
Concept explainers
(a)
To determine: The type of reaction in which leucine is converted to α-ketoisocaproate.
Introduction:
The series of
β-oxidation is the oxidation of fatty acids in cells in absence of glucose and glycogen, to produce ATP. Oxidation takes place at the β carbon, thus, this reaction is termed as β-oxidation.
(a)
Explanation of Solution
Pictorial representation: Fig. 1 represents the transamination reaction.
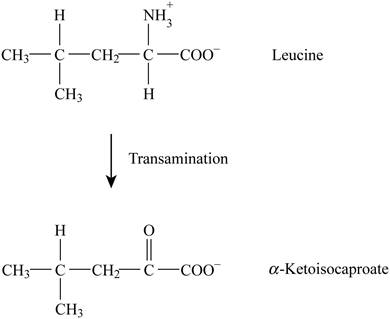
Fig. 1: Transamination reaction.
Explanation:
The transamination reaction is involved in the conversion of leucine into alpha ketoisocaproate. The co-factor involved in this reaction is PLP (pyridoxal phosphate). There is no analogous reaction to this reaction from citric acid cycle and β-oxidation.
(b)
To determine: The type of reaction in which Ketoisocaproate is converted to Isovaleryl-CoA.
Introduction:
The series of metabolic reactions, in which the energy stored in the compounds is released for the production of ATP (adenosine triphosphate), is called “Krebs’s cycle”. The pyruvate undergoes series of reactions in the citric acid cycle and converts into Acetyl CoA (acetyl coenzyme A) in the presence of oxygen. β-oxidation is the oxidation of fatty acids in cells in absence of glucose and glycogen, to produce ATP. Oxidation takes place at the β carbon, thus, this reaction is termed as β-oxidation.
(b)
Explanation of Solution
Pictorial representation: Fig. 2 represents the oxidative decarboxylation.
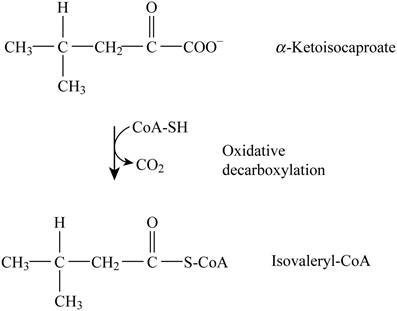
Fig. 2: Oxidative decarboxylation.
Explanation:
The oxidative decarboxylation is occurring that converts Ketoisocaproate to Isovaleryl-CoA. The cofactors involved are “NAD+ (Nicotinamide adenine dinucleotide)”, “TPP (Triphenyl phosphate)” and “FAD (Flavin adenine dinucleotide)”, and “lipoate”. The formation of acetyl CoA by oxidative decarboxylation of pyruvate is similar to this given reaction.
(c)
To determine: The type of reaction in which Isovaleryl-CoA is converted to β-methylcrotonyl-CoA.
Introduction:
The series of metabolic reactions, in which the energy stored in the compounds is released for the production of ATP (adenosine triphosphate), is called “Krebs’s cycle”. The pyruvate undergoes series of reactions in the citric acid cycle and converts into Acetyl CoA (acetyl coenzyme A) in the presence of oxygen.
β-oxidation is the oxidation of fatty acids in cells in absence of glucose and glycogen, to produce ATP. Oxidation takes place at the β carbon, thus, this reaction is termed as β-oxidation.
(c)
Explanation of Solution
Pictorial representation: Fig. 3 represents the dehydrogenation reaction.
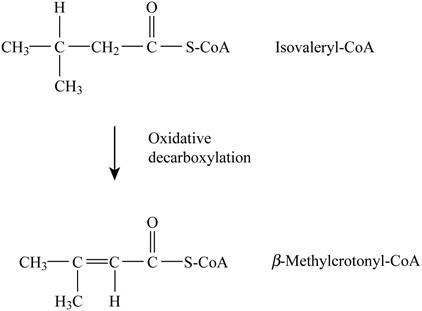
Fig. 3: Dehydrogenation reaction.
Explanation:
The type of reaction in which Isovaleryl-CoA is converted to β-methylcrotonyl-CoA is dehydrogenation. The cofactor is (FAD) Flavin adenine
(d)
To determine: The type of reaction in which beta methylcrotonyl-CoA is converted to beta-methylglutaconyl-CoA.
Introduction:
The series of metabolic reactions, in which the energy stored in the compounds is released for the production of ATP (adenosine triphosphate), is called “Krebs’s cycle”. The pyruvate undergoes series of reactions in the citric acid cycle and converts into Acetyl CoA (acetyl coenzyme A) in the presence of oxygen. β-oxidation is the oxidation of fatty acids in cells in absence of glucose and glycogen, to produce ATP. Oxidation takes place at the β carbon, thus, this reaction is termed as β-oxidation.
(d)
Explanation of Solution
Pictorial representation: Fig. 4 represents the carboxylation reaction.
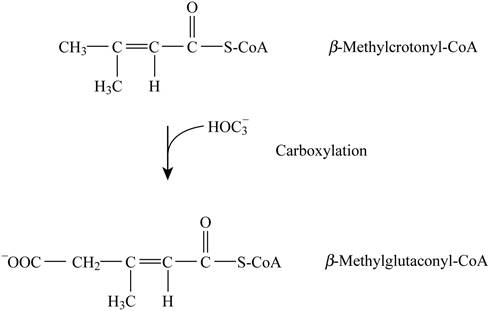
Fig. 4: Carboxylation reaction.
Explanation:
The formation of β-methylglutaconyl-CoA by β-methylcrotonyl-CoA is a type of carboxylation reaction. The cofactors are biotin and ATP. This step is not analogous to any step in β-oxidation and citric acid cycle.
(e)
To determine: The type of reaction in which beta-methylglutaconyl-CoA is converted to β-hydroxy β-methylglutaryl-CoA.
Introduction:
The series of metabolic reactions, in which the energy stored in the compounds is released for the production of ATP (adenosine triphosphate), is called “Krebs’s cycle”. The pyruvate undergoes series of reactions in the citric acid cycle and converts into Acetyl CoA (acetyl coenzyme A) in the presence of oxygen. β-oxidation is the oxidation of fatty acids in cells in absence of glucose and glycogen, to produce ATP. Oxidation takes place at the β carbon, thus, this reaction is termed as β-oxidation.
(e)
Explanation of Solution
Pictorial representation: Fig. 5 represents the hydration reaction.
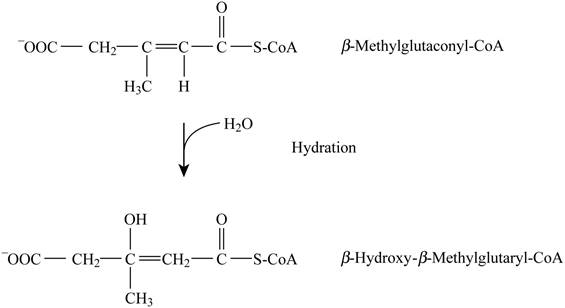
Fig. 5: Hydration reaction.
Explanation:
The conversion of beta-methylglutaconyl-CoA into β-hydroxy β-methylglutaryl-CoA is a type of hydration reaction. This reaction does not require the cofactors. The formation of malate by fumarate and formation of 3-hydroxyacyl-CoA by enoyl-CoA are analogous to this given reaction.
(f)
To determine: The type of reaction in which β-hydroxy β-methylglutaryl-CoA is converted to acetyl-CoA and acetoacetate.
Introduction:
The series of metabolic reactions, in which the energy stored in the compounds is released for the production of ATP (adenosine triphosphate), is called “Krebs’s cycle”. The pyruvate undergoes series of reactions in the citric acid cycle and converts into Acetyl CoA (acetyl coenzyme A) in the presence of oxygen.
β-oxidation is the oxidation of fatty acids in cells in absence of glucose and glycogen, to produce ATP. Oxidation takes place at the β carbon, thus, this reaction is termed as β-oxidation.
(f)
Explanation of Solution
Pictorial representation: Fig. 6 represents the reverse aldol reaction.
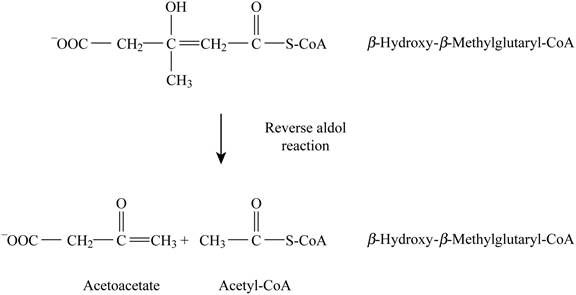
Fig. 6: Reverse aldol reaction.
Explanation:
The formation of acetate-CoA and acetoacetate by β-hydroxy β-methylglutaryl-CoA is a type of reverse aldol reaction. There are no cofactors involved in this reaction. The analogous reaction is the opposite reaction involving citrate synthase. This reaction is analogous to citrate synthase reaction in the citric acid cycle. It is identical to the cleavage of β-hydroxy-β-methylglutaryl-CoA in the formation of
Want to see more full solutions like this?
Chapter 18 Solutions
SaplingPlus for Lehninger Principles of Biochemistry (Six-Month Access)
- Which features of the curves in Figure 30-2 indicates that the enzyme is not consumed in the overall reaction? ES is lower in energy that E + S and EP is lower in energy than E + P. What does this tell you about the stability of ES versus E + S and EP versus E + P.arrow_forwardLooking at the figure 30-5 what intermolecular forces are present between the substrate and the enzyme and the substrate and cofactors.arrow_forwardprovide short answers to the followings Urgent!arrow_forward
- Pyruvate is accepted into the TCA cycle by a “feeder” reaction using the pyruvatedehydrogenase complex, resulting in acetyl-CoA and CO2. Provide a full mechanismfor this reaction utilizing the TPP cofactor. Include the roles of all cofactors.arrow_forwardB- Vitamins are converted readily into important metabolic cofactors. Deficiency inany one of them has serious side effects. a. The disease beriberi results from a vitamin B 1 (Thiamine) deficiency and ischaracterized by cardiac and neurological symptoms. One key diagnostic forthis disease is an increased level of pyruvate and α-ketoglutarate in thebloodstream. How does this vitamin deficiency lead to increased serumlevels of these factors? b. What would you expect the effect on the TCA intermediates for a patientsuffering from vitamin B 5 deficiency? c. What would you expect the effect on the TCA intermediates for a patientsuffering from vitamin B 2 /B 3 deficiency?arrow_forwardDraw the Krebs Cycle and show the entry points for the amino acids Alanine,Glutamic Acid, Asparagine, and Valine into the Krebs Cycle - (Draw the Mechanism). How many rounds of Krebs will be required to waste all Carbons of Glutamic Acidas CO2?arrow_forward
- Sodium fluoroacetate (FCH 2CO2Na) is a very toxic molecule that is used as rodentpoison. It is converted enzymatically to fluoroacetyl-CoA and is utilized by citratesynthase to generate (2R,3S)-fluorocitrate. The release of this product is a potentinhibitor of the next enzyme in the TCA cycle. Show the mechanism for theproduction of fluorocitrate and explain how this molecule acts as a competitiveinhibitor. Predict the effect on the concentrations of TCA intermediates.arrow_forwardIndicate for the reactions below which type of enzyme and cofactor(s) (if any) wouldbe required to catalyze each reaction shown. 1) Fru-6-P + Ery-4-P <--> GAP + Sed-7-P2) Fru-6-P + Pi <--> Fru-1,6-BP + H2O3) GTP + ADP <--> GDP + ATP4) Sed-7-P + GAP <--> Rib-5-P + Xyl-5-P5) Oxaloacetate + GTP ---> PEP + GDP + CO 26) DHAP + Ery-4-P <--> Sed-1,7-BP + H 2O7) Pyruvate + ATP + HCO3- ---> Oxaloacetate + ADP + Piarrow_forwardTPP is also utilized in transketolase reactions in the PPP. Give a mechanism for theTPP-dependent reaction between Xylulose-5-phosphate and Ribose-5-Phosphate toyield Glyceraldehyde-3-phosphate and Sedoheptulose-7-Phosphate.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





