
Essential University Physics: Volume 1 (3rd Edition)
3rd Edition
ISBN: 9780321993724
Author: Richard Wolfson
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 16, Problem 46P
A circular lake 1.0 km in diameter is 10 m deep (Fig. 16.14). Solar energy is incident on the lake at an average rate of 200 W/m2. If the lake absorbs all this energy and does not exchange heat with its surroundings, how long will it take to warm from 10°C to 20°C?
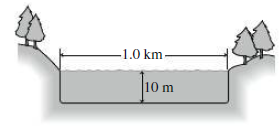
FIGURE 16.14 Problem 46
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Can someone help me
Need help on the following questions on biomechanics. (Please refer to images below)A gymnast weighing 68 kg attempts a handstand using only one arm. He plants his handat an angle resulting in the reaction force shown.A) Find the resultant force (acting on the Center of Mass)B) Find the resultant moment (acting on the Center of Mass)C) Draw the resultant force and moment about the center of mass on the figure below. Will the gymnast rotate, translate, or both? And in which direction?
Please help me on the following question (Please refer to image below)An Olympic lifter (m = 103kg) is holding a lift with a mass of 350 kg. The barexerts a purely vertical force that is equally distributed between both hands. Each arm has amass of 9 kg, are 0.8m long and form a 40° angle with the horizontal. The CoM for each armis 0.5 m from hand. Assuming the lifter is facing us in the diagram below, his right deltoidinserts 14cm from the shoulder at an angle of 13° counter-clockwise from the humerus.A) You are interested in calculating the force in the right deltoid. Draw a free body diagramof the right arm including the external forces, joint reaction forces, a coordinate system andstate your assumptions.B) Find the force exerted by the right deltoidC) Find the shoulder joint contact force. Report your answer using the magnitude and directionof the shoulder force vector.
Chapter 16 Solutions
Essential University Physics: Volume 1 (3rd Edition)
Ch. 16.1 - Is there (a) no temperature, (b) one temperature,...Ch. 16.2 - A hot rock with mass 250 g is dropped into an...Ch. 16.3 - The figure shows three slabs with the same...Ch. 16.3 - Prob. 16.4GICh. 16.4 - A houses thermostat fails, leaving the furnace...Ch. 16 - If system A is not in thermodynamic equilibrium...Ch. 16 - Does a thermometer measure its own temperature or...Ch. 16 - Compare the relative sizes of the kelvin, the...Ch. 16 - If you put a thermometer in direct sunlight, what...Ch. 16 - Why does the temperature in a stone building...
Ch. 16 - Why do large bodies of water exert a...Ch. 16 - A Thermos bottle consists of an evacuated,...Ch. 16 - Stainless-steel cookware often has a layer of...Ch. 16 - Prob. 9FTDCh. 16 - Prob. 10FTDCh. 16 - Glass and fiberglass are made from the same...Ch. 16 - To keep your hands warm while skiing, you should...Ch. 16 - Since Earth is exposed to solar radiation, why...Ch. 16 - Global warming at Earths surface is generally...Ch. 16 - In its 2014 report, the Intergovernmental Panel on...Ch. 16 - A Canadian meteorologist predicts an overnight low...Ch. 16 - Normal room temperature is 68F. Whats this in...Ch. 16 - Prob. 18ECh. 16 - At what temperature do the Fahrenheit and Celsius...Ch. 16 - The normal boiling point of nitrogen is 77.3 K....Ch. 16 - Prob. 21ECh. 16 - Prob. 22ECh. 16 - Prob. 23ECh. 16 - Whats the specific heat of a material if it takes...Ch. 16 - The average human diet contains about 2000 kcal...Ch. 16 - Prob. 26ECh. 16 - You bring a 350-g wrench into the house from your...Ch. 16 - Prob. 28ECh. 16 - Building heat loss in the United States is usually...Ch. 16 - Find the heat-loss rate through a slab of (a) wood...Ch. 16 - The top of a steel wood stove measures 90 cm by 40...Ch. 16 - Youre a builder whos advising a homeowner to have...Ch. 16 - An 8.0 m by 12 m house is built on a concrete slab...Ch. 16 - Find the -factor for a wall that loses 0.040 Btu...Ch. 16 - Compute the -factors for 1-inch thicknesses of...Ch. 16 - A horseshoe has surface area 50 cm2, and a...Ch. 16 - An oven loses energy at the rate of 14 W per C...Ch. 16 - Youre having your homes heating system replaced,...Ch. 16 - The filament of a 100-W lightbulb is at 3.0 kK....Ch. 16 - A typical human body has surface area 1.4 nr and...Ch. 16 - A constant-volume gas thermometer is filled with...Ch. 16 - A constant-volume gas thermometer is at 55-kPa...Ch. 16 - In Fig. 16.2s gas thermometer, the height h is...Ch. 16 - Prob. 44PCh. 16 - Typical fats contain about 9 kcal per gram. If the...Ch. 16 - A circular lake 1.0 km in diameter is 10 m deep...Ch. 16 - How much heat is required to raise an 800-g copper...Ch. 16 - Initially, 100 g of water and 100 g of another...Ch. 16 - Prob. 49PCh. 16 - Two neighbors return from Florida to find their...Ch. 16 - Prob. 51PCh. 16 - Prob. 52PCh. 16 - Prob. 53PCh. 16 - The temperature of the eardrum provides a reliable...Ch. 16 - Prob. 55PCh. 16 - Your young niece complains that her cocoa, at 90C,...Ch. 16 - A piece of copper at 300C is dropped into 1.0 kg...Ch. 16 - While camping, you boil water to make spaghetti....Ch. 16 - A biology labs walk-in cooler measures 3.0 m by...Ch. 16 - One end of an iron rod 40 cm long and 3.0 cm in...Ch. 16 - Prob. 61PCh. 16 - An electric stove burner has surface area 325 cm2...Ch. 16 - An electric current passes through a metal strip...Ch. 16 - Youre considering purchasing a new sleeping bag...Ch. 16 - A blacksmith heats a 1.1-kg iron horseshoe to...Ch. 16 - Whats the power output of a microwave oven that...Ch. 16 - A cylindrical log 15 cm in diameter and 65 cm long...Ch. 16 - A blue giant star whose surface temperature is 23...Ch. 16 - Prob. 69PCh. 16 - A black wood stove with surface area 4.6 nr is...Ch. 16 - Estimate the average temperature on Pluto,...Ch. 16 - Prob. 72PCh. 16 - Prob. 73PCh. 16 - Prob. 74PCh. 16 - Prob. 75PCh. 16 - In a cylindrical pipe where area isnt constant....Ch. 16 - Prob. 77PCh. 16 - Prob. 78PCh. 16 - Prob. 79PCh. 16 - Use the method outlined in Problem 76 to show that...Ch. 16 - A house is at 20C on a winter night when the...Ch. 16 - A more realistic approach to the solar greenhouse...Ch. 16 - Fiberglass is a popular, economical, and fairly...Ch. 16 - Fiberglass is a popular, economical, and fairly...Ch. 16 - Fiberglass is a popular, economical, and fairly...Ch. 16 - Fiberglass is a popular, economical, and fairly...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Plants use the process of photosynthesis to convert the energy in sunlight to chemical energy in the form of su...
Campbell Essential Biology (7th Edition)
What properties do all types of epithelia share?
Campbell Biology (11th Edition)
Draw the enol tautomers for each of the following compounds. For compounds that have more than one enol tautome...
Organic Chemistry (8th Edition)
In a population, what is the consequence of inbreeding? Does inbreeding change allele frequencies? What is the ...
Genetic Analysis: An Integrated Approach (3rd Edition)
A source of electromagnetic radiation produces infrared light. Which of the following could be the wavelength ...
Chemistry: The Central Science (14th Edition)
How can the freezing of water crack boulders?
Campbell Biology in Focus (2nd Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Similar questions
- I need help with part B. I cant seem to get the correct answer. Please walk me through what youre doing to get to the answer and what that could bearrow_forwardQuestion 6: Chlorine is widely used to purify municipal water supplies and to treat swimming pool waters. Suppose that the volume of a particular sample of Cl₂ gas is 8.70 L at 895 torr and 24°C. (a) How many grams of Cl₂ are in the sample? ⚫ Atomic mass of CI = 35.453 g/mol • Molar mass of Cl₂ = 2 x 35.453 = 70.906 g/mol Solution: Use the Ideal Gas Law: Step 1: Convert Given Values • Pressure: P = 895 torr → atm PV= = nRT 1 P = 895 × = 1.1789 atm 760 • Temperature: Convert to Kelvin: T24273.15 = 297.15 K • Gas constant: R = 0.0821 L atm/mol. K Volume: V = 8.70 L Step 2: Solve for n . PV n = RT n = (1.1789)(8.70) (0.0821)(297.15) 10.25 n = = 0.420 mol 24.405 Step 3: Calculate Mass of Cl₂ Final Answer: 29.78 g of Cl₂. mass nx M mass= (0.420)(70.906) mass= 29.78 garrow_forwardE1 R₁ w 0.50 20 Ω 12 R₁₂ ww ΒΩ R₂ 60 E3 C RA w 15 Ω E2 0.25 E4 0.75 Ω 0.5 Ωarrow_forward
- What is the force (in N) on the 2.0 μC charge placed at the center of the square shown below? (Express your answer in vector form.) 5.0 με 4.0 με 2.0 με + 1.0 m 1.0 m -40 με 2.0 μCarrow_forwardWhat is the force (in N) on the 5.4 µC charge shown below? (Express your answer in vector form.) −3.1 µC5.4 µC9.2 µC6.4 µCarrow_forwardAn ideal gas in a sealed container starts out at a pressure of 8900 N/m2 and a volume of 5.7 m3. If the gas expands to a volume of 6.3 m3 while the pressure is held constant (still at 8900 N/m2), how much work is done by the gas? Give your answer as the number of Joules.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781305952300Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:9781133104261
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

Physics for Scientists and Engineers: Foundations...
Physics
ISBN:9781133939146
Author:Katz, Debora M.
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781285737027
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning

Physics for Scientists and Engineers, Technology ...
Physics
ISBN:9781305116399
Author:Raymond A. Serway, John W. Jewett
Publisher:Cengage Learning

College Physics
Physics
ISBN:9781938168000
Author:Paul Peter Urone, Roger Hinrichs
Publisher:OpenStax College

College Physics
Physics
ISBN:9781305952300
Author:Raymond A. Serway, Chris Vuille
Publisher:Cengage Learning
Heat Transfer: Crash Course Engineering #14; Author: CrashCourse;https://www.youtube.com/watch?v=YK7G6l_K6sA;License: Standard YouTube License, CC-BY