
Chemistry: Principles and Reactions
8th Edition
ISBN: 9781305079373
Author: William L. Masterton, Cecile N. Hurley
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17, Problem 96QAP
Choose the figure that best represents the results after the 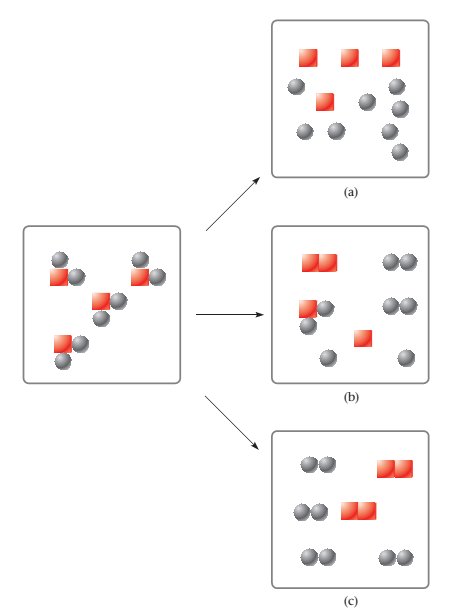
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using Benzene as starting materid show
how each of the Following molecules Contel
Ve syntheswed
CHI
9.
b
-50311
с
CHY
503H
Ночто
d.
อ
•NOV
e
11-0-650
NO2
The molecule PYRIDINE,
6th electrons and is therefore aromatre
and is Assigned the Following structure
contering
Since aromatk moleculoy undergo electrophilic
anomatic substitution, Pyridine shodd undergo
The Following reaction
+ HNO3
12504
a. write all of the possible Mononitration Products
that could Result From this reaction
18. Bared upon the reaction mechanison determime
which of these producty would be the major
Product of the hegetion
a. Explain Why electron withdrawing groups
tend to be meta-Directors. Your answer Should
lyclude all apropriate. Resonance contributing
Structures
fo. Explain why -ll is an outho -tura
drccton even though chlorine has a very High
Electronegativity
Chapter 17 Solutions
Chemistry: Principles and Reactions
Ch. 17 - Balance the following half-equations. Balance (a)...Ch. 17 - Balance the following half-equations. Balance (a)...Ch. 17 - Prob. 3QAPCh. 17 - Balance the following reactions in acid: (a)...Ch. 17 - Write balanced equations for the following...Ch. 17 - Write balanced equations for the following...Ch. 17 - Prob. 7QAPCh. 17 - Write balanced net ionic equations for the...Ch. 17 - Write balanced net ionic equations for the...Ch. 17 - Prob. 10QAP
Ch. 17 - Write a balanced chemical equation for the overall...Ch. 17 - Write a balanced net ionic equation for the...Ch. 17 - Draw a diagram for a salt bridge cell for each of...Ch. 17 - Follow the directions in Question 13 for the...Ch. 17 - Consider a voltaic salt bridge cell represented by...Ch. 17 - Consider a salt bridge voltaic cell represented by...Ch. 17 - Consider a salt bridge cell in which the anode is...Ch. 17 - Follow the directions in Question 17 for a salt...Ch. 17 - Prob. 19QAPCh. 17 - Which species in each pair is the stronger...Ch. 17 - Using Table 17.1, arrange the following reducing...Ch. 17 - Use Table 17.1 to arrange the following oxidizing...Ch. 17 - Consider the following species. Cr3+ Hg(l) H2...Ch. 17 - Follow the directions of Question 23 for the...Ch. 17 - For the following half-reactions, answer these...Ch. 17 - For the following half-reactions, answer the...Ch. 17 - Use Table 17.1 to select (a) a reducing agent in...Ch. 17 - Use Table 17.1 to select (a) an oxidizing agent in...Ch. 17 - Calculate E° for the following voltaic cells: (a)...Ch. 17 - Calculate E° for the following voltaic cells: (a)...Ch. 17 - Using Table 17.1, calculate E° for the reaction...Ch. 17 - Using Table 17.1, calculate E° for the reaction...Ch. 17 - Calculate E° for the following cells: (a)...Ch. 17 - Calculate E° for the following cells: (a)...Ch. 17 - Suppose Ered for Ag+Ag were set equal to zero...Ch. 17 - Suppose Ered for H+H2 were taken to be 0.300 V...Ch. 17 - Which of the following reactions is/are...Ch. 17 - Which of the following reactions is(are)...Ch. 17 - Use the following half-equations to write three...Ch. 17 - Follow the directions of Question 39 for the...Ch. 17 - Use Table 17.1 to answer the following questions:...Ch. 17 - Use Table 17.1 to answer the following questions....Ch. 17 - Write the equation for the reaction, if any, that...Ch. 17 - Write the equation for the reaction, if any, that...Ch. 17 - Prob. 45QAPCh. 17 - Prob. 46QAPCh. 17 - Use Table 17.1 to predict what reaction, if any,...Ch. 17 - Use Table 17.1 to predict what reaction, if any,...Ch. 17 - Consider a cell reaction at 25°C where n=2 . Fill...Ch. 17 - Consider a cell reaction at 25°C where n=4 . Fill...Ch. 17 - For a certain cell, G=25.0 kJ. Calculate E° if n...Ch. 17 - For a certain cell, E=1.08 V. Calculate G° if n is...Ch. 17 - Calculate E°, G°, and K at 25°C for the reaction...Ch. 17 - Calculate E°, G°, and K at 25°C for the reaction...Ch. 17 - Calculate G° at 25°C for each of the reactions...Ch. 17 - Calculate G° at 25°C for each of the reactions...Ch. 17 - Calculate K at 25°C for each of the reactions...Ch. 17 - Calculate K at 25°C for each of the reactions...Ch. 17 - Prob. 59QAPCh. 17 - Use Table 17.1 to find Kffor AuCl4- (aq) at 25°C.Ch. 17 - Prob. 61QAPCh. 17 - What is E° at 25°C for the following reaction?...Ch. 17 - Consider a voltaic cell at 25°C in which the...Ch. 17 - Consider a voltaic cell at 25°C in which the...Ch. 17 - Consider a voltaic cell in which the following...Ch. 17 - Consider a voltaic cell in which the following...Ch. 17 - Calculate the voltages of the following cells at...Ch. 17 - Calculate the voltages of the following cells at...Ch. 17 - Consider the reaction...Ch. 17 - Consider the reaction at 25°C:...Ch. 17 - Complete the following cell notation....Ch. 17 - Complete the following cell notation....Ch. 17 - Consider the reaction below at 25°C:...Ch. 17 - Consider the reaction low at 25°C:...Ch. 17 - Consider a cell in which the reaction is...Ch. 17 - Consider a cell in which the reaction is...Ch. 17 - An electrolytic cell produces aluminum from Al2O3...Ch. 17 - Prob. 78QAPCh. 17 - A solution containing a metal ion (M2+(aq)) is...Ch. 17 - A solution containing a metal ion (M2+(aq)) is...Ch. 17 - A baby's spoon with an area of 6.25 cm2 is plated...Ch. 17 - A metallurgist wants to gold-plate an object with...Ch. 17 - A lead storage battery delivers a current of 6.00...Ch. 17 - Calcium metal can be obtained by the direct...Ch. 17 - Given the following data:...Ch. 17 - In a nickel-cadmium battery (Nicad), cadmium is...Ch. 17 - Hydrogen gas is produced when water is...Ch. 17 - Consider the electrolysis of NiCl2 to Ni(s) and...Ch. 17 - An electrolysis experiment is performed to...Ch. 17 - Prob. 90QAPCh. 17 - Prob. 91QAPCh. 17 - Prob. 92QAPCh. 17 - Atomic masses can be determined by electrolysis....Ch. 17 - Consider the following reaction at 25°C:...Ch. 17 - Given the standard reduction potential for...Ch. 17 - Choose the figure that best represents the results...Ch. 17 - For the cell: Cr|Cr3+Co2+|Co E° is 0.46 V. The...Ch. 17 - Which of the changes below will increase the...Ch. 17 - The standard potential for the reduction of AgSCN...Ch. 17 - Consider the following standard reduction...Ch. 17 - Use Table 17.1 to answer the following questions....Ch. 17 - Consider three metals, X, Y, and Z, and their...Ch. 17 - An alloy made up of tin and copper is prepared by...Ch. 17 - In a fully charged lead storage battery, the...Ch. 17 - Consider a voltaic cell in which the following...Ch. 17 - In biological systems, acetate ion is converted to...Ch. 17 - Consider the cell Pt|H2|H+H+|H2|Pt In the anode...Ch. 17 - Prob. 108QAPCh. 17 - Prob. 109QAP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 9. Write Me product as well as the reaction Mechanism For each of the Following Vanctions +H₂504 4.50+ T C. +212 Fellz 237 b. Praw the potential energy Diagrams For each OF Mese Rauctions and account For any differences that appear in the two potential Puergy Diagrams which of here two reactions 19 Found to be Reversable, Rationalice your answer based upon the venation mechanisms and the potential energy diagrams.arrow_forward9. Write Me product as well as the reaction Mechanism For each of the Following Veritious +H2504 4.50+ + 1/₂ Felly ◎+ 7 b. Praw he potential energy Diagrams For each OF Mese Ronctions and account for any differences that appeak in the two potential Puergy Diagramsarrow_forwardDraw the major product of this reaction. Ignore inorganic byproducts. Incorrect, 3 attempts remaining 1. excess Br2, NaOH 2. neutralizing workup Qarrow_forward
- Given the electrode Pt | Ag | Ag+ (aq), describe it.arrow_forwardAt 25°C, the reaction Zn2+ + 2e ⇄ Zn has a normal equilibrium potential versus the saturated calomel electrode of -1.0048 V. Determine the normal equilibrium potential of Zn versus the hydrogen electrode.Data: The calomel electrode potential is E° = 0.2420 V versus the normal hydrogen electrode.arrow_forwardElectrochemistry. State the difference between E and E0.arrow_forward
- In an electrolytic cell, the positive pole is always assumed to be on the right side of the battery notation. Is that correct?arrow_forwardIn an electrolytic cell, the positive pole is always assumed to be on the right side of the battery. Is that correct?arrow_forwardCalculate the free energy of formation of 1 mol of Cu in cells where the electrolyte is 1 mol dm-3 Cu2+ in sulfate solution, pH 0. E° for the Cu2+/Cu pair in this medium is +142 mV versus ENH.Assume the anodic reaction is oxygen evolution.Data: EH2 = -0.059 pH (V) and EO2 = 1.230 - 0.059 pH (V); 2.3RT/F = 0.059 Varrow_forward
- If the normal potential for the Fe(III)/Fe(II) pair in acid at zero pH is 524 mV Hg/Hg2Cl2 . The potential of the saturated calomel reference electrode is +246 mV versus the NHE. Calculate E0 vs NHE.arrow_forwardGiven the galvanic cell whose scheme is: (-) Zn/Zn2+ ⋮⋮ Ag+/Ag (+). If we know the normal potentials E°(Zn2+/Zn) = -0.76V and E°(Ag+/Ag) = 0.799 V. Indicate the electrodes that are the anode and the cathode and calculate the E0battery.arrow_forwardIndicate the functions that salt bridges have in batteries.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Electrolysis; Author: Tyler DeWitt;https://www.youtube.com/watch?v=dRtSjJCKkIo;License: Standard YouTube License, CC-BY