
Introduction To Chemistry
5th Edition
ISBN: 9781259911149
Author: BAUER, Richard C., Birk, James P., Marks, Pamela
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 16, Problem 97QP
Interpretation Introduction
Interpretation:
The given equation for the reaction of an amine with hydrochloric acid is to be completed.
Concept Introduction:
The general structure of an amine is as follows:
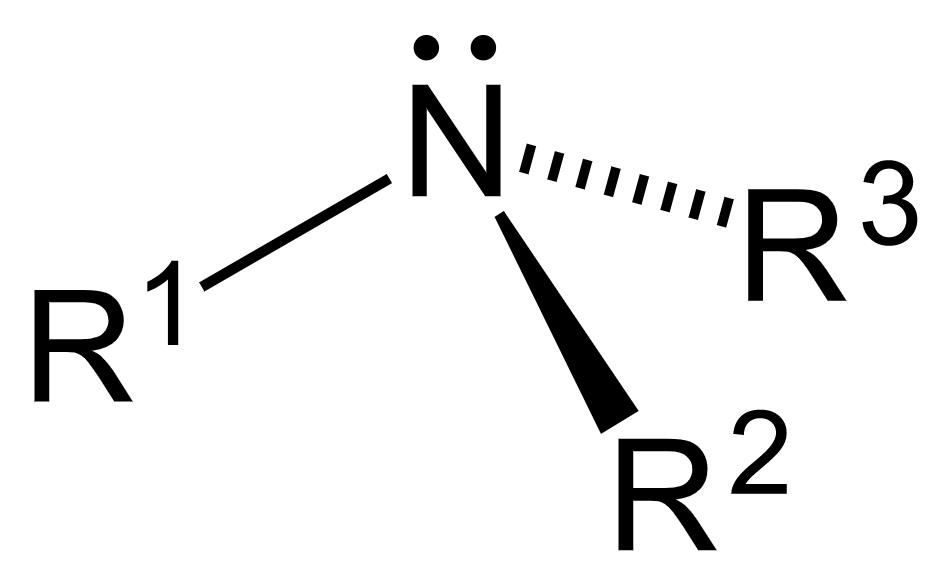
It has a nitrogen atom with a lone pair of electrons and three substituents. The amines act as bases due to the presence of an unshared electron pair on the nitrogen atom.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Label each of the seven designated regions of the following multi-component, solid-liquid phase diagram for the Zinc - Magnesium system.
22
PLEASE READ!!! I DONT WANT EXAMPLES, I DONT WANT WORDS OR PARAGRAPHS FOR THE MECHANISM!!! THANKS
First image: QUESTION 6. I have to show, with ARROWS and STRUCTURES, the mechanism of the reaction at the bottom. Also I have to show by mecanism why the reaction wouldn't work if the alcohol was primary.
I also tried to draw the mechanism, tell me what to change. Please note that its an AMIDE thats formed not an AMINE the nitrogen has ONE hydrogen and one Phenyl-C-Phenyl. I already asked for this mechanism and got as a final product ...-NH2 not whats shown on the picture, thank you
Ths second part. QUESTION 3. I just need a way to synthesize the lactone A, I already started please continue from where I left it
Second image: I simply need the products, substrates or reagents, thank you
Chapter 16 Solutions
Introduction To Chemistry
Ch. 16 - Prob. 1QCCh. 16 - Prob. 2QCCh. 16 - Prob. 3QCCh. 16 - What are cyclic hydrocarbons, and how do they...Ch. 16 - Prob. 5QCCh. 16 - Prob. 6QCCh. 16 - Prob. 7QCCh. 16 - Prob. 8QCCh. 16 - Prob. 9QCCh. 16 - Prob. 1PP
Ch. 16 - Prob. 2PPCh. 16 - Prob. 3PPCh. 16 - Prob. 4PPCh. 16 - Prob. 5PPCh. 16 - Prob. 6PPCh. 16 - Prob. 7PPCh. 16 - Prob. 8PPCh. 16 - Prob. 9PPCh. 16 - Prob. 10PPCh. 16 - Prob. 11PPCh. 16 - Prob. 1QPCh. 16 - Prob. 2QPCh. 16 - Prob. 3QPCh. 16 - Prob. 4QPCh. 16 - Prob. 5QPCh. 16 - Prob. 6QPCh. 16 - Prob. 7QPCh. 16 - Prob. 8QPCh. 16 - Prob. 9QPCh. 16 - Prob. 10QPCh. 16 - Prob. 11QPCh. 16 - Prob. 12QPCh. 16 - Prob. 13QPCh. 16 - Prob. 14QPCh. 16 - Prob. 15QPCh. 16 - Prob. 16QPCh. 16 - Prob. 17QPCh. 16 - Prob. 18QPCh. 16 - Prob. 19QPCh. 16 - Prob. 20QPCh. 16 - Prob. 21QPCh. 16 - Prob. 22QPCh. 16 - Prob. 23QPCh. 16 - Prob. 24QPCh. 16 - Prob. 25QPCh. 16 - Prob. 26QPCh. 16 - Prob. 27QPCh. 16 - Prob. 28QPCh. 16 - Prob. 29QPCh. 16 - Prob. 30QPCh. 16 - Prob. 31QPCh. 16 - Prob. 32QPCh. 16 - Prob. 33QPCh. 16 - Prob. 34QPCh. 16 - Prob. 35QPCh. 16 - Prob. 36QPCh. 16 - Prob. 37QPCh. 16 - Prob. 38QPCh. 16 - Prob. 39QPCh. 16 - Prob. 40QPCh. 16 - Prob. 41QPCh. 16 - Write IUPAC names for the following compounds....Ch. 16 - Prob. 43QPCh. 16 - Prob. 44QPCh. 16 - Prob. 45QPCh. 16 - Prob. 46QPCh. 16 - Prob. 47QPCh. 16 - Prob. 48QPCh. 16 - Prob. 49QPCh. 16 - Prob. 50QPCh. 16 - Prob. 51QPCh. 16 - Prob. 52QPCh. 16 - Prob. 53QPCh. 16 - Prob. 54QPCh. 16 - Prob. 55QPCh. 16 - Prob. 56QPCh. 16 - Prob. 57QPCh. 16 - Prob. 58QPCh. 16 - Prob. 59QPCh. 16 - Prob. 60QPCh. 16 - Prob. 61QPCh. 16 - Prob. 62QPCh. 16 - Prob. 63QPCh. 16 - Prob. 64QPCh. 16 - Prob. 65QPCh. 16 - Prob. 66QPCh. 16 - Prob. 67QPCh. 16 - Prob. 68QPCh. 16 - Prob. 69QPCh. 16 - Prob. 70QPCh. 16 - Prob. 71QPCh. 16 - Prob. 72QPCh. 16 - Prob. 73QPCh. 16 - Prob. 74QPCh. 16 - Prob. 75QPCh. 16 - Prob. 76QPCh. 16 - Prob. 77QPCh. 16 - Prob. 78QPCh. 16 - Prob. 79QPCh. 16 - Prob. 80QPCh. 16 - Prob. 81QPCh. 16 - Prob. 82QPCh. 16 - Prob. 83QPCh. 16 - Prob. 84QPCh. 16 - Prob. 85QPCh. 16 - Prob. 86QPCh. 16 - Prob. 87QPCh. 16 - Prob. 88QPCh. 16 - Predict and explain the relative boiling points of...Ch. 16 - Prob. 90QPCh. 16 - Prob. 91QPCh. 16 - Prob. 92QPCh. 16 - Prob. 93QPCh. 16 - Prob. 94QPCh. 16 - Prob. 95QPCh. 16 - Prob. 96QPCh. 16 - Prob. 97QPCh. 16 - Prob. 98QPCh. 16 - Prob. 99QPCh. 16 - Prob. 100QPCh. 16 - Prob. 101QPCh. 16 - Prob. 102QPCh. 16 - Prob. 103QPCh. 16 - Prob. 104QPCh. 16 - Prob. 105QPCh. 16 - Prob. 106QPCh. 16 - Prob. 107QPCh. 16 - Prob. 108QPCh. 16 - Prob. 109QPCh. 16 - Prob. 110QPCh. 16 - Prob. 111QPCh. 16 - Prob. 112QPCh. 16 - Prob. 113QPCh. 16 - Prob. 114QPCh. 16 - Prob. 115QPCh. 16 - Prob. 116QPCh. 16 - Prob. 117QPCh. 16 - Prob. 118QPCh. 16 - Prob. 119QPCh. 16 - Prob. 120QPCh. 16 - Prob. 121QPCh. 16 - Draw the line structures for the ether and two...Ch. 16 - Prob. 123QPCh. 16 - Prob. 124QPCh. 16 - Prob. 125QPCh. 16 - Prob. 126QPCh. 16 - Write structural formulas for all the isomers of...Ch. 16 - Prob. 128QPCh. 16 - Prob. 129QPCh. 16 - Prob. 130QPCh. 16 - Prob. 131QPCh. 16 - Prob. 132QPCh. 16 - Prob. 133QPCh. 16 - Prob. 134QPCh. 16 - Prob. 135QPCh. 16 - Prob. 136QPCh. 16 - Prob. 137QPCh. 16 - Prob. 138QPCh. 16 - Prob. 139QPCh. 16 - Prob. 140QPCh. 16 - Prob. 141QPCh. 16 - Prob. 142QPCh. 16 - Prob. 143QP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Indicate how to prepare a 10% sodium hydroxide (NaOH) solution to a slightly alkaline pH.arrow_forwardCH, CH CH₂ CH₂ Phytyl side chain 5. What is the expected order of elution of compounds A-D below from a chromatography column packed with silica gel, eluting with hexane/ethyl acetate? C D OHarrow_forwardPlease analze my gel electrophoresis column of the VRK1 kinase (MW: 39.71 kDa). Attached is the following image for the order of column wells and my gel.arrow_forward
- 2.0arrow_forwardWrite the electron configuration of an atom of the element highlighted in this outline of the Periodic Table: 1 23 4 5 6 7 He Ne Ar Kr Xe Rn Hint: you do not need to know the name or symbol of the highlighted element! ☐arrow_forwardCompare these chromatograms of three anti-psychotic drugs done by HPLC and SFC. Why is there the difference in separation time for SFC versus HPLC? Hint, use the Van Deemter plot as a guide in answering this question. Why, fundamentally, would you expect a faster separation for SFC than HPLC, in general?arrow_forward
- A certain inorganic cation has an electrophoretic mobility of 5.27 x 10-4 cm2s-1V-1. The same ion has a diffusion coefficient of 9.5 x 10-6cm2s-1. If this ion is separated from cations by CZE with a 75cm capillary, what is the expected plate count, N, at an applied voltage of 15.0kV? Under these separation conditions, the electroosmotic flow rate was 0.85mm s-1 toward the cathode. If the detector was 50.0cm from the injection end of the capillary, how long would it take in minutes for the analyte cation to reach the detector after the field was applied?arrow_forward2.arrow_forwardPlease solve for the following Electrochemistry that occursarrow_forward
- Commercial bleach contains either chlorine or oxygen as an active ingredient. A commercial oxygenated bleach is much safer to handle and less likely to ruin your clothes. It is possible to determine the amount of active ingredient in an oxygenated bleach product by performing a redox titration. The balance reaction for such a titration is: 6H+ +5H2O2 +2MnO4- à 5O2 + 2Mn2+ + 8H2O If you performed the following procedure: “First, dilute the Seventh Generation Non-Chlorine Bleach by pipetting 10 mL of bleach in a 100 mL volumetric flask and filling the flask to the mark with distilled water. Next, pipet 10 mL of the diluted bleach solution into a 250 mL Erlenmeyer flask and add 20 mL of 1.0 M H2SO4 to the flask. This solution should be titrated with 0.0100 M KMnO4 solution.” It took 18.47mL of the KMnO4 to reach the endpoint on average. What was the concentration of H2O2 in the original bleach solution in weight % assuming the density of bleach is 1g/mL?arrow_forward10.arrow_forwardProper care of pH electrodes: Why can you not store a pH electrode in distilled water? What must you instead store it in? Why?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning  Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning