H E. H CI H C.arrow_forwardWhy doesn't this carry on to form a ring by deprotonating the alpha carbon and the negatively-charged carbon attacking the C=O?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning
Nomenclature: Crash Course Chemistry #44; Author: CrashCourse;https://www.youtube.com/watch?v=U7wavimfNFE;License: Standard YouTube License, CC-BY